Abstract
We propose a system of simple rules which give the binding energy and approximate ground state structure of a nucleus, dependent only on the number of protons and neutrons making it up. This “nuclear code” is based on theN-quark Schrödinger equation and uses ideas familiar from atomic chemistry. We demonstrate some of the important ramifications by computing the4He elastic form factor. The nuclear code performs better than its atomic counterpart.
Similar content being viewed by others
References
For a review, see Ynduŕain, F.J.: Quantum chromodynamics, an introduction to the theory of quarks and gluons. Berlin, Heidelberg, New York: Springer 1983
See for example Otto, S.W., Stack, J.D.: Phys. Rev. Lett.52, 2328 (1984); Barkai, D. et al: Phys. Rev. D30, 2201 (1984);
Koite, Y.: Phys. Rev. Lett.59, 962 (1987)
Politzer, H.D.: Phys. Rep.14C, 129 (1974); Close, F.E.: An introduction to quarks and partons. New York: Academic Press 1979; Perkins, Donald H.: Introduction to high energy physics, 3rd Edn. Reading: Addison-Wesley 1987
Laermann, E. et al.: Phys. Lett. B173, 437 (1986)
Willey, R.S.: Phys. Rev. D18, 270 (1978); Fishbane, P.M., Grisaru, M.T.: Phys. Lett. B74, 98 (1978); Gavela, M.B. et al.: Phys. Lett. B82, 431 (1979)
Gleeson, A., Morley, P.D., Plümer, M.: Phys. Rev. D36, 2575 (1987)
Kiefer, Mark Linden: Ph. D. dissertation, Iowa State University (1983), available from University Microfilm International, 300 North Zeeb Road, Ann Arbor, MI 48106 USA
Isgur, N., Karl, G.: Phys. Lett. B72, 109 (1977)
Williams, S.A., Margetan, F.J., Morley, P.D., Pursey, D.L.: Phys. Rev. Lett.49, 771 (1982); Morley, P.D., Pursey, D.L., Williams, S.A.: Constituent quark model of the nucleon-nucleon interaction, Phys. Rev. C (accepted); Williams, S.A., Margeton, F.J., Morley, P.D., Pursey, D.L.: Constituent quark model of the nucleon-nucleon interaction, application to the deuteron. Phys. Rev. C (accepted)
Reed-Margeton, D.: Ph. D. dissertation, Iowa State University (1983), available from University Microfilms International, 300 North Zeeb Road, Ann Arbor, MI 48106 USA
Johnson, K.: Acta. Phys. Pol. B6, 865 (1975)
Janz, George J.: Estimation of thermodynamic properties of organic compounds. New York: Academic Press 1958
Janz, George J.: Loc. cit., Table 7.4
Morley, P.D.: Universidad Tecnica Federico Santa MariaUSM-24 (1984) unpublished, Valparaiso, Chile
Robson, D.: Nucl. Phys. A308, 381 (1978)
Pauling, Linus: Science150, 297 (1965); Nature208, 174 (1965); Phys. Rev. Lett.15, 499 (1965); Proc. Nat. Acad. Sci.54, 984 (1965); Rev. Rowmaine Phys.II, 825 (1966); Proc. Nat Acad. Sci.46, 2175 (1967);64, 807 (1969); Phys. Rev. C22, 1585 (1980); Proc. Nat. Acad. Sci.78, 5296 (1981)
deShalit, Amos, Feshbach, Herman: Theoretical nuclear physics, Vol. 1. New York: John Wiley & Sons 1974
Hauge, P.S., Williams, S.A., Duffey, G.H.: Phys. Rev. C4, 1044 (1971)
Janz, George J.: Loc. cit., Chap. 7, Sect. 3
This is the Coulomb corrected binding energy in MeV predicted for a nucleus ofA nucleons,Z of which are protons. The Coulomb energy is calculated from (16)
Experimental binding energy per nucleon for theAZ nucleus, except forA=5, 8 where it is the total unbound energy for5He,8Be respectively
This is the H(6) solution taken from [9]
Because of geometry, the maximum binding energy is not realizable; see text
The bonds predict the Coulomb uncorrected BE: E(8)=2E(4). The 1.89 MeV is the estimated theoretical Coulomb energy
Morley, P.D., Williams, S.A.: Z. Phys. A — Atomic Nuclei331, 239 (1988)
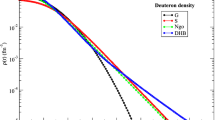
McCarthy, J.S., Sick, I., Whitney, R.R.: Phys. Rev. C15, 1396 (1977); Arnold, R.G. et al.: Phys. Rev. Lett.40, 1429 (1978)
Day, D.: Private communication
See Arnold, R.G. et al.: Phys. Rev. Lett.40, 1429 (1978)
Hamermesh, M.: Group theory. Reading: Addison-Wesley 1962
Williams, S.A., Pursey, D.L.: J. Math. Phys.17, 1383 (1976)
Donnelly, T., Williams, S.A., Sick, I.: Rev. Mod. Phys.56, 461 (1984)