Abstract
The electromotive force of the concentration cell

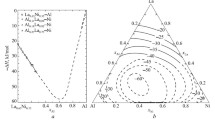
has been determined at 1000°C for molar ratios NaF/AlF3 on the right-hand side 4.0 to 0.46. On the assumption of unit transport number of Na+, relative activities of NaF and A1F3 are derived. Absolute values at a molar ratio of 2.48 were established by measuring the sodium content of aluminum in equilibrium with the melt. At a molar ratio of 3, –NaF = 0.31 andaAlF3 = 0.00058. At molar ratios below 0.65, where the melts are saturated with AlF3, the calculated value ofaAlF3 is 0.81 instead of 1. Possible reasons for the discrepancy are discussed.
Similar content being viewed by others
References
E. W. Dewing:Met. Trans., 1972, vol. 3, pp. 495–501.
E. W. Dewing:Met. Tran., 1970, vol. 1, pp. 1691–94.
E. W. Dewing:Met. Tran., 1970, vol. 1, pp. 2211–15.
JANAF Thermochemical Tables. PB168370, -1, and -2, Dow Chemical Co., Midland, Mich., 1965,1966, and 1967.
W. B. Frank and L. M. Foster:J. Phys. Chem., 1957, vol. 61, pp. 1531–36.
E. W. Dewing:J. Electrochem. Soc, 1970, vol. 117, pp. 780–81.
J. E. Boner:Helb. Chim. Acta, 1950, vol. 33, pp. 1137–53.
K. Grjotheim, J. L. Holm, C. Krohn, and J. Thonstad: paper inSelected Topics in High-Temperature Chemistry, T. Førland, K. Grjotheim, K. Motzfeldt, and S. Urnes, eds., pp. 151–78, Universitets-forlaget, Oslo, 1966.
Anne Fenerty and E. A. Hollingshead:J. Electrochem. Soc, 1960, vol. 107, pp. 993–97.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, K., Dewing, E.W. Activities in NaF-AlF3 melts saturated with Al2O3 . Metall Trans 3, 683–686 (1972). https://doi.org/10.1007/BF02642751
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02642751