Abstract
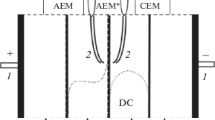
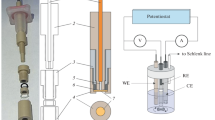
The pH on the surface of an ion-exchange membrane was measured with a microprobe in a laboratory scale chlor-alkali cell to study the leakage of OH− through the membrane. The solution pH in the vicinity of the membrane facing the anode was found to be considerably higher than that in the bulk solution because of penetration and leakage of OH−. The pH varied with the membrane type. This explains why the carboxylate membrane is not protonated and can be used as a separator in chlor-alkali cells.
Similar content being viewed by others
References
C. W. Walton and R. E. White, paper presented at the Electrochemical Society meeting, Honolulu, HI, October 1987.
Y. Ogata, T. Kojima, S. Uchiyama, M. Yasuda and F. Hine,J. Electrochem. Soc. 136 (1989) 91.
Z. Twardowski, H. L. Yeager and B. O'Dell,J. Electrochem. Soc. 129 (1982) 328.
D. J. G. Ives, ‘Reference Electrodes’, (edited by D. J. G. Ives and G. J. Janz), Academic Press, New York (1961) Chapter 7.
R. S. Yeo and H. L. Yeager, ‘Modern Aspects of Electrochemistry’, Vol. 16, (edited by B. E. Conway, R. E. White and J. O'M. Bockris), Plenum Press, New York (1985) Chapter 6.
W. G. F. Grot, G. E. Munn and P. N. Walmsley, paper presented at the Electrochemical Society Meeting, Houston, Texas, May 1972.
R. R. Chandran, R. S. Yeo and D-T. Chin,Electrochim. Acta 30 (1985) 158.
Information supplied by Asahi Glass Co.
E. Heitz and G. Kreysa, ‘Principles of Electrochemical Engineering’, VCH, Weinheim (1986) p. 197.
S. Rodinini and M. Fettari, ‘Proceedings of the Symposium on Diaphragms, Separators, and Ion-Exchange Membranes’, Electrochem. Soc., Pennington, NJ (1986) p. 120.
Author information
Authors and Affiliations
Additional information
Deceased.
Rights and permissions
About this article
Cite this article
Ogata, Y., Uchiyama, S., Hayashi, M. et al. Studies of the pH of the membrane surface in a laboratory chlor-alkali cell. J Appl Electrochem 20, 555–558 (1990). https://doi.org/10.1007/BF01008863
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01008863