Abstract
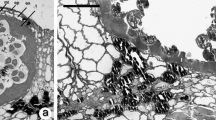
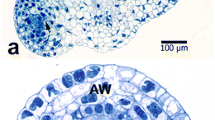
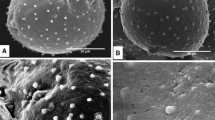
The organization ofPinus sylvestris pollen tubes during growth was studied by video microscopy of living cells and by electron microscopy after freeze-fixation and freeze-substitution (FF-FS). Pollen germinated and the tubes grew slowly for a total period of about 7 days. Some of the grains formed two tubes, while 10–50% of the tubes ramified. These features are in accordance with development in vivo. The cytoplasmic hyaline cap at the tip disappeared during the 2nd or 3rd day of culture. Aggregates of starch grains progressively migrated from the grain into the tube and later into the branches. Vacuoles first appeared at day 2 and eventually filled large parts of the tube. The tube nucleus was located at variable distances from the tip. Some of the organelles showed linear movements in a mostly circulatory pattern, but the majority of the organelles showed brownian-like movements. Rhodamine-phalloidin-stained actin filaments had a gross axial orientation and were found throughout the tube including at the tip. The ultrastructure of pollen tubes was well preserved after FF-FS, but signs of shrinkage were visible. The secretory vesicles in growing tips were not organized in a vesicle cone, and coated pits had a low density with only local accumulations, which is in accordance with slow growth. The mitochondria contained small cristae and a darkly stained matrix and were located more towards the periphery of the tube, indicating low respiratory activity and low oxygen levels. The dictyosomes carried typical trans-Golgi networks, but some contained less than the normal number of cisternae. Other elements of the cytoplasm were irregularly spaced rough endoplasmic reticulum, many multivesicular bodies, lipid droplets and two types of vacuoles. The typical organization associated with tip growth in angiosperm pollen tubes, e.g.Nicotiana tabacum, was not present inP. sylvestris pollen tubes. The different morphology may relate to the growth rate and not to the type of growth.
Similar content being viewed by others
References
Chamberlain CJ (1966) The male gametophyte. In: Gymnosperms, structure and evolution. Dover, New York, pp 306–320
Cutsem P van, Messiaen J (1994) Biological effects of pectic fragments in plant cells. Acta Bot Neerl 43:231–245
Derksen J, Wilms FHA, Pierson ES (1990) The plant cytoskeleton: its significance in plant development. Acta Bot Neerl 39:1–18
Derksen J, Rutten T, Lichtscheidl IK, Win AHN de, Pierson ES, Rongen G (1995a) Quantitative analysis of the distribution of organelles in tobacco pollen tubes: implications for exocytosis and endocytosis. Protoplasma 188:267–276
Derksen J, Rutten T, Amstel A van, Win AHN de, Doris F, Steer MW (1995b) Regulation of pollen tube growth. Acta Bot Neerl 44:93–119
Driessen MNBM, Derksen JWM, Spieksma FThM, Roetman E (1988) Pollenatlas van de nederlandse atmosfeer. Onkenhout, Hilversum, The Netherlands
Gong M, Zhang SX, Li XS, Yang ZH, Cao ZX (1993) The catabolism of lipids and changes of fatty acid composition in the germinating pollen ofPinus yunnanensis and effects of calcium and calmodulin. Chin J Bot 5:159–167
Hackenbrock CR (1968) Chemical and physical fixation of isolated mitochondria in low-energy and high-energy states. Proc Natl Acad Sci 61:598–605
Heath IB (ed) (1990) Tip growth in plant and fungal cells. Academic Press, San Diego, Calif
Iwanami Y (1959) Physiological studies on pollen. Yokohama Municipal Univ (C-34, Biol 13) 116:1–137
Johri BM (1992) Haustorial role of pollen tubes. Ann Bot 70:471–475
Joos U, Aken J van, Kristen U (1994) Microtubules are involved in maintaining the cellular polarity in pollen tubes ofNicotiana sylvestris. Protoplasma 179:5–15
Kroh M, Knuiman B (1982) Ultrastructure of cell wall and plugs of tobacco pollen tubes after chemical extraction of poly-saccharides. Planta 154:241–250
Linskens HF (1967) Pollen. Handbuch der Pflanzenpbysiologie XVIII:368–406
Lloyd CW (1989) The plant cytoskeleton. Curr Opin Cell Biol 1:30–35
Miki-Hirosige H, Nakamura S (1982) Process of metabolism during pollen tube wall formation. J Electron Microsc 31:51–62
Miller DD, Scordilis SP, Hepler PK (1995) Identification and localization of three classes of myosins in pollen tubes ofLilium longiflorum andNicotiana alata. J Cell Sci 108:2549–2563
Picton JM, Steer MW (1981) Determination of secretory vesicle production by dictyosomes in pollen tubes ofTradescantia using cytochalasin D. J Cell Sci 49:261–272
Pierson ES, Cresti M (1992) Cytoskeleton and cytoplasmic organization of pollen and pollen tubes. Int Rev Cytol 140:73–125
Singh H (1978) Embryology of gymnosperms. Borntraeger, Berlin
Tanaka K (1955) The pollen germination and pollen tube development inPinus densiflora Sieb. et Zucc. I. The effect of storage, temperature and sugars. Sci Rep Tohoku Univ Ser 4 B 21:185–198
Tanaka K (1956) The pollen germination and pollen tube development inPinus densiflora Sieb. et Zucc. II. The tube growth and tube nucleus. Sci Rep Tohoku Univ Ser 4 B 22:219–224
Terasaka O, Niitsu T (1994) Differential roles of microtubule and actin-myosin cytoskeleton in the growth ofPinus pollen tubes. Sex Plant Reprod 7:264–272
Traas JA, Doonan JH, Rawlins DJ, Shaw PJ, Watts J, Lloyd CW (1987) An actin network is present in the cytoplasm throughout the cell cycle of carrot cells and associates with the dividing nucleus. J Cell Biol 105:387–395
Willemse MThM (1968) Development of the micro- and macrogametophyte ofPinus sylvestris L., an electron microscopic investigation. Acta Bot Neerl 17:330–331
Willemse MThM (1971a) Gamétogenèse male dePinus sylvestris: morphologie et évaluation approximative des organites. Ann Univ ARERS 9:127–132
Willemse MThM (1971b) Morphological and quantitative changes in the population of cell organelles during microsporogenesis ofPinus sylvestris L. I. Morphological changes from zygotene until prometaphase I. Acta Bot Neerl 20:261–274
Willemse MThM (1971c) Morphological and quantitative changes in the population of cell organelles during micro sporogenesis ofPinus sylvestris L. II. Morphological changes from prometaphase I until the tetrad stage. Acta Bot Neerl 20:411–427
Willemse MThM (1971d) Morphological and quantitative changes in the population of cell organelles during microsporogenesis ofPinus sylvestris L. III. Morphological changes during the tetrad stage and in the young microspore. A quantitative approach to the changes in the population of cell organelles. Acta Bot Neerl 20:498–523
Willemse MThM, Linskens HF (1969) Développement du microgamétophyte chez lePinus sylvestris entre la méiose et la fécondation. Rev Cytol Biol Vég 32:121–128
Yokota E, McDonald AR, Liu B, Shimmen T, Palevitz BA (1995) Localization of a 170 kDa myosin heavy chain in plant cells. Protoplasma 185:178–187
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Win, A.H.N., Knuiman, B., Pierson, E.S. et al. Development and cellular organization ofPinus sylvesfris pollen tubes. Sexual Plant Reprod 9, 93–101 (1996). https://doi.org/10.1007/BF02153056
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02153056