Abstract
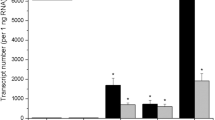
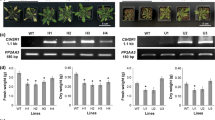
The enzyme tryptophan decarboxylase (TDC) (EC 4.1.1.28) catalyses a key step in the biosynthesis of terpenoid indole alkaloids inC. roseus by converting tryptophan into tryptamine. Hardly anytdc mRNA could be detected in hormone-independent callus and cell suspension cultures transformed by the oncogenic T-DNA ofAgrobacterium tumefaciens. Supply of tryptamine may therefore represent a limiting factor in the biosynthesis of alkaloids by such cultures. To investigate this possibility, chimaeric gene constructs, in which atdc cDNA is linked in the sense or antisense orientation to the cauliflower mosaic virus 35S promoter and terminator, were introduced inC. roseus cells by infecting seedlings with an oncogenicA. tumefaciens strain. In the resulting crown gall tumour calluses harbouring thetdc sense construct, an increased TDC protein level, TDC activity and tryptamine content but no significant increase in terpenoid indole alkaloid production were observed compared to empty-vector-transformed tumour calluses. In tumour calluses containing thetdc antisense construct, decreased levels of TDC activity were measured. Factors which might be responsible for the lack in increased terpenoid indole alkaloid production in thetdc cDNA overexpressing crown gall calluses are discussed.
Similar content being viewed by others
References
Berlin, J., Forche, E., Wray, V., Hammer, J., and Hosel, W. (1983) Formation of benzophenanthridine alkaloids by suspension cultures ofEschscholtzia californica.Z. Naturforsch. C38, 346–52.
Berlin, J., Rügenhagen, C., Dietze, P., Fecker, L., Goddijn, O.J.M. and Hoge, J.H.C. (1993) Increased production of serotonin by suspension and root cultures ofPeganum harmala transformed with a tryptophan decarboxylase cDNA clone fromCatharanthus roseus.Transgenic Res. 2, 336–44.
Bevan, M. (1984) BinaryAgrobacterium vector for plant transformation.Nucl. Acids Res. 12, 8711–21.
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding.Anal. Biochem. 72, 248–54.
DeLuca, V. and Cutler, A.J. (1987) Subcellular localization of enzymes involved in indole alkaloid biosynthesis inCatharanthus roseus.Plant Physiol. 85, 1099–1102.
DeLuca, V., Marineau, C. and Brisson, N. (1989) Molecular cloning and analysis of cDNA encoding a plant tryptophan decarboxylase: comparison with animal dopa decarboxylases.Proc. Natl Acad. Sci. USA 86, 2582–86.
Devereux, J., Haeberli, P. and Smithies, O. (1984) A comprehensive set of sequence analysis programs of the VAX.Nucl. Acids Res. 12, 387–95.
Gamborg, O.L., Miller, R.A. and Ojima, K. (1968) Nutrient requirements of suspension cultures of soybean root cells.Exp. Cell. Res. 50, 151–58.
Geliebter, J. (1987) Dideoxynucleotide sequencing of RNA and uncloned cDNA.Focus 9, 5–8.
Goddijn, O.J.M., de Kam, R.J., Zanetti, A., Schilperoort, R.A. and Hoge, J.H.C. (1992) Auxin rapidly down-regulates transcription of the tryptophan decarboxylase gene fromCatharanthus roseus.Plant Mol. Biol. 18, 1113–20.
Goddijn, O.J.M., vanderDuyn Schouten, P.M., Schilperoort, R.A. and Hoge, J.H.C. (1993) A chimaeric tryptophan decarboxylase gene as a novel selectable marker in plant cells.Plant Mol. Biol. 22, 907–12.
Goddijn, O.J.M., Lohman, F.P., Kam, R.J. de, Schilperoort, R.A. and Hoge, J.H.C. (1994) Nucleotide sequence of the tryptophan decarboxylase gene ofCatharanthus roseus and expression oftdc-gus A gene fusions inNicotiana tabacum.Mol. Gen. Gen. 242, 217–25.
Hay, C.A., Anderson, L.A., Roberts, M.F. and Phillipson, J.D. (1988) Alkaloid production by plant cell cultures. In Mizrahi, A. ed.,Biotechnology in Agriculture, Advances in Biotechnological Processes, pp. 97–140. New York: Alan R Liss Inc.
Hooykaas, P.J.J. and Schilperoort, R.A. (1992)Agrobacterium and plant genetic engineering.Plant Mol. Biol. 19, 15–38.
Hunkapillar, M.W., Lujan, E., Ostrander, F. and Hood, L.E. (1983) Isolation of microgram quantities of proteins from polyacrylamide gels for amino acid sequence analysis.Meth. Enzymol. 91, 227–236.
Huyng, T.V., Young, R.A. and Davis, R.W. (1985) Construction and screening cDNA libraries in λgt10 and λgt11. In Glover, D.M. ed.,Cloning: a Practical Approach, pp. 79–88. Oxford: IRL Press Ltd.
Knobloch, K.H. and Berlin, J. (1980) Influence of medium composition on the formation of secondary compounds in cell suspension cultures ofCatharanthus roseus (L.) G. Don.Z. Naturforsch. 35C, 551–6.
Knobloch, K.H., Hansen, B. and Berlin, J. (1981) Medium-induced formation of indole alkaloids and concomitant changes of interrelated enzyme activities in cell suspension cultures ofCatharanthus roseus.Z. Naturforsch. C 36, 40–3.
Koekman, B.P., Ooms, G., Klapwijk, P.M. and Schilperoort, R.A. (1979) Genetic map of an octopine Ti plasmid.Plasmid 2, 347–57.
Kutchan, T.M., Hampp, N., Lottspeich, F., Beyreuther, K. and Zenk, M.H. (1988) The cDNA clone for strictosidine synthase fromRauvolfia serpentina DNA sequence determination and expression inEscherichia coli.FEBS Lett. 237, 40–44.
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4.Nature 227, 680–5.
Linsmaier, E.M. and Skoog, F. (1965) Organic growth factor requirements of tobacco tissue cultures.Physiol. Plant. 18, 100–27.
Marsch, J.L., Erfle, M. and Wykes, E.J. (1984) The pIC plasmid and phage vectors with versatile cloning sites for recombinant selection by insertional inactivation.Gene 32, 481–5.
Mattonovich, D., Ruker, F., Camara, M.A. da, Laimer, M., Regner, F., Steinkellner, H., Himmler, G. and Katinger, H. (1989) Efficient transformation ofAgrobacterium spp. by electroporation.Nucl. Acids Res. 17, 6747.
McKnight, T.D., Roessner, C.A., Devagupta, R., Scott, A.I. and Nessler, C.L. (1990) Nucleotide sequence of a cDNA encoding the vacuolar protein strictosidin synthase fromCatharanthus roseus.Nucl. Acids Res. 18, 4939.
Mérillon, J.-M., Doireau, P., Guillot, A., Chénieux, J.-C. and Rideau, M. (1986) Indole alkaloid accumulation and tryptophan decarboxylase activity inCatharanthus roseus cells cultured in three different media.Plant Cell Rep. 5, 23–26.
Mérillon, J.-M., Ouelhazi, L., Doireau, P., Chénieux, J.-C., and Rideau, M. (1989) Metabolic changes and alkaloid production in habituated and non-habituated cells ofCatharanthus roseus grown in hormone-free medium. Comparing hormone-deprived non-habituated cells with habituated cells.J. Plant. Physiol. 134, 54–60.
Noé, W. and Berlin, J. (1985) Induction ofde novo synthesis of tryptophan decarboxylase in cell suspensions ofCatharanthus roseus.Planta 166, 500–4.
Pasquali, G., Goddijn, O.J.M., Waal, A. de, Verpoorte, R., Schilperoort, R.A., Hoge, J.H.C. and Memelink, J. (1992) Coordinated regulation of two indole alkaloid biosynthetic genes fromCatharanthus roseus by auxin and elicitors.Plant Mol. Biol. 18, 1121–31.
Pennings, E.J.M., Hegger, I., Heijden van der R., Duine, J.A. and Verpoorte, R. (1987) Assay of tryptophan decarboxylase fromCatharanthus roseus plant cell cultures by HPLC.Anal. Biochem. 165, 133–6.
Pennings, E.J.M., Goddijn, O.J.M., Hoge, J.H.C., and Verpoorte, R. (1989) Purification of tryptophan decarboxylase from aCatharanthus roseus cell suspension culture.J. Chrom. 483, 311–8.
Phillipson, J.D. (1990) Plants and sources of valuable products. In Charlwood, B.V., Rhodes, M.J.C. eds,Secondary Products from Plant Tissue Culture, pp. 1–21. Oxford: Clarendon Press.
Pietrzak, M., Shillito, D., Hohn, T. and Potrykus, I. (1986) Expression in plants of two bacterial antibiotic resistance genes after protoplast transformation with a new plant expression vector.Nucl. Acids Res. 14, 5857–68.
Poulsen, C., Goddijn, O.J.M., Hoge, J.H.C. and Verpoorte, R. (1994) Anthranilate synthase and chorismate mutase activities in transgenic tobacco plants overexpressing tryptophan decarboxylase fromCatharanthus roseus.Transgenic Res. 3, 43–9.
Roewer, I.A., Cloutier, N., Nessler, C.L., and DeLuca, V. (1992) Transient induction of tryptophan decarboxylase (TDC) and strictosidine synthase (SS) genes in cell suspension cultures ofCatharanthus roseus.Plant Cell Rep. 11, 86–9.
Sambrook, J., Fritsch, E.F. and Maniatis, T. (1989) Molecular Cloning:a Laboratory Manual, Cold Spring Harbour, New York: Cold Spring Harbour Laboratory Press.
Sanger, F., Nickler, S. and Coulson, A.R. (1977) DNA sequencing with chain terminating inhibitors.Proc. Natl. Acad. Sci. USA 74, 5463–7.
Schripsema, J. and Verpoorte, R. (1992) Search for factors related to the indole alkaloid production in cell suspension cultures ofTabernaemontana divaricata.Planta Med. 58, 245–249.
Songstad, D.D., DeLuca, V., Brisson, N., Kurz, W.G.W. and Nessler, C.L. (1990) High levels of tryptamine accumulation in transgenic tobacco expressing tryptophan decarboxylase.Plant Physiol. 94, 1410–3.
Stevens, L.H., Blom, T.J.M. and Verpoorte, R. (1993) Subcellular localization of tryptophan decarboxylase, strictosidine synthase and strictosidine glucosidase in cell suspension cultures ofCatharanthus roseus andTabernamontana divaricata.Plant Cell Rep. 12, 573–6.
Van der Heijden, R., Lamping, R. J., Out, P. P., Wijnsma, R. and Verpoorte, R. (1987) High performance liquid chromatographic determination of indole alkaloids in a suspension culture ofTabernamontana divaricata.J. Chrom. 396, 287–95.
Van Slogteren, G.M.S., Hoge, J.H.C., Hooykaas, P.J.J. and Schilperoort, R.A. (1983) Clonal analysis of heterogeneous crown gall tumor tissues induced by wild-type and shooter mutant strains ofAgrobacterium tumefaciens-expression of T-DNA genes.Plants Mol. Biol. 2, 321–33.
Von Heijne, G. (1986) A new method for predicting signal cleavage sites.Nucl. Acids Res. 14, 4683–90.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goddijn, O.J.M., Pennings, E.J.M., van der Helm, P. et al. Overexpression of a tryptophan decarboxylase cDNA inCatharanthus roseus crown gall calluses results in increased tryptamine levels but not in increased terpenoid indole alkaloid production. Transgenic Research 4, 315–323 (1995). https://doi.org/10.1007/BF01972528
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01972528