Abstract
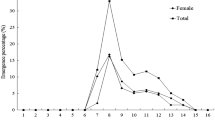
Egg depositional rates of onion flies, Delia antiqua(Meigen), injected thoracically with extracts of male paragonial glands were identical (14.5 eggs/female/ day) to those of normally mated females. Moreover, when continuously exposed to males, extract-injected females refused to mate and produced unfertilized eggs for the duration of the > 15- day experiment. For this normally monocoitic dipteran, <1 male equiv of paragonial secretion completely reproduced the ovipositional responses characteristic of normal mating, and this effect required no involvement of the genitalia or genital chamber. We suggest that the receptor for the active chemical (s) (sex peptide?) would be an excellent target for biorational insect control by sterilization. Moreover, these primer sex pheromones might play an important role in insect reproductive isolation and evolution.
Similar content being viewed by others
References
Burnet, B., Connolly, K., Kearney, M., and Cook, R. (1973). Effects of male paragonial gland secretion on sexual receptivity and courtship behaviour of femaleDrosophila melanogaster.J. Insect Physiol. 19: 2421–2431.
Chen, P. S. (1976). Species-specific protein patterns inDrosophila paragonial glands.Experientia 32: 549–551.
Chen, P. S., and Balmer, J. (1989). Secretory proteins and sex peptides of the male accessory gland inDrosophila sechellia.J. Insect Physiol. 35: 759–764.
Chen, P. S., and Diem, C. (1961). A sex-specific ninhydrin-positive substance found in the paragonia of adult males ofDrosophila melanogaster.J. Insect Physiol. 7: 289–298.
Chen, P. S., Stumm-Zollinger, E., Aigaki, T., Balmer, J., Bienz, M., and Bohlen, P. (1988). A male accessory gland peptide that regulates reproductive behavior of female D. melanogaster.Cell 54: 291–298.
Craig, G. B. (1967). Mosquitoes: Female monogamy induced by male accessory gland substance.Science 156: 1499–1501.
Fox, A. S. (1956). Chromatographie differences between males and females inDrosophila melanogaster and the role of the X and Y chromosomes.Physiol. Zool. 29: 288–298.
Fuchs, M. S., Craig, G. B., and Hiss, E. A. (1968). The biochemical basis of female monogamy in mosquitoes. I. Extraction of the active principle fromAedes aegypti.Life Sci. 7: 835–839.
Fuyama, Y. (1983). Species-specifity of paragonial substances as an isolating mechanism inDrosophila.Experientia 39: 190–192.
Garcia-Bellido, A. (1964). Das Sekret der Paragonien als Stimulus der Fekundität bei Weibchen vonDrosophila melanogaster.Z. Naturforsch. 19b: 491–495.
Gillott, C. (1988). Arthropoda-Insecta. In Adyodi, K. G., and Adyodi, R. D. (eds.),Reproductive Biology of Invertebrates, Vol. III. Accessory Sex Glands, Oxford and IBH, New Delhi, pp. 319–471.
Gottschewski, G. (1937). Künstliche Befruchtung beiDrosophila.Naturwissenschaften 25: 650.
Harris, M. O., Keller, J. E., and Miller, J. R. (1987). Responses to n-dipropyl disulfide by ovipositing onion flies: effects of concentration and site of release.J. Chem. Ecol. 13: 1261–1277.
Havukkula, I. J., and Miller, J. R. (1987). Daily periodicity in the ovipositional behavior of the onion fly,Delia antiqua (Diptera: Anthomyiidae).Environ. Entomol. 16: 41–44.
Hliss, E. A., and Fuchs, M. S. (1972). The effect of matrone on oviposition in the mosquito,Aedes aegypti.J. Insect Physiol. 18: 2217–2227.
Knipling, E. F. (1979).The Basic Principles of Insect Population Suppression and Management, U.S. Department of Agriculture, Agriculture Handbook No. 512, Washington, DC.
Kummer, H. (1960). Experimentelle Untersuchengen zur Wirkung von Forpflanzungs-Faktoren auf die Lebensdaur vonDrosophila melanogasrer-Weibchen. Z. vergl. Physiol.44: 642–679.
Leahy, M. G. (1966). Egg deposition inD. elanogaster increased by transplant of male paragonia.Drosoph. Info. Serv. 41: 145.
Leahy, M. G., and Craig, G. B. (1965). Male accessory gland substances as a stimulant for oviposition inAedes aegypti andA. albopictus.Mosq. News 25: 448–452.
Leahy, M. G., and Lowe, M. L. (1967). Purification of the male factor increasing egg deposition inDrosophila melanogaster.Life Sci. 6: 151–156.
Leopold, R. A. (1976). The role of male accessory glands in insect reproduction.Annu. Rev. Entomol. 21: 199–221.
Leopold, R. A., Terranova, A. C., Thorson, B. J., and Degrugillier, M. E. (1971). The biosynthesis of the male housefly accessory secretion and its fate in the mated female.J. Insect Physiol. 17: 987–1003.
Manning, A. (1962). A sperm factor affecting the receptivity ofDrosophila melanogaster females.Nature 194: 252–253.
Martin, J. S., and McEwen, F. L. (1982). Frequency of mating in the onion maggot,Hylemya antiqua (Diptera: Anthomyiidae).Can. Entomol. 114: 647–648.
Morrison, P. E., Venkatesh, K., and Thompson, B. (1982). The role of male accessory-gland substance on female reproduction with some observations of spermatogenesis in the stable fly.J. Insect Physiol. 28: 607–614.
Mowry, T. M., Adams, T., and Miller, J. R. (1987). Mechanoptical transducer for quantifying activity in small insect muscles.J. Insect Physiol. 33: 867–873.
Mowry, T. M., Keller, J. E., and Miller, J. R. (1989). Onion fly (Diptera: Anthomyiidae) oviposition as influenced by substrate holes and particle size.Ann. Entomol. Soc. Am. 82: 126–131.
Raina, A. K., and Gade, G. (1988). Insect peptide nomenclature.Insect Biochem. 18: 785–787.
Ribeiro, J. M. C. (1988). Can satyrs control pests and vectors?J Med. Entomol. 25: 431–440.
Ribeiro, J. M. C., and Spielman, A. (1986). The satyr effect: A model predicting parapatry and species extinction.Am. Nat. 128: 513–528.
Riemann, J. G., and Thorson, B. J. (1969). Effect of male accessory material on oviposition and mating by female houseflies.Ann. Entomol. Soc. Am. 62: 828–834.
Riemann, J. G., Moen, D. J., and Thorson, B. J. (1967). Female monogamy and its control in houseflies.J. Insect Physiol. 13: 407–418.
Smith, P. H., Barton Browne, L., and van Gerwin, A. C. M. (1989). Causes and correlates of loss and recovery of sexual receptivity inLucilia cuprina females after their first mating.J. Insect Behav. 2: 325–337.
Smith, P. H., Gillott, C., Barton Browne, L., and van Gerwin, A. C. M. (1990). The mating-induced refractoriness ofLucilia cuprina females: Manipulating the male contribution.Physiol. Entomol. 15: 469–481.
Swailes, G. E. (1971). Reproductive behavior and effects of the male accessory gland substance in the cabbage maggot,Hylemya brassicae.Ann. Entomol. Soc. Am. 64: 176–179.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Spencer, J.L., Bush, G.L., Keller, J.E. et al. Modification of female onion fly,Delia antiqua (Meigen), reproductive behavior by male paragonial gland extracts (Diptera: Anthomyiidae). J Insect Behav 5, 689–697 (1992). https://doi.org/10.1007/BF01047980
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01047980