Summary
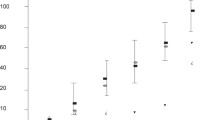
In the pregnant uterus unique immunological conditions must prevail. Since compounds of possible fetoplacental origin may modulate the local maternal immunological response, higher concentrations of such compounds in the vicinity of the conceptus might be expected. We have measured the concentrations of several compounds in peripheral and retroplacental plasma, mainly by immunological methods, from women delivered by caesarean section in pregnancy week 38–40. Significantly higher (P<0.001) retroplacental concentrations were found for human placental lactogen, prolactin, alpha-fetoprotein, tissue polypeptide antigen, non-conjugated estriol and progesterone. Peripheral plasma levels were significantly higher for cortisol (P<0.01), transcortin (P<0.001) and sex-hormone binding globulin (P<0.002). No difference was found for albumin, alpha2-macroglobulin, estradiol, placental alkaline phosphatase and human chorionic gonadotrophin. Significant correlations between retroplacental and maternal peripheral plasma levels were found for progesterone, cortisol, estradiol, estriol, transcortin, sex-hormone binding globulin, prolactin, human placental lactogen, human chorionic gonadotrophin, placental alkaline phosphatase, albumin and alpha2-macroglobulin. There were also significant correlations between estradiol and sex-hormone binding globulin as well as transcortin in peripheral maternal plasma.
Similar content being viewed by others
References
Adcock III EW, Teasdale F, August CS, Cox S, Meschia G, Battaglia FC, Naughton MA (1973) Human chorionic gonadotropin: its possible role in maternal lymphocyte suppression. Science 181:845–847
Beden M, Ferre F, Alsat E, Cedard L (1980) Regulation of steroidogenesis in the human placenta. J Steroid Biochem 12:17–24
Beham A, Weybora W, Lackinger E, Denk H, Björklund V, Björklund B (1986) Distribution of TPA and cytokeratins in gastrointestinal carcinomas as revealed by immunohistochemistry. Virchows Arch (A) 409:641–655
Ben-Jonathan N, Munsick RA (1980) Dopamine and prolactin in human pregnancy. J Clin Endocrinol Metab 51:1019–1025
Björklund B, Lundblad G, Björklund V (1958) Antigenicity of pooled human malignant and normal tissues by cyto-immunological technique: II Nature of tumor antigen. Int Arch Allergy 12:241–261
Boyer SH (1961) Alkaline phosphatase in human sera and placentae. Science 134:1002–1006
Braunsberg H, Reed MJ, Short F, Dias VO, Baxendale PM (1981) Changes in plasma concentrations of oestrogens and progesterone in women during anaesthesia and gynaecological operations. J Steroid Biochem 14:749–755
Bremme K, Eneroth P, Nilsson B Olsson M (1985) Maternal and cord serum levels of tissue polypeptide antigen (TPA) in normal pregnancies. Gynecol Obstet Invest 19:118–123
Briese Von V, Straube W, Mißner J (1983) Zur Konzentration einiger Serum-, Schwangerschafts-und Immunglobuline sowie Östriol im Retroplazentarblut. Zentralbl Gynäkol 105:845–854
Caldwell JL, Stites DP, Fudenberg HH (1975) Human chorionic gonadotropin: effects of crude and purified preparations on lymphocyte responses to phytohemagglutinin and allogeneic stimulation. J Immunol 115:1249–1253
De Moore P, Steeno O, Brosens I, Hendrikx A (1966) Data on transcortin in human plasma as studied by gel filtration. J Clin Endocrinol Metab 26:71–78
Durandy A, Fischer A, Mamas S, Dray F, Griscelli C (1982) Respective roles and interactions of T-lymphocyte and PGE2-mediated monocyte suppressive activities in human newborns and mothers at the time of delivery. Am J Reprod Immunol 2:127–134
Englebienne P (1984) The serum steroid transport proteins: biochemistry and clinical significance. Mol Aspects Med 7:313–396
Eriksson C-G, Eneroth P (1987) Determination of transcortin in serum by polyethylenglycol enhanced immunonephelometry. A comparison with equilibrium dialysis and radioimmunoassay. Clin Chim Acta 170:245–254
Farquharson RG, Klopper AI (1984) Progesterone concentrations in maternal and fetal blood. Br J Obstet Gynecol 91:133–137
Fishman WH, Bardawil WA, Habib HG, Anstiss CL, Green S (1972) The placental isoenzyme of alkaline phosphatase in sera of normal pregnancy. Am J Clin Pathol 57:65–74
Fuchs T, Hammarström L, Smith CIE, Brundin J (1981) In vitro induction of human suppressor T-cells by a chorionic gonadotropin preparation. J Reprod Immunol 3:75–84
Gaugas JM, Curzen P (1978) Polyamine interaction with pregnancy serum in suppression of lymphocyte transformation. Lancet I:18–20
Golander A, Barrett J, Hurley T, Barry S, Handwerger S (1979) Failure of bromocriptine, dopamine and thyrotropin-releasing hormone to affect prolactin secretion by human decidual tissue in vitro. J Clin Endocrinol Metab 49:787–789
Grudzinskas JG, Menabawey M, Wiley BA, Teisner B, Chard T (1979) The relation between the concentration of placental specific proteins in retroplacental and peripheral blood. Br J Obstet Gynecol 86:891–893
Hammarström L, Fuchs T, Smith CIE (1979) The immunodepressive effect of human glucoproteins and their possible role in the nonrejection process during pregnancy. Acta Obstet Gynecol Scand 58:417–422
Hussa RO (1981) Human chorionic gonadotropin, a clinical marker: review of its biosynthesis. Ligand Review, 3, [Suppl 2]:6–44
Jaffe RB (1986) Endocrine physiology of the fetus and fetoplacental unit. In: Yen SSC, Jaffe RB (eds) Reproductive endocrinology. WB Saunders, Philadelphia, p 741
Klopper A, Hughes G (1978) Placental secretion of oestrogens and protein hormones. Arch Gynecol 225:171–177
Lau HL, Linkins SE (1976) Alpha-fetoprotein. Am J Obstet Gynecol 124:533–554
Liggins GC, Forster CS, Grieves SA, Schwartz AL (1977) Control of parurition in man. Biol Reprod 16:39–56
Lizana J, Blad E (1983) Immunonephelometry of specific proteins in human seminal plasma. Clin Chem 29:618–623
Maslar IA, Kaplan BM, Luciano AA, Riddick DH (1980) Prolactin production by the endometrium of early human pregnancy. J Clin Endocrinol Metab 51:78–83
Morse JH, Stearns G, Arden J, Agosto GM, Canfield RE (1976) The effects of crude and purified human gonadotropin on in vitro stimulated human lymphocyte cultures. Cell Immunol 25:178–188
Nathrath WBJ, Heidenkummer P, Björklund V, Björklund B (1985) Distribution of tissue polypeptide antigen (TPA) in normal human tissues: immunohistochemical study on unfixed, methanol-, ethanol-, and formalin-fixed tissues. J Histochem Cytochem 33:99–109
Ohrlander S, Gennser S, Eneroth P (1976) Plasma cortisol levels in human fetus during parturition. Obstet Gynecol 48:381–387
Ryan KJ (1980) Placental synthesis of steroid hormones. In: Tulchinsky D, Ryan KJ (eds) Maternal-fetal endocrinology. WB Saunders, Philadelphia, p 3
Schindler AE (1982) Hormones in human amniotic fluid. Springer, Berlin Heidelberg new York
Siiteri PK, Murai JT, Hammond GL, Nisker JA, Raymoure WJ, Kuhn RW (1982) The serum transport of steroid hormones. Rec Prog Horm Res 38:457–510
Siiteri PK, Stites DP (1982) Immunologic and endocrine inter-relationships in pregnancy. Biol Reprod 26:1–14
Sippell WG, Gaede P, Fleischmann O, Bidlingmaier F, Knorr D, Troelle D (1978) Plasma concentrations of eight unconjugated steroids in mother and fetoplacental unit at term delivery. Acta Endocrinol (Copenh) [Suppl] 125:34
Smith ID, Shutt A, Shearman RP (1975) Fetal plasma steroid concentrations related to gestational age and method of delivery. J Steroid Biochem 6:657–662
Smith R, Klopper A, Hughes G, Wilson G (1979) The compartmental distribution of oestrogens and pregnancy specific β1 glyco-protein. Br J Obstet Gynecol 86:119–124
Stites DP, Siiteri PK (1983) Steroids as immunosuppressants in pregnancy. Immunol Rev 75:117–138
Szekeres-Bartho J, Csernus V, Hadnagy J, Pacsa AS (1983) Immunosuppressive effect of serum progesterone during pregnancy depends on the progesterone binding capacity of the lymphocytes. J Reprod Immunol 5:81–88
Szekeres-Bartho J, Hadnagy J, Pacsa AS (1985) The suppressive effect of progesterone on lymphocyte cytotoxicity: unique progesterone sensitivity of pregnancy lymphocytes. J Reprod Immunol 7:121–128
Szekeres-Bartho J, Kilar F, Falkay G, Csernus V, Török A, Pacsa AS (1985) The mechanism of the inhibitory effect of progesterone on lymphocyte cytotoxicity: 1. Progesterone-treated lymphocytes release a substance inhibiting cytotoxicity and prostaglandin synthesis. Am J Reprod Immunol Microbiol 9:15–18
Tulchinsky D, Simmer HH (1972) Sources of plasma 17 alpha-hydroxyprogesterone in human pregnancy. J Clin Endocrinol Metab 35:799–808
Tulchinsky D (1973) Placental secretion of unconjugated estrone, estradiol and estriol into the maternal and the fetal circulation. J Clin Endocrinol Metab 36:1079–1087
Tulchinsky D, Okada DM (1975) Hormones in human pregnancy, IV. Plasma progesterone. Am J Obstet Gynecol 121:293–299
Uriel J, Dupiers M, Rimbaut C, Buffe D (1981) Maternal serum levels of sex steroid-binding protein during pregnancy. Br J Obstet Gynecol 88:1229–1232
Weber K, Osborn M, Moll R, Wiklund B, Lüning B (1984) Tissue polypeptide antigen (TPA) is related to the nonepidermal keratins 8, 18 and 19 typical of simple and non-squamous epithelia: re-evaluation of a human tumor marker. EMBO J 3:2707–2714
Wilson EA, Finn AE, Rayburn W, Jawad MJ (1979) Corticosteroid-binding globulin and estrogens in maternal and cord blood. Am J Obstet Gynecol 135:215–217
Yachnin S (1976) Demonstration of the inhibitory effect of human alpha-fetoprotein on in vitro transformation of human lymphocytes. Proc Natl Acad Sci USA 73:2857–2861
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Norman, M., Eriksson, C.G. & Eneroth, P. A comparison between the composition of maternal peripheral plasma and plasma collected from the retroplacental compartment at caesarean section. Arch Gynecol Obstet 244, 215–226 (1989). https://doi.org/10.1007/BF01560085
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01560085