Summary
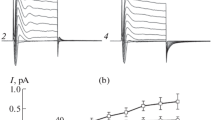
The influence of Ba++ and Cs+ on the K+ conductive properties of the rabbit isolated perfused cortical collecting tubule were assessed using electrophysiological methods. As before, elevation of K+ from 5 to 25 or 50mm (choline+ for K+ substitution) in the luminal perfusate, caused a marked hyperpolarization (lumen negative) of the transepithelial voltage,V te, and increase in the transepithelial conductance,G te-indicative of a high luminal (apical) K+ conductance-where-as a similar elevation of K+ in the bath caused only minor changes inV te andG te. In the presence of 5mm K+ in perfusate and bath, addition of 5mm Ba++ to the perfusate causedG te to decrease from 16.1 to 7.4 mScm−2 (10mm Cs+ had qualitatively similar effects) and greatly diminished the response ofV te andG te to K+ elevation in the lumen, reflecting a decrease in the apical membrane K+ conducatance. In contrast, a similar addition of 5mm Ba++ to the bath caused only a modest reduction inG te of 0.4 mScm−2, consistent with a relatively low K+ conductance of the basolateral membrane and tight junction. In other studies the effects of luminal addition of Ba++ and amiloride were found to be relatively independent, with the magnitude of the Ba++-sensitiveG te (8.7 mScm−2) being several-fold greater than the amiloride-sensitiveG te (1.4 mScm−1), indicative of a dominat K+ conductance at that border which is spatially distinct from the Na+ conductance. Furthermore, from the input current-voltage relation of the tubule, the effects of Ba++ (0.1–5mm) and Cs+ (10mm) at the luminal border on tissue conductance were found to be highly voltage-dependent — the effects on conductance being diminished with lumennegative voltages and more pronounced, approaching a maximum, with lumen-positive voltages. It is concluded that the apical (luminal) cell membrane contains a dominant K+ conductive pathway that is blocked by luminal addition of Ba++ and Cs+. The voltage-dependent nature of the block is consistent with a K+ conductive pathway which has a binding site for Ba++ and Cs+, and presumably K+, located deep within the channel.
Similar content being viewed by others
References
Adelman, W.J., French, R.J. 1978. Blocking of the squid axon potassium channel by external caesium ions.J. Physiol. (London) 276:13–25
Armstrong, C.M., Taylor, S.R. 1980. Interaction of barium ions with potassium channels in squid giant axons.Biophys. J. 30:473–488
Begenisich, T., De Weer, P. 1980. Potassium flux ratio in voltage-clamped squid giant axons.J. Gen. Physiol. 76:83–98
Biagi, B., Sohtell, M., Giebisch, G. 1981. Intracellular potassium activity in the rabbit proximal straight tubule.Am J. Physiol. 241:F677-F686
Boudry, J., Stoner, L.C., Burg, M.B. 1976. Effect of acid lumen pH on potassium transport in renal cortical collecting tubules.Am. J. Physiol. 230:239–244
Burg, M., Grantham, J., Abramow, M., Orloff, J. 1966. Preparation and study of fragments of single rabbit nephrons.Am. J. Physiol. 210:1293–1298
Coronado, R., Miller, C. 1979. Voltage-dependent caesium blockade of a cation channel from fragmented sarcoplasmic reticulum.Nature (London) 280:807–810
Eaton, D.C., Brodwick, M.S. 1980. Effects of barium on the potassium conductance of squid axon.J. Gen. Physiol. 75:727–750
Gogelein, H., Driessche, W. van 1981. noise analysis of the K+ current through the apical membrane ofNecturus gallbladder.J. Membrane Biol. 60:187–198
Grantham, J.J., Burg, M.B., Orloff, J. 1970. The nature of transtubular Na and K transport in isolated rabbit renal collecting tubules.J. Clin. Invest. 49:1815–1826
Greger, R. 1981. Coupled transport of Na+ and Cl− in the thick ascending limb of Henle's loop of rabbit nephron.J. Physiol. Scand. (Suppl.)14:1–15
Hagiwara, S., Miyazaki, S., Rosenthal, N.P. 1976. Potassium and the effect of cesium on this current during anomalous rectification of the egg cell membrane of a starfish.J. Gen. Physiol. 67:621–638
Helman, S.I., Grantham, J.J., Burg, M.B. 1971. Effect of vasopressin on electrical resistance of renal cortical collecting tubules.Am. J. Physiol. 220:1825–1832
Helman, S.I., O'Neil, R.G. 1977. Model of active transepithelial Na and K transport or renal collecting tubules.Am. J. Physiol. 233:F559-F571
Hille, B., Schwarz, W. 1978. Potassium channels as multi-ion single-file pores.J. Gen. Physiol. 72:409–442
Iino, Y., Imai, M. 1978. Effects of prostaglandins on Na transport in isolated collecting tubules.Pfluegers Arch. 373:125–132
Koeppen, B.M., Biagi, B.A., Giebisch, G.H. 1983. Intracellular microelectrode characterization of the rabbit cortical collecting duct.Am. J. Physiol. 244:F35-F47
Labarca, P.P., Miller, C. 1981. A K+-selective, three-state channel from fragmented sarcoplasmic reticulum of frog leg muscle.J. Membrane Biol. 61:31–38
Nagel, W., Hirschmann, W. 1980. K+-permeability of the outer border of the frog skin (R. Temporaria).J. Membrane Biol. 52:107–113
Nielsen, R. 1979. Coupled transepithelial sodium and potassium transport across isolated frog skin: Effect of ouabain, amiloride, and the polyene antibiotic Filipin.J. Membrane Biol. 51:161–184
O'Neil, R.G. 1981. Potassium secretion by the cortical collecting tubule.Fed. Proc. 40:2403–2407
O'Neil, R.G. 1982a. Apical cell membrane K conductance properties of the cortical collecting tubule: Influence of Ba and Cs.Kidney. Int. 21:283
O'Neil, R.G. 1982b. Effect of luminal H+ and Ba++ on the apical cell membrane K+ conductance of the cortical collecting tubule (CCT).Fed. Proc. 41:1006
O'Neil, R.G. 1982c. Microelectrode assessment of the luminal cell membrane ionic conductive properties of the cortical collecting tubule (CCT).Physiologist 25:334
O'Neil, R.G., Boulpaep, E.L. 1979. Effect of amiloride on the apical cell membrane cation channels of a sodium-absorbing, potassium-secreting renal epithelium.J. Membrane Biol. 50:365–387
O'Neil, R.G., Boulpaep, E.L. 1982. Ionic conductive properties and electrophysiology of the rabbit cortical collecting tubule.Am. J. Physiol. 243:F81-F95
O'Neil, R.G., Helman, S.I. 1977. Transport characteristics of renal collecting tubules: Influences of DOCA and diet.Am. J. Physiol. 233:F544-F558
Planelles, G., Teulon, J., Anagnostopoulos, T. 1981. The effects of barium on the electrical properties of the basolateral membrane in proximal tubule.Naunyn Schmiedebergs Arch. Pharmacol. 318:135–141
Reuss, L., Cheung, L.Y., Grady, T.P. 1981. Mechanisms of cation permeation across apical cell membrane ofNecturus gallbladder: Effects of luminal pH and divalent cations on K+ and Na+ permeability.J. Membrane Biol. 59:211–224
Schwartz, G.J., Burg, M.B. 1978. Mineralocorticoid effects on cation transport by cortical collecting tubulesin vitro.Am. J. Physiol. 235 (6):F576-F585
Sperelakis, N., Schneider, M.F., Harris, E.J. 1967. Decreased K+ conductance produced by Ba++ in frog sartorius fibers.J. Gen. Physiol. 50:1565–1583
Standen, N.B., Stanfield, P.R. 1978. A potential and time-dependent blockade of inward rectification in frog skeletal muscle fibres by barium and strontium ions.J. Physiol. (London) 280:169–191
Stokes, J.B. 1981. Potassium secretion by cortical collecting tubule: Relation to sodium absorption, luminal sodium concentration, and transepithelial voltage.Am. J. Physiol. 241:F395-F402
Stoner, L.C., Burg, M.B., Orloff, J. 1974. Ion transport in cortical collecting tubule: Effect of amiloride.Am. J. Physiol. 227:453–459
Van Driessche, W., Zeiske, W. 1981. Ba2+-induced conductance fluctuations of spontaneously fluctuating K+ channels in the apical membrane of frog skin (Rana temporaria).J. Membrane Biol. 56:31–42
Wills, N.N., Eaton, D.C., Lewis, S.A., Ifshin, M.S. 1979. Current-voltage relationship of the basolateral membrane of a tight epithelium.Biochim. Biophys. Acta 55:519–523
Zeiski, W., Van Driessche, W. 1979. Saturable K+ pathway across the outer border of frog skin (Rana temporaria): kinetics and inhibition by Cs+ and other cations.J. Membrane Biol. 47:77–96
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
O'Neil, R.G. Voltage-dependent interaction of Barium and Cesium with the Potassium Conductance of the Cortical Collecting Duct Apical cell membrane. J. Membrain Biol. 74, 165–173 (1983). https://doi.org/10.1007/BF01870505
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01870505