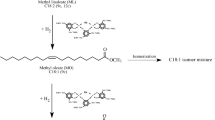
Abstract
Studies have been made on the conjugated hydrogenation of peanut and safflower oils using nickel catalyst and with alcohols as hydrogen donors. Peanut oil was reduced with ethyl alcohol to a final iodine value of 64–71 and the reaction is selective because little saturated acid is formed but some linoleic acid is retained. When the same oil is reduced with isopropyl or secondary butyl alcohols an iodine value of ∼25 is reached while all linoleic acid has disappeared. With safflower oil and isopropyl alcohol an iodine value of ∼46 is reached and the reaction is selective since all linoleic acid has disappeared. Thus the amount of monoene retained depends on initial unsaturation of the oil when a secondary alcohol is used as hydrogen donor.
Similar content being viewed by others
References
Lyubarskii, E. I., Ph. prikl. Khim., Leningr.5, 1025 (1932).
Rush, V. A., I. L. Dvinyaninkova and E. I. Lyubarskii, Ibid.10, 702 (1937).
Ivanova, G. A., and M. F. Belyakov, Ibid.11, 61 (1938).
Puzanov, V. M., Ibid.11, 668, 670 (1938).
Sokol'skii, D. V., and R. Z. Gumarova, Proc. Acad. Sci. USSR111, 693 (1956).
Basu, H. N., and M. M. Chakrabarty, J. Sci. Ind. Res. (India)21D, 467 (1962).
Hilditch, T. P., R. A. Morton and J. P. Riley, Analyst70, 68 (1945).
Cocks, L. V., B. C. Christian and G. Harding, Ibid.,56, 368 (1931).
Schlenk, H., and D. M. Sand, Anal. Chem.,33, 1624 (1961).
Govt. of India. Dept. of Food and Vegetable Oil Products Control Order, “The Capillary Slip Method,” specification under clause 4(1) 1947; Govt. of India, Ministry of Agriculture, S. R. 780 dated Oct. 21, 1950; Mehlenbacher, V. C., The Analysis of Fats and Oils, The Garrard Press, Publishers, Champaign, Ill. 1960, p. 361–362.
Feuge, R. O., M. B. Pepper, Jr., R. T. O'Connor and E. T. Field, JAOCS28, 420 (1951).
Feuge, R. O., and E. R. Cousins, Ibid.37, 267 (1960).
Author information
Authors and Affiliations
About this article
Cite this article
Basu, H.N., Chakrabarty, M.M. Studies on conjugated hydrogenation: Nickel catalyst with alcohols. J Am Oil Chem Soc 43, 119–121 (1966). https://doi.org/10.1007/BF02646281
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02646281