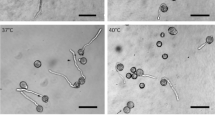
Summary
Microtubules inNicotiana tabacum pollen tube subprotoplasts reassembled in wave-like to concentric cortical arrays. Crosslinks between microtubules were either 15 or 80 nm in length. Cortical actin filaments showed different distributions; no colocalization like that in pollen tubes was observed. Degradation of actin filaments by cytochalasin D had no influence on microtubule organization. Degradation of microtubules and/or actin filaments did not affect outgrowth of the subprotoplasts. Organization of the microtubules occurred independent of the presence of the generative cell and/or the vegetative nucleus. No relation of actin filament and microtubule organization with organelle distribution could be detected.
Similar content being viewed by others
Abbreviations
- AFs:
-
actin filaments
- DAPI:
-
4′,6-diamidino-2-phenylindole
- EGTA:
-
ethylene glycol bis (2-amino ethylether) N,N,N′,N′-tetraacetic acid
- FITC:
-
fluorescein isothiocyanate
- MTs:
-
microtubules
- SPPs:
-
subprotoplasts
- TRITC:
-
tetramethyl rhodamine B isothiocyanate
References
Clayton L, Black CM, Lloyd CW (1985) Microtubule nucleating sites in higher plant cells identified by an auto-antibody against pericentriolar material. J Cell Biol 101: 319–324
Derksen J, Emons AM (1990) Microtubules in tip growth systems. In: Heath IB (ed) Tip growth in plant and fungal cells. Academic Press, San Diego, pp 147–181
- Traas JA (1984) Growth of tobacco pollen tubes in vitro; effects of drug interference with the cytoskeleton. In: Willemse MTM, Van West JL (eds) Proceedings 8th international symposium on sexual reproduction in seed plants, ferns and mosses, 20–24 August 1984. Pudoc, Wageningen, pp 64–70
—, Pierson ES, Traas JA (1985) Microtubules in vegetative and generative cells of pollen tubes. Eur J Cell Biol 38: 142–148
—, Wilms FHA, Pierson ES (1990) The plant cytoskeleton: its significance in plant development. Acta Bot Neerl 39: 1–8
Hagestedt T, Lichtenberg B, Wille H, Mandelkow E-M, Mandelkow E (1989) Tau protein becomes long and stiff upon phosphorylation: correlation between paracrystalline structure and degree of phosphorylation. J Cell Biol 109: 1643–1651
Kroh M, Knuiman B (1988) Development of subprotoplasts from in vitro-grown tobacco pollen tubes. Sex Plant Reprod 1: 103–113
Lancelle SA, Cresti M, Hepler PK (1987) Ultrastructure of the cytoskeleton in freeze-substituted pollen tubes ofNicotiana alata. Protoplasma 140: 141–150
Menzel D (1986) Visualization of cytoskeletal changes through the life cycle inAcetabularia. Protoplasma 134: 30–42
Nishida E, Kotani S, Kuwaki T, Sakai H (1982) Phosphorylation of microtubule associated proteins (MAPs) controls both microtubule assembly and MAPs actin interaction. In: Sakai H, Mahri H, Borisy JJ (eds) Biological functions of microtubules and related structures. Academic Press, New York, pp 285–295
Pierson ES, Derksen J, Traas JA (1986) Organization of microfilaments and microtubules in pollen tubes grown in vitro or in vivo in various angiosperms. Eur J Cell Biol 41: 14–18
—, Kengen HMP, Derksen J (1989) Microtubules and actin filaments colocalize in pollen tubes ofNicotiana tabacum L. andLilium longiflorum Thunb. Protoplasma 150: 75–77
—, Lichtscheidl IK, Derksen J (1990) Structural behaviour of organelles in living pollen tubes ofLilium longiflorum. J Exp Bot 41: 1461–1468
Rutten TLM, Derksen J (1990) Organization of actin filaments in regenerating and outgrowing subprotoplasts from pollen tubes ofNicotiana tabacum L. Planta 180: 471–479
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26: 31–42
Steer MW (1990) Role of actin in tip growth. In: Heath IB (ed) Tip growth in plant and fungal cells. Academic Press, San Diego, pp 119–145
—, Steer JM (1989) Pollen tube tip growth. New Phytol 111: 323–358
Tiwari SC, Polito VS (1988) Spatial and temporal organization of actin during hydration activation and germination of pollen inPyrus communis L.: a population study. Protoplasma 147: 5–15
Traas JA, Doonan JH, Rawlins DJ, Shaw PJ, Watts J, Lloyd CW (1987) An actin network is present in the cytoplasm throughout the cell cycle of carrot cells and associates with the dividing nucleus. J Cell Biol 105: 387–395
Wick SM, Duniec J (1983) Immunofluorescence microscopy of tubulin and microtubule arrays in plant cells. I. Preprophase band development and concomitant appearance of nuclear envelop-associated tubulin. J Cell Biol 97: 235–243
Yamauchi T, Fujisawa H (1988) Regulation of the interaction of actin filaments with microtubule-associated protein 2 by calmodulin-dependent protein kinase II. Biochim Biophys Acta 968: 77–85
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rutten, T.L.M., Derksen, J. Microtubules in pollen tube subprotoplasts: organization during protoplast formation and protoplast outgrowth. Protoplasma 167, 231–237 (1992). https://doi.org/10.1007/BF01403386
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01403386