Abstract
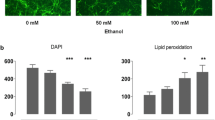
We used two experimental paradigms to examine the influence of the neurotrophins, NGF, EGF, and bFGF on normal neuroblast survival and also after ethanol insult. In the first paradigm, chick embryos received in ovo at embryonic day 1 and 2 (E1 and E2) saline (control) ethanol (10mg/50 μl/day), NGF (50 ng/50 μl/day), or EGF (25 ng/50 μl/day), or ethanol+NGF or EGF. At E3, cultures were prepared from whole embryos separately from each group. At C2, all cultures were labeled with [3H]thymidine and assessed for effects or neuronal survival. In the second paradigm, cultures were prepared from 3-day-old whole embryos and at CO, cultures were treated with either ethanol (50 mM) alone, NGF (50 ng/ml) alone, EGF (25 ng/ml) alone, bFGF (50 ng/ml) alone, or were treated concomitantly with ethanol plus one of the neurotrophins; control had only the culture medium, DMEM+5% FBS. We obtained the following findings. 1) Cultures derived from embryos treated with either of the three neurotrophins exhibited a higher neuronal survival as compared to controls (1st paradigm). 2) The survival-promoting effect was also observed when the neurotrophins were added directly to the cultures (2nd paradigm). 3) As reported previously, cultures derived from ethanol-treated embryos exhibited a marked decline in neuronal survival as compared to controls. 4) All three neurotrophins attenuated the decline in neuronal survival produced by ethanol. The ‘rescuing’ effects of the neurotrophins support our early hypothesis that ethanol administration during early neurogenesis interferes with microenvironmental trophic signals essential for neuroblast survival and differentiation.
Similar content being viewed by others
References
Arimatsu, Y., and Mayamoto, M. 1991. Survival-promoting effect of NGF on in vitro septohippocampal neurons with cholinergic and GABAergic phenotypes. Devel. Brain Res. 58:189–201.
Bannerman, P. G., and Pleasure, D. 1993. Protein growth factor requirements of rat neural crest cells. J. Neurosci Res. 36:46–57.
Barakat, I., Sensenbrenner, M., and Labourdette, G. 1982. Stimulation of chick neuroblast proliferation in culture by brain extracts. J. Neurosci Res. 8:303–314.
Bennett, G. S., and DiLullo, C. 1985. Expression of a neurofilament protein by the precursors of a subpopulation of ventral spinal cord neurons. Devel. Biol. 107:94–106.
Bennett, G. S., and DiLullo, C. 1985. Transient expression of a neurofilament protein by replicating neuroepithelial cells of the embryonic chick brain. Devel. Biol. 107:107–127.
Brodie, C., Kentroti, S., and Vernadakis, A. 1991. Growth factors attenuate the cholinotoxic effects of ethanol during early neuroembryogenesis in the chick embryo. Intl. J. Neurosci 9:203–213.
Brodie, C., and Vernadakis, A. 1990. Critical periods to ethanol exposure during early neuroembryogenesis in the chick embryo: Cholinergic neurons. Devel. Brain Res. 56:223–228.
Calamandrei, G., and Alleva, E. 1989. Epidermal growth factor has both growth-promoting and growth-inhibiting effects on physical and neurobehavioral development of neonatal mice. Brain Res. 477:1–6.
Carmignoto, G., Maffei, L., Candeo, P., Canella, R., and Comelli, C. 1989. Effect of NGF on the survival of rat retinal ganglion cells following optic nerve section. J. Neurosci. 9:1263–1272.
Carpenter, G., and Cohen, S. 1979. Epidermal growth factor. Ann Rev. Biochem. 48:193–216.
Cattaneo, E., and McKay, R. 1990. Proliferation and differentiation of neuronal stem cells regulated by nerve growth factor. Nature 347:762–765.
Ernfors, P., Hallböök, Ebendl, T., Shooter, E. M., Radeke, M. J., Misko, T. P., and Persson, H. 1983. Developmental and regional expression ofB-nerve growth factor receptor mRNA in the chick and rat. Neuron 1:983–996.
Fischer, W., Wictorin, K., Björklund, A., Williams, L. R., Varon, S., and Gage, F. H. 1987. Amelioration of cholinergic neuron atrophy and spatial memory impairment in aged rats by nerve growth factor. Nature 329:65–68.
Gage, F. H., Batchelor, P., Chen, K. S., Chin, D., Higgins, G. A., Koh, S., Deputy, S., Rosenberg, M. B., Fischer, W., and Björklund, A. 1989. NGF receptor reexpression and NGF-mediated cholinergic neuronal hypertrophy in the damaged adult neostriatum. Neuron 2:1177–1184.
Hamburger, V. 1992. History of the discovery of neuronal death in embryos. J. Neurobiol. 23:1116–1123.
Hatanaka, H., Nihonmatsu, I., and Tsukui, H. 1988. Nerve growth factor promotes survival of cultured magnocellular cholinergic neurons from nucleus basalis of Meynert in postnatal rats. Neursci. Ltrs. 90:63–68.
Hatanaka, H., Nihonmatsu, I., and Tsukui, H. 1988. Developmental change in the nerve growth factor action from induction of choline acetyltransferase to promotion of cell survival in cultured basal forbrain cholinergic neurons from postnatal rats. Devel. Brain Res. 39:85–95.
Heuer, J. G., Von Barthheld, C. S., Kinoshita, Y., and Evers, P. C. 1990. Alternating phases of FGF reception and NFG receptor expression in the developing chicken nervous system. Neuron 5: 283–296.
Johnson, E. M., and Deckwerth, T. L. 1993. Molecular mechanisms of developmental neuronal death. Annu. Rev. Neurosci. 16: 31–46.
Johnson, E. M. Jr., Chang, J. R. S., Koikes, T., and Martin, D. P. 1989. Why do neurons die when deprived of trophic factor? Neurobiology of Aging 10:549–552.
Kalcheim, C., and Neufeld, G. 1990. Expression of basic fibroblast growth factor in the nervous system of early avian embryos. Development 109:203–215.
Kentroti, S., and Vernadakis, A. 1990. Neuronal plasticity in the developing chick brain: Interaction of ethanol and neuropeptides. Devel. Brain Res. 56:205–210.
Kentroti, S., and Vernadakis, A. 1991. Survival and proliferation in developing neuroblasts in cultures derived from embryos treated with ethanol during early neuroembryogenesis: Effects attenuated by somatostatin. J. Neurosci. Res. 30:618–641.
Kentroti, S., and Vernadakis, A. 1991. Correlation between morphological and biochemical effects of ethanol on neuroblast-enriched cultures derived from 3-day-old chick embryos. J. Neurosci. Res. 30:484–492.
Knowles, A. F. 1988. Inhibition of growth and induction of enzyme activities in a clonal human hepatoma cell line (Li-7A): Comparison of the effects of epidermal growth factor and an anti-epidermal growth factor receptor antibody. J. Cell Physiol. 134: 109–116.
Koliatsos, V. E., Clatterbuck, R. E., Nauta, H. J. W., Knüsel, B., Burton, L. E., Mobley, W. C., and Price, D. L. 1991. Human nerve growth factor prevents degeneration of basal forebrain cholinergic neurons in primates. Ann Neurol. 30:831–840.
Levi-Montalcini, R. 1987. The nerve growth factor 35 years later. Science 237:1154–1162.
Mangoura, D., Sakellaridis, N., and Vernadakis, A. 1988. Factor influencing neuronal growth in primary cultures derived from 3-day-old chick embryos. Int. J. Devel. Neurosci. 6:89–102.
Mangoura, D., Sakellaridis, N., and Vernadakis, A. 1988. Cholinergic neurons in cultures derived from three, six, or eight-day-old chick embryos. A biochemical and immunocytochemical study. Devel. Brain Res. 40:37–46.
Mangoura, D., and Vernadakis, A. 1988. Gabaergic neurons in cultures derived from three, six, or eight-day-old chick embryos. A biochemical and immunocytochemical study. Devel. Brain Res. 40:25–35.
Mayer, E., Dunnett, S. B., Pellitteri, R., and Fawcett, J. W. 1993. Basic fibroblast growth factor promotes the survival of embryonic ventral mesencephalic dopaminergic neurons—I. Effects in vitro. Neurosci. 56:379–388.
Mayer, E., Dunnett, S. B., Pellitteri, R., and Fawcett, J. W. 1993. Basic fibroblast growth factor promotes the survival of embryonic ventral mesencephalic dopaminergic neurons—II. Effects on nigral transplants in vitro. Neurosci. 56:389–398.
Mayer, E., Dunnett, S. B., and Fawcett, J. W. 1993. Basic FGF promotes the survival of embryonic ventral mesencephalic dopamine neurons. I. Effects in vitro. Neurscience 56:374–388.
Monnet-Tschudi, F., Honegger, P. 1989. Influence of epidermal growth factor on the maturation of fetal rat brain cells in aggregate culture. Devel. Neurosc. 11:30–40.
Morgan, D. G. 1989. Considerations in the treatment of neurological disorders with trophic factors. Neurobiol. Aging 10:547–549.
Morrison, R. S., Kornblum, H. I., Leslie, F. M., and Bradshaw, R. A. Trophic stimulation of cultured neurons from neonatal rat brain by epidermal growth factor. Science 238:72–75.
Mytilineou, C., Park, J. H., and Shen, J. 1992. Epidermal growth factor-induced survival and proliferation of neuronal precursor cells from embryonic rat mesencephalon. Neurosci. Ltr. 135:62–66.
Oppenheim, R. W. 1991. Cell death during development of the nervous system. Annu. Rev. Neurosci. 14:453–501.
Oppenheim, R. W., Prevette, D., and Fuller, F. 1992. The lack of effect of basic and acid fibroblast growth factors on the naturally occurring death of neurons in the chick embryo. J. Neurosci. 12: 2726–2734.
Raff, M. C., Barres, B. A., Burne, J. F., Coles, H. S., Ishizaki, Y., and Jacobson, M. D. 1993. Programmed cell death and the control of cell survival: Lessons from the nervous system. Science 262: 695–700.
Rahman, H., Kentroti, S., and Vernadakis, A. 1994. The critical period for ethanol effects on cholinergic neuronal expression in neuroblast-enriched cultures derived from 3-day-old chick embryo: NGF attenuation of the cholinergic effect of ethanol. Intl. J. Devel. Neurosci. 12:397–404.
Schlessinger, J., Schriber, A. B., Levi, A., Lex, I., Libermann, T., and Yarden, Y. 1983. Regulation of cell proliferation by epidermal growth factor. CRC. Crit. Rev. Biochem. 4:93–111.
Smith, C. J., Wion, D., and Brachet, P. 1991. Nerver growth factor-induced neuronal differentiation is accompanied by differential splicing ofB-amyloid precursor mRNAs in the PC12 cell line. Mole Brain Res. 10:351–354.
Snider, W. W., and Johnson, E. M. Jr. 1989. Neurotrophic molecules. Ann Neurol 26:489–506.
Tanaka, H., and Landmeser, L. T. 1986. Cell death of lumbosacral motoneurons in chick, quail, and chick-quail chimeral embryos: A test of the quantative matching hypothesis of neuronal cell death. J. Neurosci. 6:2889–2899.
Thoenen, H., and Barde, Y-A. 1980. Physiology of nerve growth factor. Physiol. Rev. 60:1284–1335.
Tiffany-Castiglioni, E., and Perez-Polo, J. R. 1979. The role of nerve growth factor in vitro in cell resistance to 6-hydroxydopamine toxicity.
Tishler, A. S., Riseberg, J. C., Hardenbrook, M. A., and Cherington, V. 1993. Nerve growth factor is a potent inducer of proliferation and neuronal differentiation for adult rat chromaffin cells in vitro. J. Neurosci. 13:1533–1542.
Torelli, S., Dell-Era, P., Ennas, M-G., Soyos, V., Gremo, F., Ragnott, G., and Presto, M. 1990. Basic fibroblast growth factor in neuronal cultures of human fetal brain. J. Neurosci. Res. 27:78–83.
Tuszynski, M. H., U, H. S., Amaral, D. G., and Gage, F. H. 1990. Nerve growth factor infusion in the primate brain reduces lesion-induced cholinergic neuronal degeneration. J. Neurosci. 10:3604–3614.
Walicke, P., Cowan, W. M., Ueno, N., Baird, A., and Guillenmin, R. Fibroblast growth factor promotes survival of dissociated hippocampal neurons and enhances neurite extension. Proc. Natl. Acad. Sci. USA. 83:3012–3016.
Weise, B., Janet, T., and Grothe, C. 1993. Localization ofBFGF and FGF-receptor in the developing nervous system of the embryonic and newborn rat. J. Neurosci. Res. 34:442–453.
Verndakis, A., Sakellaridis, N., and Mangoura, D. 1986. Growth patterns of primary cultures dissociated from 3-day-old chick embryos: Morphological and biochemical comparisons. J. Neurosci. Res. 16:397–407.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rhaman, H., Kentroti, S. & Vernadakis, A. Neuroblast cell death in ovo and in culture: Interaction of ethanol and neurotrophic factors. Neurochem Res 19, 1495–1502 (1994). https://doi.org/10.1007/BF00968996
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00968996