Abstract
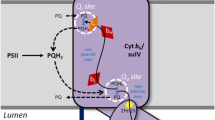
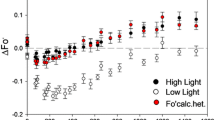
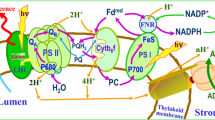
Flash-driven ATP formation by spinach chloroplast thylakoids, using the luciferin luminescence assay to detect ATP formed in single turnover flashes, was studied under conditions where a membrane protein amine buffering pool was either protonated or deprotonated before the beginning of the flash trains. The flash number for the onset of ATP formation was delayed by about 10 flashes (from 15 to about 25) when the amine pool was deprotonated as compared to the protonated state. The delay was substantially reversed again by reprotonating the pool upon application of 20–30 single-turnover flashes and 8 min of dark before addition of ADP, Pi, and the luciferin system. In the case of deprotonation by desaspidin, the uncoupler was removed by binding to BSA before the reprotonating flashes were given. Reprotonation was carried out before addition of ADP and Pi, to avoid a possible interference by the ATP-ase, which can energize the system by pumping protons. The reprotonated state, as indicated by an onset lag of about 15 flashes rather than 25 for the deprotonated state, was stable in the dark over extended dark times. The number of protons released by 10 flashes is approximately 30 nmol H+ (mg chl)−1, an amount similar to the size of the reversibly protonated amine group buffering pool. The data are consistent with the hypothesis that the amine buffering groups must be in the protonated state before any protons proceed to the coupling complex and energize ATP formation. Other work has suggested that the amine buffering pool is sequestered within membrane proteins rather than being exposed directly to the inner aqueous bulk phase. Therefore, it is possible that the sequested amine group array may provide localized association-dissociation sites for proton movement to the coupling complex.
Similar content being viewed by others
References
Abbott, M. S., and Dilley, R. A. (1983).Arch. Biochem. Biophys. 222 95–104.
Arnon, D. I. (1949).Plant Physiol. 24 1–15.
Baker, G. M. (1983). Ph.D. Thesis, Purdue University.
Baker, G. M., Bhatnagar, D., and Dilley, R. A. (1982).J. Bioenerg. Biomembr. 14 249–264.
Baker, G. M., Bhatnagar, D., and Dilley, R. A. (1981).Biochemistry 20 2307–2315.
Boyer, P. D., Chance, B., Ernster, L., Mitchell, P., Racker, E. and Slater, E. (1977).Ann. Rev. Biochem. 46 955–1026.
Davenport, J. W., and McCarty, R. E. (1980).Biochim. Biophys. Acta 589 353–357.
Davenport, J. W., and McCarty, R. E. (1981).J. Biol. Chem. 256 8947–8954.
Dilley, R. A., Prochaska, L. J., Baker, G. M., Tandy, N. E., and Millner, P. A. (1982).Curr. Top. Membr. Transp. 16 345–369.
Dunker, A. K., and Marvin, D. A. (1978).J. Theor. Biol. 22 9–16.
Galmiche, J. M., and Girault, G. (1982).FEBS Lett. 146 123–127.
Giaquinta, R. T., Ort, D. R., and Dilley, R. A. (1975).Biochemistry 14 4392–4396.
Giaquinta, R. T., Dilley, R. A., Anderson, B. J., and Horton, P. (1974).Bioenergetics 6 167–177.
Graan, T., and Ort, D. (1981).Biochim. Biophys. Acta 637 447–456.
Graan, T., and Ort, D. R. (1982).Biochim. Biophys. Acta 682 395–403.
Graan, T., and Ort, D. R. (1983).J. Biol. Chem. 258 2831–2836.
Graan, T., Flores, S., and Ort, D. R. (1981). InEnergy Coupling in Photosynthesis (Selman, B., and Selman-Reiner, S., eds.), Elsevier North-Holland, pp. 25–34.
Gräber, P. (1982).Curr. Top. Membr. Transp. 16 215–248.
Hope, A. B., Ranson, D., and Dixon, P. G. (1982).Aust. J. Plant Physiol. 9 399–407.
Johnson, J. D., Pfister, V. R., and Homann, P. H. (1983).Biochim. Biophys. Acta 723 256–265.
Karlish, S. J. D., Shavit, N., and Auron, M. (1969).Eur. J. Biochem. 9 291–298.
Karu, A. E., and Moudrianakis, E. N. (1969).Arch. Biochem. Biophys. 129 655–671.
Laszlo, J. A., Baker, G. M., and Dilley, R. A. (1984a).J. Bioenerg. Biomembr.,16 37–51.
Laszlo, J. A., Baker, G. M., and Dilley, R. A. (1984b).Biochim. Biophys. Acta 764 160–169.
Mitchell, P. (1966).Biol. Rev. Cambridge Philos. Soc. 41 445–502.
Moudrianakis, E. N., and Tiefert, M. A. (1976).J. Biol. Chem. 251 7796–7801.
Nagle, J. F., and Morowitz, H. J. (1978).Proc. Natl. Acad. Sci. U.S.A. 75 298–302.
Nagle, J. F., and Tristram-Nagle, S. (1983).J. Membr. Biol. 74 1–14.
Ort, D. R., and Izawa, S. (1973).Plant Physiol. 52 595–600.
Ort, D. R., and Dilley, R. A. (1976).Biochim. Biophys. Acta 449 95–107.
Ort, D. R., Dilley, R. A., and Good, N. E. (1976).Biochim. Biophys. Acta 449 108–124.
Portis, A. R., and McCarty, R. E. (1974).J. Biol. Chem. 249 6250–6254.
Prochaska, L. J., and Dilley, R. A. (1978).Arch. Biochem. Biophys. 187 61–71.
Schreiber, U., and Del Valle-Tascon, S. (1983).FEBS Lett. 150 32–37.
Shavit, N., and Strotmann, H. (1980).Methods Enzymol. 69 321–324.
Tandy, N. E., Dilley, R. A., Hermodson, M. A., and Bhatnagar, D. (1982).J. Biol. Chem. 257 4301–4307.
Theg, S. M., and Junge, W. (1983).Biochim. Biophys. Acta 723 294–307.
Theg, S. M., Johnson, J. D., and Homann, P. H. (1982).FEBS Lett. 145 25–29.
Vinkler, C., Avron, M., and Boyer, P. D. (1980).J. Biol. Chem. 255 2263–2266.
Williams, R. J. P. (1962).J. Theor. Biol. 3 209–229.
Yamamoto, Y., Shimada, S., and Nishimura, M. (1983).FEBS Lett. 151 49–53.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dilley, R.A., Schreiber, U. Correlation between membrane-localized protons and flash-driven ATP formation in chloroplast thylakoids. J Bioenerg Biomembr 16, 173–193 (1984). https://doi.org/10.1007/BF00751048
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00751048