Abstract
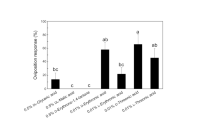
Ovipositing black swallowtail butterflies,Papilio polyxenes, make their final host-selection decisions on the basis of compounds present on the leaf surface. Little information is available, however, on the chemistry of leaf surfaces. The purpose of this study was to develop a technique to extract and quantify the concentrations of compounds from the leaf surfaces ofDaucus carota, one of the main host species forP. polyxenes, with particular reference to compounds already identified as contact oviposition stimulants, namelytrans-chlorogenic acid (CA) and luteolin-7-O-(6″-O-malonyl)-β-d-glucopyranoside (L7MG), as well as its degradation product luteolin-7-glucoside (L7G). Plant surfaces were extracted by dipping leaves sequentially in pairs of solvents: (1) CHCl3-MeOH, (2) near-boiling H2O, (3) CHCl3-near-boiling H2O, and (4) CH2Cl2-CH2Cl2. The resulting extracts were fractionated and analyzed using high-performance liquid chromatography. The leaf-surface concentrations of each compound were calculated using regressions relating leaf surface area to leaf weight that were obtained from measurements of field-collected carrot plants. All four methods removed the three compounds from carrot leaf surfaces, but the solvent systems differed in effectiveness. The chloroform-near-boiling water solvent system performed better than the other solvent combinations, but not significantly so. This system also extracted the highest number of polar, UV-absorbing compounds. Methylene chloride was significantly less efficient than the other methods. An additional test confirmed that the chloroform-near-boiling water method removed compounds from the surface alone and probably not from the apoplast or symplast. Surface concentrations of CA (up to 600 ng/cm2 leaf surface) were substantially greater than those of the two flavonoid compounds. No clear seasonal trend in concentrations was evident from the limited number of sampling dates.
Similar content being viewed by others
References
Bart, K. M., andWilliams, E. H. 1993. Use of dental wax for the study of insect behavior by scanning electron microscopy.Microsc. Res. Tech. 26:180–181.
Baur, R., Birch, A. N. E., Hopkins, R. J., Griffiths, D. W., Simmonds, M. S. J., andStadler, E. 1996. Oviposition and chemosensory stimulation of the root fliesDelia radicum andD. floralis in response to plants and leaf extracts from resistant and susceptibleBrassica genotypes.Entomol. Exp. Appl. 78:61–75.
Blau, W. S. 1981. Life history variation in the black swallowtail butterfly.Oecologia 48:116–122.
Bohm, B. A. 1993. External and vacuolar flavonoids ofRibes viscossisimum.Biochem. Syst. Ecol. 21:745.
Bohm, B. A., andChan, J. 1992. Flavonoids and affinities of Greyiaceae with a discussion of the occurrence of b-ring deoxyflavonoids in dicotyledonous families.Syst. Bot. 17:272–281.
Bohm, B. A., andConstant, H. 1990. Leaf surface flavonoids ofEriodictyon tricholaclyx.Biochem. Syst. Ecol. 18:491–492.
Boppré, M. 1978. Chemical communication, plant relationships, and mimicry in the evolution of danaid butterflies.Entomol. Exp. Appl. 13:363–376.
Boppré, M. 1983. Leaf-scratching-a specialized behaviour of danaine butterflies (Lepidoptera) for gathering secondary plant substances.Oecologia 59:414–416.
Conover, W. J. 1980. Practical Nonparametric Statistics, 2nd ed. John Wiley & Sons, New York.
Derridj, S., Finala, V., andJolivet, E. 1987. Low molecular carbohydrates ofZea mays L. leaves and the egg-laying ofOstrinia nubilalis Hbn. Lepid. Pyralidae, pp. 295–299,in V. Labeyrie, G. Fabres, and D. Lachaise (eds.). Proceedings of the 6th International Symposium on Insect-Plant Relationships, Pau 1986. W. Junk, Dordrecht.
Derridj, S., Wu, B. R., Stammitti, L., Garrec, J. P., andDerrien, A. 1996. Chemicals on the leaf surface, information about the plant available to insects.Entomol. Exp. Appl. 80:197–201.
Feeny, P., Rosenberry, L., andCarter, M. 1983. Chemical aspects of ovipositional behavior in butterflies, pp. 27–76,in S. Ahmad (ed.). Herbivorous Insects: Host-Seeking Behavior and Mechanisms. Academic Press, New York.
Feeny, P., Blau, W. S., andKareiva, P. M. 1985. Larval growth and survivorship of the black swallowtail butterfly in central New York.Ecol. Monogr. 55:167–187.
Feeny, P., Sachdev, K., Rosenberry, L., andCarter, M. 1988. Luteolin 7-O-(6″-O-malonyl)-β-d-glucoside andtrans-chlorogenic acid: Oviposition stimulants for the black swallowtail butterfly.Phytochemistry 27:3439–3448.
Feeny, P., Städler, E., Ahman, I., andCarter, M. 1989. Effects of plant odor on oviposition by the black swallowtail butterfly,Papilio polyxenes (Lepidoptera: Papilionidae).J. Insect Behavior. 2:803–827.
Fiala, V., Glad, C., Martin, M., Jolivet, E., andDerridj, S. 1990. Occurrence of soluble carbohydrates on the phylloplane of maize (Zea mays L.): Variations in relation to leaf heterogeneity and position on the plant.New Phytol. 115:609–615.
Harborne, J. B. 1973. Phytochemical Methods. Chapman and Hall, London.
Hunt, G. M., andBaker E. A. 1980. Phenolic constituents of tomato fruit cuticles.Phytochemistry 19:1415–1419.
Linley, J. R. 1989. Tergal spines ofMansonia titillans andMa. dyari (Diptera: Culicidae) and their effect on a leaf surface during oviposition.J. Med. Entomol. 26:402–406.
Merrall, G. T. 1981. Physical factors that influence the behavior of chemicals on leaf surfaces, pp. 265–281.in J. P. Blakeman (ed.). Microbial Ecology of the Phylloplane. Academic Press, London.
Miller, R. H. 1985. The prevalence of pores and canals in leaf cuticular membranes.Ann. Bot. 55:459–471.
Miller, R. H. 1986. The prevalence of pores and canals in leaf cuticular membranes. 2. Supplemental studies.Ann. Bot. 57:419–434.
Peterson, C. E., andSimon, P. W. 1986. Carrot breeding, pp. 321–356,in M. J. Bassett (ed.). Breeding Vegetable Crops. AVI Publishing, Wesport, Conn.
Price, C. E. 1982. A review of the factors influencing the penetration of pesticides through plant leaves, pp. 237–252,in D. F. Cutler, K. L. Alvin, and C. E. Price (eds.). The Plant Cuticle. Academic Press, London.
Proksch, M., Weissenbock, G., andRodriguez, E. 1985. Flavonoids and phenolic acids inAdenostoma, a dominant genus of the Californian chaparral.Phytochemistry 24:2889–2892.
Proksch, P., Mitsakos, A., Bodden, J., andWollenweber, E. 1986. Benzofurans and methylated flavonoids ofGeraea (Asteraceae).Phytochemistry 25:2367–2370.
Reid, A. R., andBohm, B. A. 1993. External and vacuolar flavonoids ofBrachyglottis cassinioides.Biochem. Syst. Ecol. 21:746.
Renwick, J. A. A., Radke, C. D., Sachdev-Gupta, K. S., andStadler, E. 1992. Leaf surface chemicals stimulating oviposition byPieris rapae (Lepidoptera: Pieridae) on cabbage.Chemoecology 3:33–38.
Richmond, D. V., andMartin, J. T. 1959. Studies on plant cuticle. III. The composition of the cuticle of apple leaves and fruits.Ann. Appl. Biol. 47:583–592.
Robinson, T. 1991. The Organic Constituents of Higher Plants: Their Chemistry and Interrelationships. Cordus Press, North Amhurst, Massachusetts.
Roessingh, P., Stadler, E., Schöni, R., andFeeny, P. 1991. Tarsal contact chemoreceptors of the black swallowtail butterfly.Papilio polyxenes: Responses to phytochemicals from host- and non-host plants.Physiol. Entomol. 16:485–495.
Sokal, R., R., andRohlf, F. J. 1981. Biometry, 2nd ed. W. H. Freeman, New York.
Städler, E. 1986. Oviposition and feeding stimuli in leaf surface waxes, pp. 105–121,in B. Juniper and R. Southwood (eds.). Insects and the Plant Surface. Edward Arnold, London.
Städler, E., andBuser, H. R. 1984. Defense chemicals in leaf surface wax synergistically stimulate oviposition by a phytophagous insect.Experientia 40:1157–1159.
Städler, E., andRoessingh, P. 1991. Perception of surface chemicals by feeding and ovipositing insects.Symp. Bio. Hung. 39:71–86.
Tietz, H. M. 1972. An index to the described life histories, early stages and hosts of the Macrolepidoptera of the continental United States and Canada, Volumes 1 and 2. A. C. Allyn, Sarasota, Florida.
Traynier, R. M. M., andHines, E. R. 1987. Probes by aphids indicated by stain induced fluorescence in leaves.Entomol. Exp. Appl. 45:198–201.
Tukey, H. B., Jr., 1970. The leaching of substances from plants.Annu. Rev. Plant Physiol. 21:305–324.
van Loon, J. J. A., andvan Meer, M. M. M. 1991. Chemosensory perception of leaf surface chemicals by ovipositingPieris brassicae L. butterflies.Proc. Exp. Appl. Entomol. 2:56–61.
van Loon, J. J. A., Blaakmeer, A., Griepink, F. C., van Beek, T. A., Schoonhoven, L. M., andde Groot, A. 1992. Leaf surface compound fromBrassica oleracea (Cruciferae) induces oviposition byPieris brassicae (Lepidoptera: Pieridae).Chemoecology 3:39–44.
Wagner, G. J. 1982. Compartmentation in plant cells: The role of the vacuole, pp. 1–45,in L. L. Creasy and G. Hrazdina (eds.). Cellular and Subcellular Localization in Plant Metabolism. Vol. 16. Plenum Press, New York.
Zobel, A. M., andBrown, S. A. 1988. Determination of furanocoumarins on the leaf surface ofRuta graveolens with an improved extraction technique.J. Nat. Prod. 51:941–946.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brooks, J.S., Williams, E.H. & Feeny, P. Quantification of contact oviposition stimulants for black swallowtail butterfly,Papilio polyxenes, on the leaf surfaces of wild carrot,Daucus carota . J Chem Ecol 22, 2341–2357 (1996). https://doi.org/10.1007/BF02029551
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02029551