Abstract
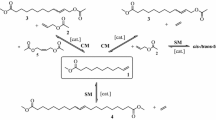
The reaction of methyl 4-hydroxy-trans-2-hexadecenoate [1b] with diiodomethane in the presence of zinc-copper couple yielded methyl 4-methoxy-trans-2,3-methylenehexadecanoate [2] in a 70% yield, together with methyl 4-hydroxy-trans-2,3-methylenehexadecanoate [3] (∼20%). The presence of an allylic hydroxyl group in the α,β-unsaturated ester increases the yield of the cyclopropanation product. The formation ofo-methyl ether reveals the dual role of cyclopropanation and etherification by S.S. reagent of a hydroxylated olefinic compound.
Similar content being viewed by others
References
Gunstone, F.D., M. Lie Ken Jie, and R.T. Wall, Chem. Phys. Lipids 6:147 (1971).
Wood, R. and R. Reiser, JAOCS 42:315 (1965).
Gunstone, F.D. and B.S. Perera, Chem. Phys Lipids 10:303 (1973).
Ansari, F.H., S.M. Osman and M.R. Subbaram, Ind. J. Chem. 10:1053 (1973).
Ansari, A.A. and S.M. Osman, JAOCS 53: 118 (1976).
Ansari, A.A., F. Ahmad and S.M. Osman, Ibid. 53:541 (1976).
Ansari, A.A., F. Ahmad and S.M. Osman, Fette Seifen Anstrichm. 79:328 (1977).
Ahmad, M.U., M.S. Ahmad and S.M. Osman, JAOCS 55:491 (1978).
Ahmad, M.U., M.S. Ahmad, Jr. and S.M. Osman, Ibid., 55:669 (1978).
Ahmad, M.S. Jr., M.U. Ahmad and S.M. Osman, Ibid. 56:867 (1979).
Christie, W.W. and R.T. Holman, Lipids 1:176 (1966).
Author information
Authors and Affiliations
About this article
Cite this article
Ahmad, M.S., Osman, S.M. Simmons-Smith reaction of allylic hydroxylated long chain α, β-unsaturated esters. J Am Oil Chem Soc 57, 363–364 (1980). https://doi.org/10.1007/BF02662060
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02662060