Abstract
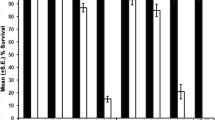
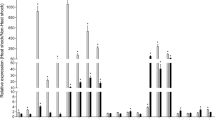
The selective inducibility ofhsrω gene by heat shock and several chemical agents and its selective non-inducibility by heat shock under certain conditions led to suggestion that this locus is subject to multiple controls at the level of transcription. With a view to delimit these different control elements, transgenic lines horbouringhsrω 5’ promoter deletion variants tagged to thelacZ reporter gene were used. Three different assays, viz., staining forβ-galactosidase activity in different larval tissues using chromogenic X-gal substrate, [3H] uridine labelling of polytene nuclei andin situ DNA-DNA hybridization with a non-radioactive probe to polytene chrmosome spreads for checking the puffing status of the resident and the transgene in larval salivary glands, were applied to monitor the activiy of the reporter gene following different treatments. Our results showed that the − 844 bp to +107 bp sequence was sufficient for heat shock induction of the transgene in all tissues. An analysis of the base sequence of thehsrω promoter revealed the presence of three consensus heat shock elements at − 466, − 250 and at − 57 bp and of two GAGA factor binding sites at − 496 and at − 68bp within the − 844 bp region. Germline transformants carrying the − 346 bp to − 844 bp region of thehsrω promoter showed only a very weak heat shock inducibility of the reporter gene in agreement with the presence of only one of the three putative heat shock elements and one of the two GAGA factor binding sites in this region. Interestingly, neither of the transformed lines (carrying the − 844 bp to + 107 bp or the − 844 bp to −346 bp of thehsrω promoter region) showed any response of the transgene to benzamide or colchicine treatments. These results showed that while the heat shock response elements of thehsrω are included within the − 844 bp region the response elements for benzamide and colchicine treatments are outside this region.
Similar content being viewed by others
References
Amin J J, Ananthan J and Voellmy R 1988 Key features of heat shock regulatory elements;Mol. Cell. Biol. 8 3761–3769
Beermann W 1967 Gene action at the level of the chromosome; in;Heritage from Mendel (ed.) R A Brink (Madison, Wisconsin: University of Wisconsin Press) pp 179–201
Fernandes M, O’Brien T and Lis J T 1994 Structure and regulation of heat shock gene promoters; inThe biology of heat shock proteins and molecular chaperones (ed.) R I Morimoto (Cold Spring Harbor, New York: Cold Spring Harbor Press) pp 375–393
Garbe J C, Bendena W H and Pardue M L 1989 Sequence evolution of theDrosophila heat shock locus hsr-omega. The non-repeated portion of the gene;Genetics 122 403–415
Granok H, Leibovitch B A, Schaffer C D and Elgin SCR 1995 Ga-ga over GAGA factor;Curr. Biol. 5 238–241
Hovemann B, Walldorf U and Ryseck R P 1986 Heat shock locus of93D of Drosophila melanogaster. an RNA with limited coding capacity accumulates precursor transcripts after heat shock:Mol. Gen. Genet. 204 334–340
Lakhotia S C 1987 The93D heat shock locus inDrosophila: a review;J. Genet. 66 139–157
Lakhotia S C 1989 The93D heat shock locus ofDrosophila melanogaster. modulation by genetic and developmental factors;Genome 31 677–683
Lakhotia S C, KarChowdhuri D and Burma P K 1990 Mutations affecting β-alanine metabolism influence inducibility of the 93D puff by heat shock inDrosophila melanogaster, Chromosoma 99 296–305
Lakhotia S C and MukherjeeT 1980 Specific activation of puff 93D ofDrosophila melanogaster by benzamide and the effect of benzamide treatment on the heat shock puffing activity;Chromosoma 81 125–136
Lakhotia S C and Sharma A 1995 RNA metabolismin situ at the 93D heat shock locus in polytene nuclei ofDrosophila melanogaster after various treatments;Chromosome Res. 3 151–161
Lakhotia S C and Sharma A 1996 The93D (hsrω) locus ofDrosophila: non-coding gene with house-keeping functions;Genetica (in press)
Lakhotia S C and Singh A K 1989 A novel set of heat shock polypeptides in Malpighian tubules ofDrosophila melanogaster, J. Genet. 68 129–137
Lindsley D L and Zimm G G 1992The genome of Drosophila melanogaster (San Diego: Academic Press)
Lis J and Wu C 1993 Protein traffic on the heat shock promoter: parking, stalling and trucking along;Cell 74 1–4
Lu Q, Wallrath L L, Granok H and Elgin S C R 1993 Distinct role of (CT)n (GA)n repeats and heat shock elements in chromatin structure and transcriptional activation of theDrosophila hsp26 gene;Mol. Cell Biol. 13 2802–2814
Mukherjee T and Lakhotia S C 19793H-uridine incorporation in the puff93D and in chromocentric heterochromatin of heat shocked salivary glands ofDrosophila melanogaster, Chromosoma 74 75–82
Mutsuddi M and Lakhotia S C 1995 Spatial expression of thehsr-omega (93D) gene in different tissues ofDrosophila melanogaster and identification of promoter elements controlling its development;Dev. Genet. 17 301–311
Pelham H R B 1982 A reulatory upstream promoter element in theDrosophila hsp70 heat shock gene;Cell 30 517–528
Perisic O, Xiao H and Lis J T 1989 Stable binding ofDosophila heat shock factor to-head-to head and tail-to-tail repeats of a conserved 5 bp recognition unit;Cell 59 797–806
Singh B N and Lakhotia S C 1995 The non-induction of hsp70 in heat shocked Malpighian tubules ofDrosophila larvae is not due to consitutive presence of hsp70 or hsc70;Curr. Sci. 69 178–182
SrinivasU, Swamynathan S K 1996 Role of heat shock transcription factors in stress response and during development;J. Biosci. 21 103–121
Tapadia M G and Lakhotia S C 1993 Effect of a P-element mediated deletion on induction of93D heat shock locus ofDrosophila melanogaster by benzamide and colchicine;Abstr. Drosophila Meeting (University of Mysore, India) p 27
Vazquez J, Pauli D and Tissieres A 1993 Transcription regulation inDrosophila during heat shock: a nuclear run-on analysis;Chromosoma 102 233–248
Westwood J T, Clos J and Wu C 1991 Siress induced oligomerisation and chromosomal relocalisiition of heat shock factor;Nature (London) 353 822–827
Wu C, Clos J, Giorgi G, Haroun R I, Kim S J, Rabindran S K, Westwood J T, Wisniewski J and Yim G 1994 Structure and regulation of heat shock transcription factor; inThe biology of heat shock proteins and molecular chaperones (ed.) R I Morimoto (Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press) pp 395–416
Xiao H and Lis J T 1988 Germline transformation used to define key features of heat shock response element;Science 239 1139–1142
Xiao H, Olga P and Lis J T 1991 Co-operative binding ofDrosophila heat shock factor to arrays of a conserved 5 bp unit;Cell 64 585–593
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lakhotia, S.C., Mutsuddi, M. Heat shock but not benzamide and colchicine response elements are present within the — 844 bp upstream region of thehrsω gene ofDrosophila melanogaster . J Biosci 21, 235–246 (1996). https://doi.org/10.1007/BF02703111
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02703111