Summary
In contrast to chemically induced mutants of tobacco mosaic virus (TMV) in which we have found replacement of one or at most of two amino acids per coat protein chain, the protein chains of naturally occurring TMV strains differ from each other in numerous positions. The complete amino acid sequence of the naturally occurring TMV straindahlemense isolated byMelchers (1940) has been determined. It differs in 30 of the 158 amino acid positions from the TMV wild strainvulgare (Fig. 1). This is the first case in which complete amino acid sequences of the coat proteins of two virus strains can be compared. Such a comparison permits conclusions about the structure of the protein subunits and about certain aspects of the genetic code to be drawn.
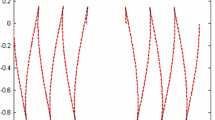
The electrophoretic mobility curves for the virus rods and the A proteins ofvulgare anddahlemense (Fig. 4) can be explained on the basis of the amino acid sequences of the two strains. Spatial distribution of the positive and negative groups within the protein subunits are discussed. One particular segment of the protein chain appears to be so important for the secondary and/or tertiary structure of the protein subunit that amino acid replacements within this segment in general lead to a loss of infectivity.
The 46 cases in which we have exactly located the positions of amino acid differences betweenvulgare and various TMV mutants and strains are summarized in Table 1. Combination of the data in Table 1 with the base compositions of the triplets as obtained from the cell free system ofE. coli permits conclusions about the nucleotide sequence within the triplets to be drawn. The triplets shown in Table 2 represent, at present, the best agreement between the data from the cell free system and the work with TMV mutants.
Similar content being viewed by others
Literatur
Anderer, F. A.: Die Reihenfolge der Aminosäuren im Protein des Tabakmosaikvirus. Z. Naturforsch.17b, 526–543 (1962).
Anderer, F. A., H. Uhlig, E. Weber andG. Schramm: Primary structure of the protein of tobacco mosaic virus. Nature (Lond.)186, 922–925 (1960).
Benzer, S., andS. W. Champe: A change from nonsense to sense in the genetic code. Proc. nat. Acad. Sci. (Wash.)48, 1114–1121 (1962).
Crick, F. H. C., L. Barnett, S. Brenner andR. J. Watts-Tobin: General nature of the genetic code for proteins. Nature (Lond.)192, 1227–1232 (1961).
Ehrenstein, G. v., andF. Gonano: Degenerate transfer RNAs as amino adaptors in protein synthesis. Cold Spr. Harb. Symp. quant. Biol.28 (im Druck).
Hanada, M., andD. L. Rucknagel: The abnormality of the primary structure of haemoglobin Shimonoseki. Biochem. biophys. Res. Commun.11, 229–234 (1963).
Jones, O. W., andM. W. Nirenberg: Qualitative survey of RNA codewords. Proc. nat. Acad. Sci. (Wash.)48, 2115–2123 (1962).
Jukes, T. H.: Observations on the possible nature of the genetic code. Biochem. biophys. Res. Commun.10, 155–159 (1963a).
—: The genetic code. Amer. Sci.51, 227–245 (1963b).
Knight, C. A.: Chemistry of viruses. In: Protoplasmatologia, vol. IV, 177 p. Wien: Springer 1963.
—,D. M. Silva, D. Dahl andA. Tsugita: Two distinctive strains of tobacco mosaic virus. Virology16, 236–243 (1962).
Kramer, E., u.H. G. Wittmann: Elektrophoretische Untersuchungen der A-Proteine dreier Tabakmosaikvirusstämme. Z. Naturforsch.13b, 30–33 (1958).
Melchers, G.: Die biologische Untersuchung des “Tomatenvirus Dahlem 1940”. Biol. Zbl.60, 527–537 (1940).
Nirenberg, M. W., andJ. H. Matthaei: The dependence of cell free protein synthesis inE. coli upon naturally occurring or synthetic poly-ribonucleotides. Proc. nat. Acad. Sci. (Wash.)47, 1588–1602 (1961).
Schramm, G.: Die Reindarstellung und die chemischen Eigenschaften des “Tomatenmosaikvirus Dahlem 1940”. Biol. Zbl.60, 538–546 (1940).
Sueoka, N., andT. Yamane: Fractionation on amino acyl acceptor RNA on a methylated albumin column. Proc. nat. Acad. Sci. (Wash.)48, 1454–1461 (1962).
Tsugita, A.: The proteins of mutants of TMV. J. molec. Biol.5, 284–300 (1962).
—, andH. Fraenkel-Conrat: The amino acid composition and C-terminal sequence of a chemically evoked mutant of TMV. Proc. nat. Acad. Sci. (Wash.)46, 636–642 (1960).
——: The composition of proteins of chemically evoked mutants of TMV-RNA. J. molec. Biol.4, 73–82 (1962).
—D. T. Gish, J. Young, H. Fraenkel-Conrat, C. A. Knight andW. M. Stanley: The complete amino acid sequence of the protein of tobacco mosaic virus. Proc. nat. Acad. Sci. (Wash.)46, 1436–1469 (1960).
Wahba, A. J., C. Basilio, J. F. Speyer, P. Lengyel, R. S. Miller andS. Ochoa: Synthetic polynucleotides and the amino acid code. V. Proc. nat. Acad. Sci. (Wash.)48, 1683–1686 (1962).
—R. S. Miller, C. Basilio, R. S. Gardner, P. Lengyel andJ. F. Speyer: Synthetic polynucleotides and the amino acid code. IX. Proc. nat. Acad. Sci. (Wash.)49, 880–885 (1963).
Weisblum, B., S. Benzer andR. W. Holley: A physical basis for the degeneracy in the amino acid code. Proc. nat. Acad. Sci. (Wash.)48, 1449–1454 (1962).
Wittmann, H. G.: Vergleich der Proteine des Normalstamms und einer Nitritmutante des TMV. Z. Vererb.-Lehre90, 463–475 (1959).
Wittmann, H. G.: Studies on the nucleic acid-protein correlation in tobacco mosaic virus. Proc. V. Internat. Congr. Biochem., Moscow1961, vol. I, pp. 240–254.
—: Proteinuntersuchungen an Mutanten des Tabakmosaikvirus als Beitrag zum Problem des genetischen Codes. Z. Vererb.-Lehre93, 491–530 (1962).
Wittmann, H. G., andB. Wittmann-Liebold: Tobacco mosaic virus mutants and the genetic coding problem. Cold Spr. Harb. Symp. quant. Biol.28 (im Druck).
Wittmann-Liebold, B., andH. G. Wittmann: Die primäre Proteinstruktur von Stämmen des Tabakmosaikvirus: Teil I. Hoppe-Seylers Z. physiol. Chem. (im Druck) (1963a).
Wittmann-Liebold, B., andH. G. Wittmann: Die primäre Proteinstruktur von Stämmen des Tabakmosaikvirus: Teil II. Z. Naturforsch. (im Druck) (1963b).
Author information
Authors and Affiliations
Additional information
Mit 4 Textabbildungen
Rights and permissions
About this article
Cite this article
Wittmann-Liebold, B., Wittmann, H.G. Die primäre Proteinstruktur von Stämmen des Tabakmosaikvirus. Zeitschrift für Vererbungslehre 94, 427–435 (1963). https://doi.org/10.1007/BF00897598
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00897598