Summary
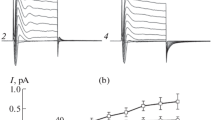
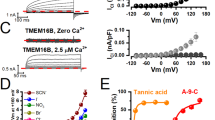
The effect of pH buffers and related compounds on the conductance of an outwardly rectifying anion channel has been studies using the patch-clamp technique. Single-channel current-voltage relationships were determined in solutions buffered by trace amounts of bicarbonate and in solutions containing N-substituted taurines (HEPES, MES, BES, TES) and glycines (glycylglycine, bicine and tricine), Tris andbis-Tris at millimolar concentrations. HEPES (pKa=7.55) reduced the conductance of the channel when present on either side of the membrane. Significant inhibition was observed with 0.6mm HEPES on the cytoplasmic side (HEPES i ) and this effect increased with [HEPES i ] so that conductance at the reversal potential was diminished ≈25% with 10mm HEPES i )and ≈70% at very high [HEPES i ]. HEPES i block was relieved by applying positive voltage but positive currents were not consistent with a Woodhulltype blocking scheme in that calculated dissociation constants and electrical distances depended on HEPES concentration. Results obtained by varying total HEPES i concentration at constant [HEPES−] and vice versa suggest both the anionic and zwitterionic (protonated) forms of HEPES inhibit. Structure-activity studies with related compounds indicate the sulfonate group and heterocyclic aliphatic groups are both required for inhibition from the cytoplasmic side. TES (pKa=7.54), substituted glycine buffers (pKa=8.1–8.4) andbis-Tris (pKa=6.46) had no measurable effect on conductance and appear suitable for use with this channel.
Similar content being viewed by others
References
Ascher, P., Marty, A., Neild, T.O. 1978. Lifetime and elementary conductance of the channels mediating the excitatory effects of acetylcholine inAplysia neurones.J. Physiol. (London) 278:177–206
Blatz, A.L., Magleby, K.L. 1984. Ion conductance and selectivity of single calcium-activated potassium channels in cultured rat muscle.J. Gen. Physiol. 84:1–23
Bormann, J., Hamill, O.P., Sakmann, B. 1987. Mechnism of anion permeation through channels gated by glycine and γ-aminobutyric acid in mouse cultured spinal neurones.J. Physiol. (London) 385:243–286
Eisenthal, R., Cornish-Bowden, A. 1974. The ddirect linear plot.Biochem. J. 139:715–720
Frizzell, R.A. 1987. Cystic fibrosis: A disease of ion channels?Trends Neurosci. 10:190–193
Good, N.E., Izawa, S. 1968. Hydrogen ion buffers.Methods Enzymol. 24((Part B):53–68
Good, N.E., Winget, G.D., Winter, W., Connolly, T.N., Izawa, S., Singh, R.M.M. 1966. Hydrogen ion buffers for biological research.Biochemistry 5:467–477
Gray, R., Johnston, D. 1985. Retification of single GABA-gated chloride channels in adult hippocampal neurons.J. Neurophysiol. 54:134–142
Halm, D.R., Frizzell, R.A. 1988. Ion permeation through the apical membrane chloride channel in a secretory epithelial cell.FASEB J. 2:A1283
Halm, D.R., Rechkemmer, G.R., Schoumacher, R.A., Frizzell, R.A. 1988. Apical membrane chloride channels in a colonic cell line activated by secretory agonists.Am. J. Physiol. 254:C505-C511
Hamill, O.P., Bormann, J., Sakmann, B. 1983. Activation of multiple-conductance state chloride channels in spinal neurones by glycine and GABA.Nature (London) 305:805–808
Hamill, O.P., Marty, A., Neher, E., Sakmann, B., Sigworth, F.J. 1981. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches.Pfluegers Arch. 391:85–100
Hanke, W., Miller, C. 1983. Single chloride channels fromTorpedo electroplax: Activation by protons.J. Gen. Physiol. 82:25–45
Hanrahan, J.W., Tabcharani, J.A. 1989. Possible role of the outwardly rectifying anion channel in epithelial Cl and HCO3 transport.Ann. NY Acad. Sci. 574:30–43
Li, M., McCann, J.D., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh, M.J. 1988. Cyclic AMP-dependent protein kinase opens chloride channels in normal but not cystic fibrosis airway epithelium.Nature (London) 331:358–360
Low, W., Tabcharani, J.A., Hanrahan, J.W. 1990. Outwardly rectifying anion channels: Activation by salts, inhibition by IAA-94 and NPPB.FASEB J. 4:A726
Norris, C.H., Guth, P.S. 1985. Buffers may have pharmacological actions.Trends Pharmacol. Sci. 6:315
Prod'hom, B., Pietrobon, D., Hess, P. 1987. Direct measurement of proton transfer rates to a group controlling the dihydropyridine-sensitive Ca2+ channel.Nature (London) 329:243–246
Shoumacher, R.A., Shoemaker, R.L., Halm, D.R., Tallant, E.A., Wallace, R.W., Frizzell, R.A. 1987. Phosphorylation fails to activate chloride channels from cystic fibrosis airway cells.Nature (London) 330:752–754
Sigworth, F.J. 1977. Sodium channels in nerve apparently have two conductance states.Nature (London) 270:265–267
Tabcharani, J.A., Jensen, T.J., Riordan, J.R., Hanrahan, J.W. 1989. Bicarbonate permeability of the outwardly rectifying anion channel.J. Membrane Biol. 112:109–122
Tunnicliff, G., Smith, J.A. 1981. Competitive inhibition of γ-aminobutyric acid receptor binding by N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid and related buffers.J. Neurochem. 36:1122–1126
Vaughan, P.C. 1986. Anion permeability sequence and voltageclamp measurements in frog muscle at pH 9.Can. J. Physiol. Pharmacol. 64:A28
Vergara, C., Latorre, R. 1983. Kinetics of Ca2+-activated K+ channels from rabbit muscle incorporated into planar bilayers.J. Gen. Physiol. 82:543–568
Wilson, W.A., Clark, M.T., Pellmar, T.C. 1977. Tris buffer attenuates acetylcholine responses inAplysia neurons.Science 196:440–441
Witte, O.W., Speckmann, E.-J., Walden, J. 1985. Acetylcholine responses of identified neurons inHelix pomatia. II. Pharmacological properties of acetylcholine responses.Comp. Biochem. Physiol. 80:C25–35
Woodhull, A.M. 1973. Ionic blockage of sodium channels in nerve.J. Gen. Physiol. 61:687–708
Yamamoto, D., Suzuki, N. 1987. Blockage of chloride channels by HEPES buffer.Proc. R. Soc. London B 230:93–100
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hanrahan, J.W., Tabcharani, J.A. Inhibition of an outwardly rectifying anion channel by HEPES and related buffers. J. Membrain Biol. 116, 65–77 (1990). https://doi.org/10.1007/BF01871673
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01871673