Summary
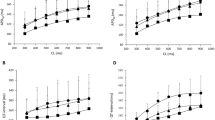
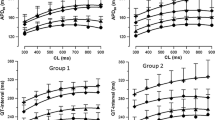
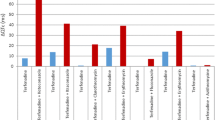
We evaluated the antiarrhythmic efficacy and the minimal effective concentrations of propafenone and its metabolite 5-hydroxy-propafenone during a) acute intravenous infusion (1.5 mg/kg in bolus followed by 45 minutes infusion), b) an acute oral single-dose test (450 mg), and c) 14-day chronic therapy (300 mg tid) followed by a washout. Oxidative metabolism was assessed by a debrisoquine oral test in every patient. Eleven patients with stable ventricular premature beats (VPBs)≥300/hr and Lown class ≥ 3 completed the study. The main results emphasized a certain discrepancy between the clinical effect of the acute intravenous infusion (efficacy in 5 out of 11 patients) and of the acute oral test and chronic therapy (efficacy in 11/11), with a time lag of the ECG changes during the acute intravenous infusion. The minimal effective concentrations were lower after acute oral administration compared with chronic treatment both for propafenone (200±189 ng/ml vs. 492±530 ng/ml; p<0.05) and for 5-hydroxy-propafenone (82±40 ng/ml vs. 149±80 ng/ml; p<0.02). A linear correlation was demonstrated between drug/metabolite ratios of propafenone and debrisoquine, either after acute oral (r=0.91) or after chronic administration (r=0.84). The pharmacokinetics of propafenone was nonlinear and showed wide interindividual variations. In conclusion, a) the lower efficacy and delayed electrophysiologic effects of propafenone after intravenous administration suggest that longer infusion times are necessary for complete antiarrhythmic efficacy; b) the differences observed in the minimal effective concentrations of acute versus chronic oral therapy suggest the development of partial tolerance to propafenone during chronic treatment.
Similar content being viewed by others
References
Connolly SJ, Kates RE, Lebsack CS, et al. Clinical pharmacology of propafenone.Circulation 1983;68:589–596.
Podrid PJ, Cytryn R, Lown B. Propafenone: Noninvasive evaluation of efficacy.Am J Cardiol 1984;54:53D–59D.
Naccarella F, Bracchetti D, Palmieri M, et al. Propafenone for refractory ventricular arrhythmias: Correlation with drug plasma levels during long term treatment.Am J Cardiol 1984;54:1008–1014.
Hammil SC, Sorenson PB, Wood DL, et al. Propafenone for the treatment of refractory complex ventricular ectopic activity.Mayo Clin Proc 1986;61:98–103.
Kates RE, Yee GY, Winkle RA. Metabolite cumulation during chronic propafenone dosing in arrhythmia.Clin Pharmacol Ther 1985;37:610–614.
Siddoway LA, Thompson KA, McAllister CB, et al. Polymorphism of propafenone metabolism and disposition in man: Clinical and pharmacokinetic consequences.Circulation 1987;75:785–791.
Phillisborn VB, Gries J, Hoffman HP, et al. Pharmacological studies on propafenone and its main metabolite 5-hydroxypropafenone.Arzneim Forsh Drug Res 1984; 34(I):1489–1497.
Valenzuela C, Delgado C, Tamargo J. Electrophysiological effects of 5-hydroxypropafenone on guinea pig ventricular muscle fibers.J Cardiovasc Pharmacol 1987;40:523–529.
Frabetti L, Marchesini B, Capucci A, et al. Antiarrhythmic efficacy of propafenone: Evaluation of effective plasma levels following single and multiple doses.Eur J Clin Pharmacol 1986;30:665–671.
Brode E, Sachse R, Hoffman HD. Untersuchungen der analitik von propafenone mittels interner analogstandardsierung.Arzneim Forsh Drug Res 1982;32:1–6.
Marchesini B, Tomasi L. Effects of modifiers on the chromatographic behaviour of propafenone and its major metabolites.Chromatographia 1987;34:753–758.
Harrison PM, Tonkin AM, Dixon ST, et al. Determination of debrisoquine and its 4-hydroxy-metabolite in urine by high performance liquid chromatography.J Chromatography 1986;374:204–208.
Connolly S, Lebsack C, Winkle RA, et al. Propafenone disposition kinetics in cardiac arrhythmias.Clin Pharmacol Ther 1984;36:163–168.
Giani P, Landolina M, Giudici V, et al. Pharmacokinetics and pharmacodynamics of propafenone during acute and chronic administration.Eur J Clin Pharmacol 1988;34:187–194.
Evans PDA, Mahgoub A, Sloan TP, et al. A family and population study on the genetic polymorphism of debrisoquine oxidation in white British population.J Med Genetics 1980;17:102–105.
Nakamura K, Goto F, Ray WA, et al. Interethnic differences in genetic polymorphism of debrisoquine and mephenytoin hydroxylation between Japanese and Caucasian population.Clin Pharmacol Ther 1985;38:402–408.
Steiner E, Iselius L, Alvan G, et al. A family study of genetic and environmental factors determining polymorphic hydroxylation of debrisoquine.Clin Pharmacol Ther 1985; 38:394–401.
Barbey JT, Thompson KA, Echt DS, et al. Antiarrhythmic activity, electrocardiographic effects and pharmacokinetics of encainide metabolites o-desmethyl-encainide and 3-methoxy-o-desmethyl-encainide in man.Circulation 1988;77: 380–391.
Gillis AM, Kates RE. Myocardial uptake kinetics and pharmacodynamics of propafenone in the isolated perfused rabbit heart.J Pharmacol Exp Ther 1986;237:708–712.
Furlanello F, Disertori M, Vergara G, et al. Clinical evaluation of new antiarrhythmic agents: Experiences with propafenone.Int J Clin Pharm Res 1983;3:101–105.
Lullmann H, Timmermans PB, Ziegler A. Accumulation of drugs by resting or beating cardiac tissue.Eur J Pharmacol 1979;60:277–285.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Capucci, A., Boriani, G., Marchesini, B. et al. Minimal effective concentration values of propafenone and 5-hydroxy-propafenone in acute and chronic therapy. Cardiovasc Drug Ther 4, 281–287 (1990). https://doi.org/10.1007/BF01857646
Issue Date:
DOI: https://doi.org/10.1007/BF01857646