Summary
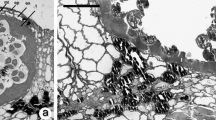
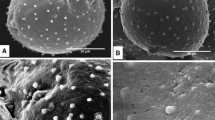
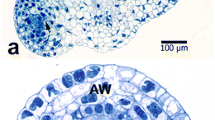
Anthers ofBetula pendula were collected at regular intervals during the dormancy period until anthesis. Ultrathin sections of maturing pollen grains were especially stained for polysaccharides and proteins and examined with TEM to determine whether structural or/and chemical changes in the pollen wall occur during the dormancy period of the plant life cycle. At the beginning of the dormancy period, the microspore wall consists of a well developed tectum, columellae and a foot layer. Spinules and supratectal elements are prominent. Microchannels are present in the tectum but not obvious in the foot layer. Some of the columellae are not clearly connected with the foot layer, but some connections are evident. Pores are filled with a thick fibrillar network “flocculent material”. The cytoplasm is packed with starch grains and lipid globules. The stainability for acidic and neutral polysaccharides and protein was tested, and variations in the pollen wall are illustrated. As temperature increased towards the end of dormancy and before anthesis there is obvious differentiation in the morphology of the pollen wall. The granular fibrillar layer beneath the pore and the Zwischenkörper are the most variable part of the wall. Different histochemical reactions observed in different layers at the aperture sites indicate different functions of these layers.
Similar content being viewed by others
References
Beer R (1906) On the development of the pollen grain and anther of some Onagraceae. Bot Zentralbl 19: 286–313
Benedetti EL, Bertolini B (1963) The use of phosphotungstic acid as a stain for the plasma membrane. J R Microsc Soc 81: 219–222
Charzynska M, Murgia M, Cresti M (1990) Microspore ofSecale cereale as a transfer cell type. Protoplasma 158: 26–32
Christensen J, Horner HT (1974) Pollen pore development and its special orientation during microsporogenesis in the grassSorghum bicolor. Amer J Bot 61: 604–623
Ciampolini F, Nepi M, Pacini E (1993) Tapetum development inCucurbita pepo (Cucurbitaceae). In: Hesse M, Pacini E, Willemse M (eds) The tapetum. Cytology, function, biochemistry and evolution. Plant Syst Evol [Suppl] 7: 13–22
Claugher D, Rowley JR (1987)Betula pollen grain substructure revealed by fast atom etching. Pollen Spores 29: 5–20
Doyle A, Van Campo M, Lugardon B (1975) Observations on exine structure ofEucommiidites and lower cretaceous angiosperm pollen. Pollen Spores 17: 429–486
Dunbar A, Rowley JR (1984)Betula pollen development before and after dormancy: exine and inline. Pollen Spores 24: 299–338
El-Ghazaly G, Jensen W (1986) Studies of the development of wheat (Triticum aestivum) pollen: formation of the pollen aperture. Can J Bot 64: 3141–3154
— — (1987) Development of wheat (Triticum aestivum) pollen. II. Histochemical differentiation of wall and Ubisch bodies during development. Amer J Bot 74: 1396–1418
Erdtman G, Praglowski J, Takeoka M (1962) Zur Bedeutung der Pollenmorphologie für pollenanalytische Vegetationsforschung. Veröff Geobot Inst Rübel Zürich 37: 57–59
Grote M (1989) Techniques to preserve soluble surface components in birch pollen wall: a scanning and transmission electron microscopy study. J Histochem Cytochem 37-7: 981–987
—, Fromme HG (1984 a) Ultrastructural demonstration of a glycoproteinic surface coat in allergenic pollen grains by combined cetylpyridinium chloride precipitation and silver proteinate staining. Histochemistry 81: 171
— — (1984 b) Immunoelectron microscopic localization of diffusible birch pollen antigens in ultrathin sections using the protein A/gold technique. Histochemistry 81: 489
Heslop-Harrison J, Heslop-Harrison Y (1991) Structural and functional variatin in pollen intines. In: Blackmore S, Barnes SH (eds) Pollen and spores, patterns of diversification. Oxford University Press, Oxford, pp 331–343 (The Systematics Association special volume, no 44)
Heywood VH (1978) Flowering plants of the world. Oxford University Press, Oxford
Kandasamy MK, Kristen U (1989) Ultrastructural responses of tobacco pollen tubes to heat shock. Protoplasma 153: 104–110
Knox RB, Heslop-Harrison J (1970) Pollen wall protein: localization and enzymic activity. J Cell Sci 6: 1–27
Marinozzi V (1967) Reaction de l'acide phosphotungstique avec la mucine et les glycoprotéines des plasmamembranes. J Microsc 6: 682–692
- Marinozzi V (1968) Phosphotungstic acid (PTA) as a stain for polysaccharides and glycoproteins in electron microscopy. In: Proceedings 4th European Regional Conference on Electron Microscopy, Rome, pp 55–56
Nepi M, Pacini E (1993) Pollination, pollen viability and pistil receptivity inCucurbita pepo. Ann Bot 72: 527–536
Pease DC (1968) Phosphotungstic acid as an electron stain. In: Proceedings 26th Annual EMSA Meeting, Calitor's, Baton Rouge, pp 36–37
Praglowski J (1962) Notes on the pollen morphology of Swedish trees and shrubs. Grana Palynol 3: 45–65
Rowley JR (1981) Pollen wall characters with emphasis upon applicability. Nord I Bot l: 357–380
—, Dahl AO (1977) Pollen development inArtemisia vulgaris with special reference to glycocalyx material. Pollen Spores 19: 169–284
Schrauwen JAM, Reijnen WH, Deleeuw HCGM, Van Herpen MMA (1986) Response of pollen to heat stress. Acta Bot Neerl 35: 321–327
Seiberler S, Scheiner O, Kraft D, Lonsdale D, Valenta R (1994) Characterization of a birch pollen allergen, Bet v II, representing a novel class of calcium-binding proteins: specific expression in mature pollen and dependence of patients IgE-binding on protein bound calcium. In: Heberle-Bors E, Hesse M, Vicente O (eds) Frontiers in sexual plant reproduction research, abstract book, University of Vienna, Vienna, p 100
Southworth D (1973) Cytochemical reactivity of pollen walls. J Histochem Cytochem 21: 73–80
—, Singh MB, Hough T, Smart IJ, Taylor P, Knox RB (1988) Antibodies to pollen exines. Planta 176: 482–487
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26: 31–43
Sulkinoja M, Valanne T (1980) Polyembryony and abnormal germination inBetula pubescens subsp.tortuosa. Rep Kevo Subarctic Res Stat 16: 31–37
Swoboda I, Dant TCH, Heberle-Bors E, Vicente O (1994) Expression of Bet v 1, the major birch pollen allergen, during anther development. An in situ hybridization study. In: Heberle-Bors E, Hesse M, Vicente O (eds) Frontiers in sexual plant reproduction research, abstract book, University of Vienna, Vienna, p 111
Takeoka T, Stix E (1963) On the fine structure of the pollen walls in some Scandinavian Betulaceae. Grana Palynol 4: 160–188
Thiéry JP (1967) Mise en évidence des polysaccharides sur coupes fines au microscopic électronique. J Microsc 6: 987–1018
Völker W, Sinclair NJ, Kalveram K, Rubenek H (1986) Is the surface layer from hazelnut pollen which is precipitated by Cuprolinic blue an effective antigen in hay fever patients? Histochemistry 84: 57
Wihl JÅ, Ipsen B, Nüchel Petersen B, Munch EP, Janniche H, Lövenstin H (1988) Immunotherapy with partially purified and standardized tree pollen extracts. Allergy 43: 363–369
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Ghazaly, G., Grafström, E. Morphological and histochemical differentiation of the pollen wall ofBetula pendula Roth, during dormancy up to anthesis. Protoplasma 187, 88–102 (1995). https://doi.org/10.1007/BF01280236
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01280236