Summary
The quantitative radiochemical methodology described in this report allows a major increase in information generation, increased experimental flexibility, improved statistical control, and increased diversity of information per culture. Other advantages relate to economies of technical time, supplies, cells, and test materials per individual culture.
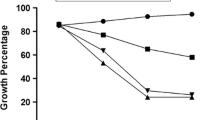
Microcultures of human synovial cells incorporate [14C]glucosamine into hyaluronic acid that accumulated primarily in the media and to a lesser extent in the cell mass. CTAP-I (from lymphoid cells), CTAP-III (from human platelets), PGE2, dibutyryl cAMP, and poly(I)·poly(C) markedly stimulated hyaluronate synthesis, whereas cortisol, cycloheximide, and tunicamycin inhibited stimulated synthesis. Time studies with cycloheximide indicated that translation, essential for the activation of synovial cells, was completed by 17 h postexposure to CTAP-I. Tunicamycin also seemed to inhibit CTAP-I induced activation primarily by interpering with translation; however, tunicamycin also caused modest post-translational inhibition of hyaluronate synthesis in activated adult human synovial cells.
Similar content being viewed by others
References
Castor, C. W. Adrenocorticoid suppression of mucopolysaccharide formation in human connective tissue cell cultures. J. Lab. Clin. Med. 60: 788–798; 1962.
Castor, C. W. Connective tissue activation. I. The nature, specificity, measurement and distribution of connective tissue activating peptide. Arthritis Rheum. 14;41–54; 1971.
Castor, C. W. Connective tissue activation. III. Observations on the mechanism of action of connective tissue acdtivating peptide. J. Lab. Clin. Med. 79:285–301; 1972.
Castor, C. W. Synovial cell activation induced by a polypeptide mediator. Perper, R. J. ed. Mechanism of tissue injury with reference to rheumatoid arthritis. Vol. 256. New York, NY Ann. NY Acad. Sci. 304–317: 1975.
Castor, C. W.; Ritchie, J. C.; Scott, M. E.; Whitney, S. L.: Connective tissue activation. XI. Stimulation of glycosaminoglycan and DNA formation by a platelet factor. Arthritis Rheum. 20:859–868; 1977.
Castor, C. W. Connective tissue activation. IV. Regulatory effects of antirheumatic drugs. Arthritis Rheum. 15:504–514; 1972.
Castor, C. W. Connective tissue activation. VI. The effects of cyclic nucleotides on human synovial cellsin vitro. J. Lab. Clin. Med. 83: 46–55; 1974.
Castor, C. W. Connective tissue activation. VII. Evidence supporting a role for prostaglandins and cyclic nucleotides. J. Lab. Clin. Med. 85: 392–404; 1975.
Castor, C. W. Connective tissue activation. IX. Modification by pharmacologic agents. Arthritis Rheum. 18:451–460; 1975.
Castor, C. W.; Whitney, S. L. Connective tissue activation. XIII. Stimulation of sulfated glycosaminoglycan synthesis by human connective tissue cells by peptide mediators from lymphocytes and platelets. J. Lab. Clin. Med. 91: 811–821; 1978.
Castor, C. W. Abnormalities of connective tissue cells cultured from patients with rheumatoid arthritis. II. Defective regulation of hyaluronate and collagen formation. J. Lab. Clin. Med. 77: 65–75; 1971.
Castor, C. W.; Ritchie, J. C.; Williams, C. H.; Scott, M. E.; Whitney, S. L.; Myers, S. L.; Sloan, T. B.; Anderson, B. Connective tissue adtivation. XIV. Composition and actions of a human platelet autacoid mediator. Arthritis Rheum. 22:260–272; 1979.
Bitter, T.; Muir, H. M. A modified uronic acid carbazole reaction. Anal. Biochem. 4:330–334; 1962.
Oyama, V. I.; Eagle, H. Measurement of cell growth in tissue culture with a phenol reagent (Folin-Ciocalteau). Proc. Soc. Exp. Biol. Med. 91:305–307;1956.
Castor, C. W.; Greene, J. A. The regional distribution of acid mucopolysaccharides in the kidney. J. Clin. Invest. 47:2125–2132; 1968.
Schmickel, R. D.; Distler, J. J.; Jourdian, G. W. Accumulation of sulfate-containing acid mucopolysaccharides in I-cell fibroblasts. J. Lab. Clin. Med. 86:672–682; 1975.
Yaron, M.; Yaron, I.; Gurari-Ritman, D.; Revel, M.; Lindner, H. R.; Zor, U. Stimulation of prostaglandin E production in cultured human fibroblasts by Poly(I)·Poly(C) and human interferon. Nature 267:457–459;1977.
Castor, C. W.; Pek, S. Connective tissue activation. XX. Stimulation of prostaglandin secretion by mediators from lymphocytes (CTAP-I) and platelets (CTAP-III). Arthritis Rheum. 24: 504–509; 1981.
Waechter, C. J.; Lennarz, W. J. The role of polyprenol-linked sugars in glycoprotein synthesis. Annu. Rev. Biochem. 45:95–112; 1976.
Mahoney, W. C.; Duksin, D. Biological activities of the two major components of tunicamycin. J. Biol. Chem. 254(14):6572–6576; 1976.
Castor, C. W.; Dorstewitz, E. L. Identification of acid mucopolysaccharides by paper chromatography. J. Chromatogr. 13:157–165; 1964.
Wasteson, A.; Uthne, K.; Westermark, B. A novel assay for the biosynthesis of sulfated polysaccharide and its application to studies on the effects of somatomatedin on cultured cells. Biochem. J. 136:1069–1074; 1973.
Pratt, R. M.; Yamada, K. M.; Olden, K.; Ohanian, S. H.; Hascall, V. C.: Tunicamycin-induced alterations in the synthesis of sulfated proteoglycans and cell surface morphology in the chick embryo fibroblast. Exp. Cell. Res. 118: 245–252; 1979.
Author information
Authors and Affiliations
Additional information
Study supported by U.S. Public Health Service Grant AM-10728, the Michigan Chapter of the Arthritis Foundation, and Michigan Memorial-Phoenix Project Grant No. 517.
Rights and permissions
About this article
Cite this article
Castor, C.W., Bignall, M.C., Hossler, P.A. et al. Connective tissue activation. XXI. In Vitro 17, 777–785 (1981). https://doi.org/10.1007/BF02618444
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02618444