Abstract
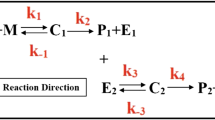
The theory of linear systems analysis is applied to the evaluation of induced drug removal processes. The rate and extent of removal are determined by deconvolution for the case of phenobarbital removal from the systemic circulation by orally administered activated charcoal. The proposed method is model independent in the sense that no specific models of intrinsic or induced pharmacokinetic processes are required, and it is readily adapted to the analysis of most types of induced removal processes (hemodialysis, peritoneal dialysis, etc.). Application of the approach indicates that phenobarbital was removed from the systemic circulation to an extent of 25–53% following multiple oral doses of activated charcoal in healthy human subjects.
Similar content being viewed by others
References
D. J. Cutler. Linear systems analysis in pharmacokinetics.J. Pharmacokin. Biopharm. 6:265–282 (1978).
P. Veng-Pedersen. Pharmacokinetic analysis by linear systems approach I: cimetidine bioavailability and second peak phenomenon.J. Pharm. Sci. 70:32–38 (1981).
D. J. Cutler. Numerical deconvolution by least squares: Use of prescribed input functions.J. Pharmacokin. Biopharm. 6:227–241 (1978).
D. J. Cutler. Assessment of rate and extent of drug absorption.Pharm. Ther. 14:123–160 (1981).
P. Veng-Pedersen. Model independent method of analyzing input in linear pharmacokinetic systems having polyexponential impulse response I: Theoretical analysis.J. Pharm. Sci. 69:298–305 (1980).
P. Veng-Pedersen. Model independent method of analyzing input in linear pharmacokinetic systems having polyexponential impulse response II: Numerical evaluation.J. Pharm. Sci. 69:305–312 (1980).
W. R. Gillespie and P. Veng-Pedersen. Gastrointestinal bioavailability: Determination ofin vivo release profiles of solid oral dosage forms by deconvolution.Biopharm. Drug Dispos. 6:351–355 (1985).
W. R. Gillespie and P. Veng-Pedersen. A polyexponential deconvolution method. Evaluation of the “gastrointestinal bioavailability” and meanin vivo dissolution time (MDT) of some ibuprofen dosage forms.J. Pharmacokin. Biopharm. 13:289–307 (1985).
P. Veng-Pedersen. An algorithm and computer program for deconvolution in linear pharmacokinetics.J. Pharmacokin. Biopharm. 8:463–481 (1980).
D. D. Schottelius, personal communication.
D. D. Schottelius. Homogeneous immunoassay system (EMIT) for quantitation of antiepileptic drugs in biological fluids. In C. E. Pippenger, J. K. Perry, and H. Kutt (eds.),Antiepileptic Drugs: Quantitative Analysis and Interpretation, Raven Press, New York, 1978, pp. 95–108.
P. Veng-Pedersen. Curve fitting and modeling in pharmacokinetics and some practical experiences with NONLIN and a new program FUNFIT.J. Pharmacokin. Biopharm. 5:513–531 (1977).
A. V. Wolf, D. G. Kemp, and J. E. Kiley. Artificial kidney function: Kinetics of hemodialysis.J. Clin. Invest. 30:1062–1070 (1951).
T. P. Gibson and H. A. Nelson. Drug kinetics and artificial kidneys.Clin. Pharmacokinet. 2:403–406 (1977).
M. J. Berg, W. G. Berlinger, M. J. Goldberg, R. Spector, and G. F. Johnson. Acceleration of the body clearance of phenobarbital by oral activated charcoal.N. Engl. J. Med. 307:642–644 (1982).
M. J. Goldberg and W. G. Berlinger. Treatment of phenobarbital overdose with activated charcoal.J. Am. Med. Assoc. 247:2400–2401 (1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gillespie, W.R., Veng-Pedersen, P., Berg, M.J. et al. Linear systems approach to the analysis of an induced drug removal process. Phenobarbital removal by oral activated charcoal. Journal of Pharmacokinetics and Biopharmaceutics 14, 19–28 (1986). https://doi.org/10.1007/BF01059281
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059281