Abstract
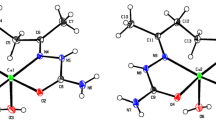
Powder samples of the new compound [Cu(C5H5N)4·Br2](C5H5N) and of its desorption product have been studied by EXAFS. The crystal structure of [Cu(C5H5N)4Br2]·(C5H5N) has been determined from three-dimensional X-ray diffraction data and refined to theR value 0.038 for 379 observed reflections. (orthorhombic,Ccca,a=12.033(8),b=14.764(3),c=16.768 Å,V=1897(3) Å3,Z=4). The copper atom lies on a 222 symmetry site and is hexacoordinated with four nitrogen and two bromine atoms, forming an elongated octahedron with the bromine atoms in apical positions (Cu−Br distance: 3.201(2) Å, Cu−N1/N2 distances: 2.02(1)/2,07(1) Å). The host molecules (Cu(C5H5N)4Br2) form layers parallel to the (001) plane. Cavities in the space between these layers, bound by pyridine ligands protruding into this space, are occupied by the guest molecules (C5H5N). The guest desorption at room temperature is accompanied by a chemical and structural destruction of the host, leading to the known compound Cu(C5H5N)2Br2.
Similar content being viewed by others
References
J. Lipkowski: inInclusion Compounds, Vol. 1 (Eds. J.L. Atwood, J.E.D. Davies and D.D. MacNicol), pp. 59–103, Academic Press, London, 1984.
N.V. Pervukhina, N.V. Podberezskaya, V.V.Bakakin, N.V. Kislykh, G.N. Chekhova and Yu.A. Dyadin:Zh. Strukt. Khim. 26, 120 (1985);Chem. Abstr. 104, 139681g (1986).
N.V. Pervukhina, N.V. Podberezskaya, I.V. Davydova, N.V. Kislykh and Yu.A. Dyadin:J. Incl. Phenom. 13, 9 (1992).
G.J. Long and P.J. Clarke:Inorg. Chem. 17, 1394 (1978) (and references therein).
H. Hartl and T. Brudgam:Acta Crystallogr. B36, 162 (1980).
H. Hartl and S. Steidl:Z. Naturforsch. 32B, 6 (1977).
M.A. Poraj-Koshits and A.S. Antsychkina:Kristallografiya 3, 686 (1958).
H. Hartl and S. Steidl:Acta Crystallogr. B36, 65 (1980).
A.C.T. North, D.C. Phillips and F.S. Mathews:Acta Crystallogr. A24, 351 (1968).
N. Walker and D. Stuart:Acta Crystallogr. A39, 158 (1983).
International Tables for X-Ray Crystallography, Vol. IV, The Kynoch Press, Birmingham, England (1974), Distr. Kluwer Academic Publishers, Dordrecht, The Netherlands.
B. Morosin:Acta Crystallogr. B31, 632 (1975).
V. Kupčik and S. Ďurovič:Czech. J. Phys. 10, 182 (1960).
D. Bonnin, P. Kaiser and J. Desbarres: EXAFS, Ecole d'été à Garchy, France, 19–24 September 1988.
B.A. Frenz:Enraf-Nonius, Structure Determination Package, V3.0, Enraf-Nonius, Delft, The Netherlands (1985).
Tripos:Molecular Modeling Software, V 5.2, Inc. Missouri, USA (1989).
C.K. Johnson:ORTEPII, Report ORNL. 5138 (Third Revision), Oak Ridge National Laboratory, Tennessee, USA (1976).
A. Sakthivel and R.A. Young: School of Physics, Georgia Institute of Technology, Atlanta, USA, GA30332 (1992).
D.E. Sayers, A.E. Stern and F.W. Lytle:Phys. Rev. Lett. 27, 1204 (1971).
A.G. McKale, B.W. Veal, A.P. Paulikas, S.K. Chan and G.S. Knappe:J. Am. Chem. Soc. 110, 3763 (1988).
J.D. Dunitz:Acta Crystallogr. 10, 307 (1957).
Author information
Authors and Affiliations
Additional information
Supplementary Data relevant to this article (lists of observed and calculated structure factors, calculated positional parameters of H atoms, root-mean-square amplitudes of thermal vibration and anisotropic thermal parameters) have been deposited with the British Library as Supplementary Publication SUP 82163 (5 pages)
Rights and permissions
About this article
Cite this article
Selkti, M., Ling, Cc. & Navaza, A. Crystal structure of the clathrate compound [dibromotetra(pyridine)copper(II)]·pyridine. Characterization by EXAFS of the pyridine desorption product. J Incl Phenom Macrocycl Chem 17, 127–135 (1994). https://doi.org/10.1007/BF00711853
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00711853