Summary
A method is presented for quantitative determination of terbutaline after administration of its prodrug Bambuterol. Terbutaline is extracted from plasma by liquid-solid extraction on small C18-cartridges. The extract is then analysed by coupled column liquid chromatography with amperometric detection.
To inhibit the esterase catalyzed hydrolysis of bambuterol to terbutaline an analogue of bambuterol is added to the plasma sampling tubes.
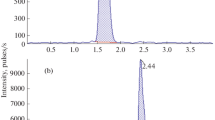
The within-day variation on spiked samples were 2.1% and 2.4% at 8.0 and 40.0 nmol/L respectively. The between-day variation on spiked samples (8.0 nmol/L) was 3.6% and on authentic samples 5.9% at the 11 nmol/L level.
The absolute recovery was in the range 83–94% for terbutaline and the internal standard. The limit of quantitation was set at 4.0 nmol/L (Cv=3.4%, n=46).
Similar content being viewed by others
References
S.-E. Jacobsson, S. Jönsson, C. Lindberg, L.-Å. Svensson, Biomed. Mass Spectrom.7, 265 (1980).
L.-Å. Svensson, A. Tunek, Drug Metab. Rev. In Press.
L.-E. Edholm, B.-M. Kennedy, S. Bergqvist, Chromatographia,16, 341 (1982).
L.-E. Edholm, L. Ögren, in “Liquid Chromatography in Pharmaceutical Development”, I. Wainer, ed., Aster, Springfield, Oregon, 1986; p. 323.
S. Bergqvist, L.-E. Edholm, J. Liq. Chromatogr.,6 (3), 559 (1983).
Author information
Authors and Affiliations
Additional information
Subsidiary of AB Astra.
Rights and permissions
About this article
Cite this article
Kennedy, B.M., Blomgren, A., Edholm, L.E. et al. Quantitative determination of terbutaline in human plasma after administration of bambuterol using coupled columns and electrochemical detection. Chromatographia 24, 895–899 (1987). https://doi.org/10.1007/BF02688606
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02688606