Summary
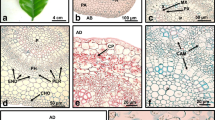
The cell ultrastructure in three types of callus obtained from leaf explants ofAesculus hippocastanum L. has been studied. Remarkable differences have been shown between the cells of the forerunner E1 callus and those of the callus arising from it, according to the culture conditions.
The peculiar characteristics of E1 are the scarcity of intercellular spaces and the occurrence of autophagic vacuoles in the cells.
An embryogenic friable callus (E2) is formed in time when E1 is maintained on solid culture medium. The E2 cells show cytological features typical of a higher metabolic level and contain starch. Diffused middle lamella digestion leads to the detachment of small embryogenic cell aggregates consisting of vacuolated parenchymatous-like cells and small meristematic cells which may be regarded as embryoids initials.
Shaking E1 in the same liquid medium and subsequent culture on solid medium lead to the differentiation of a non-embryogenic callus (NE), whose cells are very large and highly vacuolated, devoid of starch and with organelle-rich cytoplasm. The NE callus shows a high degree of growth, but does not attain embryogenic competence in time.
Similar content being viewed by others
Abbreviations
- c :
-
cell
- cr :
-
crystal
- cw :
-
cell wall
- d :
-
dictyosome
- er :
-
endoplasmic reticulum
- m :
-
mitochondrion
- mb :
-
microbody
- n :
-
nucleus
- p :
-
plastid
- s :
-
starch
- v :
-
vacuole
References
Armstrong CL, Green CE (1985) Establishment and maintenance of friable embryogenic maize callus and the involvement of L-proline. Planta 164: 207–214
Barwale UB, Kerns HR, Widholm JM (1986) Plant regeneration from callus cultures of several soybean genotypes via embryogenesis and organogenesis. Planta 167: 473–481
Benbadis A (1973) Analyse des aptitudes embriogènes de divers types de cellules isolées deDaucus carota L. Bull Soc Bot Fr Mém 120: 223–234
Button J, Kochba J, Bornman CH (1974) Fine structure of embryoid development from embryogenic ovular callus of “Shamouti orange” (Citrus sinensis Osb.). J Exp Bot 25: 446–457
Cronshaw J (1964) Crystal containing bodies of plant cells. Protoplasma 59: 318
Dameri RM, Profumo P, Gastaldo P, Caffaro L (1986) Callus formation and embryogenesis with leaf explants ofAesculus hippocastanum L. J Plant Physiol 126: 93–96
Davey MR, Street HE (1971) Studies on the growth in culture of plant cells IX. Additional features of the fine structure ofAcer pseudoplatanus L. cells cultured in suspension. J Exp Bot 22: 90–95
Dennis DT, Miernyk JA (1982) Compartimentation of nonphotosynthetic carbohydrate metabolism. Ann Rev Plant Physiol 33: 27–50
Dickinson HG, Helsop-Harrison J (1970) The ribosome cycle, nucleoli, and cytoplasm nucleoids in the meiocytes ofLilium. Protoplasma 69: 187–200
Dos Santos AVP, Cutter EG, Davey MR (1983) Origin and development of somatic embryos inMedicago sativa L. (Alfalfa). Protoplasma 117: 107–115
Dunwell JM, Sunderland N (1974) Pollen ultrastructure in anther cultures ofNicotiana tabacum. II. Changes associated with embryogenesis. J Exp Bot 74: 363–373
Hall JL, Flowers TI, Roberts RM (1982) Plant cell structure and metabolism. Longman, London New York
Halperin W, Jensen WA (1967) Ultrastructural changes during growth and embryogenesis in carrot cell cultures. J Ultrastruct Res 18: 428–443
—,Wetherell DF (1964) Adventive embryony in tissue cultures of the wild carrot,Daucus carota. Amer J Bot 51: 274–283
Huang AHC, Trelease RN, Moore TS Jr (1983) Plant peroxisomes. Academic Press, New York London
Kadej F, Kadej A (1981) Ultrastructural organization of the tomato embryosac at the time of fertilization. Acta Soc Bot Pol 50: 139–142
Kamo KK, Becwar MR, Hodges TK (1985) Regeneration ofZea mays from embryogenic callus. Bot Gaz 146: 327–334
Karlsson SB, Vasil IK (1986) Morphology and ultrastructure of embryogenic cell suspension cultures ofPanicum maximum (Guinea grass) andPennisetum purpureum (Napier grass). Amer J Bot 73: 894–901
Kochba J, Spiegel-Roy P, Neuman H, Saad S (1978) Stimulation of embryogenesis in citrus ovular callus by ABA, Ethepon, CCC and Alar and its suppression by GA3. Z Pflanzenphysiol 89: 427–432
Konar RN, Thomas E, Street HE (1972) Origin and structure of embryoids arising from epidermal cells of the stem ofRanunculus sceleratus L. J Cell Biol 11: 77–93
Kononowitcz H, Kononowitcz AK, Janick J (1984) Asexual embryogenesis via callus ofTheobroma cacao L. Z Pflanzenphysiol 113: 347–358
Lu C, Vasil IK (1981) Somatic embryogenesis and plant regeneration from freely suspended cells and cell groups ofPanicum maximum Jacq. Ann Bot 48: 543–548
Mackenzie A, Helsop-Harrison J, Dickinson HG (1967) Elimination of ribosomes during meiotic profase. Nature 215: 997–999
Nabors MW, Heyser JW, Dykes ThA, de Mott KJ (1983) Longduration, high frequency plant regeneration from cereal tissue cultures. Planta 157: 385–391
Profumo P, Dameri RM, Cremona Orsino I (1976) Frammenti cotiledonari diAesculus hippocastanum L. coltivatiin vitro. Primi dati sul comportamento dell'amido e dell'escina. Giorn Bot Ital 110: 155–171
—,Dameri Orsino RM, Modenesi P (1980) Aescin content in calluses from expiant ofAesculus hippocastanum cotyledons growthin vitro. Giorn Bot Ital 114: 25–28
—,Gastaldo P, Dameri RM, Caffaro L (1986) Histological study of calli and embryoids of leaf expiants ofAesculus hippocastanum L. J Plant Physiol 126: 97–103
Radojević L, Vujičić JR, Nešković M (1975) Embryogenesis in tissue culture inCorylus avellana L. Z Pflanzenphysiol 77: 33–41
—,Zylbergers L, Kovoor J (1980) Etude ultrastructurale des embryons androgénétiques d'Aesculus hippocastanum L. Z Pflanzenphysiol 98: 255–261
Rascio N, Casadoro G (1985) Effects of light intensity on the E 283 B olive necrotic maize mutant. Cytobios 42: 179–191
— —,Ramina A, Masia A (1985) Structural and biochemical aspects of peach fruit abscission (Prunus persica L. Batsch). Planta 164: 1–11
—,Orsenigo M, Arboit D (1976) Prolamellar body transformation with increasing cell age in the maize leaf. Protoplasma 90: 253–263
Rengel Z, Jelaska SJ (1986) Somatic embryogenesis and plant regeneration from seedling tissues ofHordeum vulgare L. J Plant Physiol 124: 385–392
Sangwan RS (1986) Formation and cytochemistry of nuclear vacuoles during meiosis inDatura. Eur J Cell Biol 40: 210–218
Sexton R, Jameson GGC, Allan MHIL (1977) An ultrastructural study of abscission zone cells with special reference to the mechanisms of enzyme secretion. Protoplasma 91: 369–387
Sharp WR, Sondahl MR, Caldas LS, Maraffa SB (1980) The physiology ofin vitro asexual embryogenesis. Hortic Rev 2: 268–310
Smith SM, Street HE (1974) The decline of embryogenic potential as callus and suspension cultures of carrot (Daucus carota L.) are serially subcultured. Amer J Bot 38: 223–241
Sutton-Jones B, Street HE (1968) Studies on the growth in culture of plant cells. III. Changes in fine structure during the growth ofAcer pseudoplatanus L. cells in suspension culture. J Exp Bot 19: 114–118
Thomas E, Konar RN, Street HE (1972) The fine structure of the embryogenic callus ofRanunculus sceleratus. J Cell Sci 11: 95–109
— — — (1973) Fine structural studies of embryogenesis in a callus ofRanunculus sceleratus. Bull Soc Bot Fr Mém 120: 207–214
Thorpe TA, Joy IV RW, Leung DWM (1986) Starch turnover in shoot-forming tobacco callus. Physiol Plant 66: 58–62
Vasil V, Vasil IK (1982) Characterization of an embryogenic cell suspension culture derived from cultured inflorescences ofPennisetum americanum (Pearl Millet,Gramineae). Amer J Bot 69: 1441–1449
— — (1986) Plant regeneration from friable embryogenic callus and cell suspension cultures ofZea mays L. J Plant Physiol 124: 399–408
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Profumo, P., Gastaldo, P. & Rascio, N. Ultrastructural study of different types of callus from leaf expiants ofAesculus hippocastanum L.. Protoplasma 138, 89–97 (1987). https://doi.org/10.1007/BF01281017
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01281017