Abstract
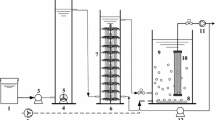
Effluent from the manufacture of acrylonitrile is difficult to biodegrade. It contains nine major organic components: acetic acid, acrylonitrile, acrylamide, acrylic acid, acrolein, cyanopyridine, fumaronitrile, succinonitrile, and maleimide. A range of bacteria have been isolated that can grow on, or convert all of the organic components of effluent from the manufacture of acrylonitrile. These bacteria can be used as the basis of a mixed culture system to treat the effluent. The bacteria were utilised in batch and continuous cultures to degrade a synthetic wastewater containing acrylonitrile, acrylamide, acrylic acid, cyanopyridine and succinonitrile. The mixed microbial population was adapted by varying the growth rate and switching from continuous to batch and back to continuous growth, to degrade these five compounds as well as acrolein, fumaronitrile and maleimide.
Similar content being viewed by others
Abbreviations
- BOD:
-
Biological Oxygen Demand
- COD:
-
Chemical Oxygen Demand
- T D :
-
Doubling Time
- ppm:
-
parts per million
- HPLC:
-
High Pressure Liquid Chromatography
- GLC:
-
Gas Liquid Chromatography
References
Arnaud A, Galzy P & Jallageas JC (1976a) Amidase activity of some bacteria. Folia Microbiologica 21: 178–184
Arnaud A, Galzy P & Jallageas JC (1976b) Etude de l'activité nitrilasique de quelques bacteries. Revue des Fermentations et des Industries Alimentaires 31:39–44 Arnaud A, Galzy P & Jallageas JC (1977) Étude de l'acetonitrilase d'une souche deBrevibacterium. Agricultural and Biological Chemistry 41: 2183–2191
Asano Y, Tani T & Yamada H (1980) A new enzyme ‘nitrile hydratase’ which degrades acetonitrile in combination with amidase. Agricultural and Biological Chemistry 44: 2251–2252
Bauchop T & Elsden SR (1960) The growth of microorganisms in relation to their energy supply. Journal of General Microbiology 23: 457–469
Cain RB (1984) Xenobiotic breakdown by mixed cultures. Biochemical Society Transactions 12: 1146–1148
Catchpole JR & Stafford DA (1977) The biological treatment of coke-oven liquors. In: Callely AG, Forster CF & Stafford DA (Eds) Treatment of Industrial Effluents (pp 258–272) D.A. Hodder & Stoughton, UK
Collins PA & Knowles CJ (1983) The utilization of nitriles and amides byNocardia rhodochrous. Journal of General Microbiology 129: 711–718
Commeyras A, Arnaud A, Galzy P & Jallageas JC (1977) Process for the preparation of amides by biological hydrolysis. United States Patent 4,001,081
Cook AM, Grossenbacher H & Hutter R (1983) Isolation and cultivation of microbes with biodegradative potential. Experienta 39: 1191–1198
Cowan ST & Steel KJ (1965) Manual for the identification of medical bacteria. Cambridge University Press
Dalton H & Stirling DI (1982) Co-metabolism. Philosophical Transactions of the Royal Society of London, Series B 297: 481–496
DiGeronimo MJ & Antoine AD (1976) Metabolism of acetonitrile and propionitrile byNocardia rhodochrous LL100-21. Applied and Environmental Microbiology 31: 900–906
Fawcett JK & Scott JE (1960) A rapid and precise method for the determination of urea. Journal of Clinical Pathology 13: 156–159
Geyer BP (1962) Reaction with water. In: Smith CW (Ed) Acrolein (pp 144–153)
Gresseli JG (Ed) (1973) Atlas of spectral data and physical constants for organic compounds. C.R.C. Press
Hall ER & Melcer H (1983) Biotechnology developments for the treatment of toxic and inhibitory wastewaters. Biotechnology Advances 1: 59–71
Harder W, Kuenen JG & Matin A (1977) Microbial selection in continuous culture. Journal of Applied Bacteriology 43: 1–24
Harper DB (1976) Purification and properties of an unusual nitrilase fromNocardia NCIB 11216. Biochemical Society Transactions 4: 502–504
Harper DB (1977a) Microbial metabolism of aromatic nitriles. Enzymology of C-N cleavage byNocardia sp. (Rhodochrous group) NCIB 11216. Biochemical Journal 165: 309–319
Harper DB (1977b) Fungal degradation of aromatic nitriles. Enzymology of C-N cleavage byFusarium solani. Biochemical Journal 167: 685–692
Harper DB (1985) Characterisation of a nitrilase fromNocardia sp. (Rhodochrous group) NCIB 11215 using p-hydroxybenzonitrile as sole carbon source. International Journal of Biochemistry 17: 677–683
Hook RH & Robinson WG (1964) Ricinine nitrilase II purification and properties. Journal of Biological Chemistry 239: 4263–427
Jallageas JC, Arnaud A & Galzy P (1978) Etude l'acetamidase d'une souche deBrevibacterium. Journal of General and Applied Microbiology 24: 103–114
Krieg NR (1981) Enrichment and isolation. In: Manual of Methods for General Bacteriology (pp 112–142) American Society for Microbiology
Kuwahara M, Yanase H, Kikuchi Y & Okazumi K (1980a) Metabolism of succinonitrile inAeromonas sp. Hako Kogaku Kaishi 58: 441–447
Kuwahara M, Yanase H, Ishida Y & Kikuchi Y (1980b) Metabolism of aliphatic nitriles inFusarium solani. Journal of Fermentation Technology 58: 573–577
Lande SS, Bosch SJ & Howard PH (1979) Degradation and leaching of acrylamide in soil. Journal of Environmental Quality 8: 133–137
Linton EA & Knowles CJ (1986) Utilization of aliphatic amides and nitriles byNocardia rhodochrous LL100-21. Journal of General Microbiology 132: 1493–1401
Miller JH (1972) Experiments in Molecular Genetics (p 431) Cold Spring Harbor Laboraotry, Cold Spring Harbor, N.Y.
Miller JM & Knowles CJ (1984) The cellular location of nitrilase and amidase enzymes ofBrevibacterium R312. FEMS Letters 21: 147–151
Mimura A, Kawano T & Yamaga K (1969) Application of microorganisms to the Petrochemical Industry. 1. Assimilation of nitrile compounds by microorganisms. Journal of Fermentation Technology 47: 631–638
Nagasawa T & Yamada H (1989) Microbial transformations of nitriles. Trends in Biotechnology 7: 153–158
Palleroni NJ (1980) Isolation and properties of a new hydrogen bacterium related toPseudomonas saccharophila. Journal of General Microbiology 117: 155–161
Robinson WG & Hook RH (1964) Ricinine nitrilase. Reaction product and substrate specificity. Journal of Biological Chemistry 239: 4257–4262
Rohm & Haas Co. (1967) Fumaronitrile as a bactericide. U.S. Patent 1,489,570
Shukla OP (1984) Microbial transformations of pyridine derivatives. Journal Scient. Ind. Res. 43: 98–116
Slater JH & Bull AT (1982) Environmental microbiology: Biodegradation. Philosophical Transactions of the Royal Society London Series B, 297: 575–597
Sinskey AJ, Akedo M & Cooney CL (1981) Acrylate fermentations, Basic Life Science 18 (Trends Biol. Ferment. Fuels Chem.) 473–492
Thijsse GJE (1964) Fatty acid accumulation by acrylate inhibition of β-=oxidation in an alkane oxidisingPesudomonas. Biochem. Biophys. Acta 84: 195–197
Thompson LA, Knowles CJ, Linton EA & Wyatt JM (1988) Microbial biotransformations of nitriles. Chemistry in Britain 900–902
Vaughan PA, Chetham PSJ & Knowles CJ (1988) The utilization of pyridine carbonitriles and carboxamides byNocardia rhodochrous LL100-21. Journal of General Microbiology 134: 1099–1107
Watanabe M, Kuginuki H, Ono T, Ohsawa T, Taki K, Isaii K & Iwaki S (1974) Antiseptic for a metal cutting fluid. Japanese patent, 74,88,907
Wittcoff HA & Rueben BG (1980) Industrial organic chemicals in perspective. 1. Raw materials and manufacture. Wiley-Interscience
Yamada H, Asano Y, Hino T & Tani Y (1979) Microbial utilization of acrylonitrile. J. Ferment. Technol. 57: 8–14
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wyatt, J.M., Knowles, C.J. The development of a novel strategy for the microbial treatment of acrylonitrile effluents. Biodegradation 6, 93–107 (1995). https://doi.org/10.1007/BF00695340
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00695340
Key words
- acrylamide
- acrylic acid
- acrolein
- acrylonitrile
- acrylonitrile effluents
- allyl alcohol
- bacterial isolates on acrylonitrile
- acrylamide
- acrylic acid
- cyanopyridine
- succinonitrile
- 3-cyanopropenoic acid
- allyl alcohol
- biotransformation of acrolein
- biotransformation of fumaronitrile
- biotransformation of of maleimide
- cyanopyridine
- 3-cyanopropenoic acid
- fumaronitrile
- maleimide
- succinonitrile