Summary
Protein content, dry weight, cell viability, and the ability of cultures to be subcultured were measured at 4-d intervals beginning on Day 14 when cell division and expansion had stopped. Measurements were continued until the cultures had died.
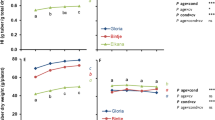
The protein content of senescing cultures decreased from approximately 780 to 500 µg between Days 14 and 18, then remained relatively constant until Day 26 whereupon it declined to approximately 350 µg/culture. The decline in protein content of parent cultures did not correlate well with cell viability and their ability to be subcultured at some ages.
In older cultures it was estimated that less than 2000 of the approximately 261×106 cells appeared to be capable of dividing when they were subcultured.
Similar content being viewed by others
References
King, P. J.; Street, H. E. Growth patterns in cell cultures. Street, H. E. ed. Plant tissue and cell culture. Berkeley: University of California Press; 1977: 307–387.
Condron, H.; Latche, A.; Pech, J. C.; Fallot, J. Mise au point d’un nouveau systeme d’etude de la senescence des cellules vegetales. Compt. Rend. Acad. Sci. Paris 287: 21–24; 1978.
Condron, H.; Latche, A.; Pech, J. C.; Nebie, B.; Fallot, J. 1979. Control of quiescence and viability in auxin-deprived pear cells in batch and continuous culture. Plant Sci. Lett. 17: 29–35; 1979.
Okamura, S.; Sueki, K.; Nishi, A. Physiological changes of carrot cells in suspension culture during growth and senescence. Physiol. Plant. 33: 251–255; 1975.
Pech, J. C.; Romani, R. J. Senescence of pear fruit cells cultured in a continuously renewed, auxin-deprived medium. Plant Physiol. 64: 814–817; 1979.
Nooden, L. D. Senescence in the whole plant. Thimann, K. V. ed. Senescence in plants. Boca Raton, Florida: CRC Press Inc.; 1980: 219–258.
Nesius, K. K.; Uchytil, L. E.; Fletcher, J. S. Minimal organic medium for suspension cultures of Paul’s Scarlet rose. Planta 106: 173–176; 1972.
Mohanty, B.; Fletcher, J. S. Influence of ammonium on the growth and development of suspension cultures of Paul’s Scarlet rose. Physiol. Plant. 42: 221–225; 1978.
Widholm, J. M. The use of fluorescein diacetate and phenosafranine for determining viability of cultured plant cells. Stain Technol. 47: 189–194; 1972.
Danks, M. L.; Fletcher, J. S.; Rice, E. L. Effects of phenolic inhibitors on growth and metabolism of glucose-UL-14C in Paul’s Scarlet rose cell-suspension cultures. Am. J. Bot. 62: 311–317; 1973.
Nesius, K. K.; Fletcher, J. S. Carbon dioxide and pH requirements of non-photosynthetic tissue culture cells. Physiol. Plant. 28: 259–263; 1973.
Street, H. E.; Henshaw, G. G. Cell division and differentiation in suspension cultures of higher plant cells. Symp. Soc. Exp. Biol. 17: 234–256; 1963.
Henshaw, G. G.; Jha, K. K.; Mehta, A. R.; Shakeshaft, D. J.; Street, H. E. Studies on the growth in culture of plant cells. I. Growth patterns in batch propagated suspension cultures. J. Exp. Bot. 17: 362–377; 1962.
Stuart, R.; Street, H. E. Studies on the growth in culture of plant cells. IV. The initiation of division of stationary-phase cells ofAcer pseudoplantus L. J. Exp. Bot. 20: 556–571; 1969.
Author information
Authors and Affiliations
Additional information
This work was supported by National Institute of Health Grant 1 R01 AG01709-02.
Rights and permissions
About this article
Cite this article
Reider, M.L., Smith, B.A. & Fletcher, J.S. Protein content and subculturing properties of senescing plant suspension cultures. In Vitro Cell.Dev.Biol.-Plant 18, 1004–1008 (1982). https://doi.org/10.1007/BF02796375
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02796375