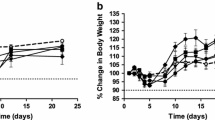
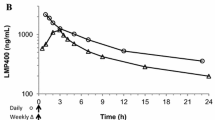
Abstract
The camptothecin derivative 7-ethyl-10-[4-(1-piperidino)-1-piperidino]-carbonyloxy camptothecin (CPT-11) has attracted the attention of clinicians because of its high antitumor activity against refractory solid cancers. We established two CPT-11-resistant cell lines, a non-small-cell lung-cancer cell line (PC-7/CPT-11) from the parental PC-7 line and an ovarian cancer cell line (HAC-2/CPT-11) from the parental HAC-2 line. The mechanisms of resistance to CPT-11 in PC-7/CPT-11 cells were reduced conversion of CPT-11 to its active metabolite SN-38 and point mutation of topoisomerase I. Those in HAC-2/CPT-11 cells were reduction of topoisomerase I activity and decreased sensitivity of topoisomerase to topoisomerase I inhibitors. No point mutation of the topoisomerase was observed in HAC-2/CPT-11 cells. We conducted two phase I trials using CPT-11 in combination with other anticancer agents. One was a phase I trial of CPT-11 and cisplatin given with a fixed dose of vindesine to patients with advanced non-small-cell lung-cancer and the other was a phase I study on a topoisomerase-targeting combination of CPT-11 and etoposide (VP-16) in patients with various malignant solid tumors. The results of the first trial indicated that the recommended dose of CPT-11 for phase II studies was 80 mg/m2 combined with 3 mg/m2 vindesine on days 1 and 8 and 60 mg/m2 cisplatin on day 1. In the second trial, the recommended dose of CPT-11/VP-16 given with recombinant granulocyte colony-stimulating factor (on days 4–17) was found to be 60/60 mg/m2 In both trials, diarrhea and granulocytopenia were considered to be dose-limiting toxicities.
Similar content being viewed by others
References
Andoh T, Ishii K, Suzuki Y, Ikegami Y, Kusunoki Y, Takemoto Y, Okada K (1987) Characterization of a mammalian mutant with a camptothecin-resistant DNA topoisomerase I. Proc Natl Acad Sci USA 84:5565–5569
Bungo M, Fujiwara Y, Kasahara K, Nakagawa K, Ohe Y, Sasaki Y, Irino S, Saijo N (1990) Decreased accumulation as a mechanism of resistance tocis-diamminedichloroplatinum(II) in human nonsmall cell lung cancer cell line; relation to DNA damage and repair. Cancer Res 50:25049–2553
Fujiwara Y, Kasahara K, Sugimoto Y, Nishio K, Ohmori T, Saijo N (1990) Detection of proteins that recognize platinum-modified DNA using gel mobility shift assay. Jpn J Cancer Res 81:1210–1213
Fukuoka M, Niitani H, Suzuki S, Motomiya M, Hasegawa K, Nishiwaki Y, Kuriyama T, Ariyoshi Y, Negoro S, Masuda N, Nakajima S, Taguchi T (1992) A phase II study of CPT-11, a new derivative of camptothecin, for previously untreated non-small cell lung cancer. J Clin Oncol 10:16–20
Gupta RS, Gupta R, Eng B, Lock RB, Ross WE, Hertzberg RR, Caranga MJ, Johnson RK (1988) Camptothecin-resistant mutants of Chinese hamster ovary cells containing a resistant form of topoisomerase I. Cancer Res 48:6404–6410
Hsiang YH, Hertzberg R, Hecht S, Liu LF (1985) Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J Biol Chem 260:14873–14875
Kaneda N, Nagata H, Furuta T, Yokokura T (1990) Metabolism and pharmacokinetics of the camptothecin analogue CPT-11 in the mouse. Cancer Res 50:1715–1720
Kanzawa F, Sugimoto Y, Minato K, Kasahara K, Bungo M, Nakagawa K, Fujiwara Y, Liu LF, Saijo N (1990) Establishment of a camptothecin analogue (CPT-11)-resistant cell line of human non small cell lung cancer: characterization and mechanism of resistance. Cancer Res 50:5919–5924
Karato A, Sasaki Y, Shinkai T, Eguchi K, Tamura T, Ohe Y, Oshita F, Nishio M, Kunikane H, Arioka H, Ohmatsu H, Nakashima H, Shiraishi J, Saijo N (1993) A phase I study of CPT-11 and VP-16. J Clin Oncol 11:2030–2035
Karato A, Nakashima H, Sasaki Y, Shiraishi J, Ohmatsu H, Oshita F, Atrioka H, Eguchi K, Kunikane H, Nishio M, Ohe Y, Tamura T, Shinkai T, Saijo N (1993) Pharmacokinetic analysis of the combination of CPT-11 and VP-16. Proc Jpn Cancer Assoc 52:2141
Kojima A, Shinkai T, Saijo N (1993) Cytogenetic effects of CPT-11 and its active metabolite, SN-38, on human lymphocytes. Jpn J Clin Oncol 23:116–122
Kasahara K, Fujiwara Y, Nishio K, Ohmori T, Sugimoto Y, Komiya K, Matsuda T, Saijo N (1991) Metallothionein content correlates with the sensitivity of human small cell lung cancer cell lines to cisplatin. Cancer Res 51:3237–3242
Kijima T, Kubota N, Nishio K (1994) Establishment of a CPT-11 resistant human ovarian cell line. Anticancer Res. (in press)
Kubota N, Kanzawa F, Nishio K, Takeda Y, Ohmori T, Fujiwaya Y, Terashima Y, Saijo N (1992) Detection of topoisomerase I gene point mutation in CPT-11 resistant lung cancer cell line. Biochem Biophys Res Commun 188:571–577
Kunimoto T, Nitta K, Tanaka T, Uehara N, Baba H, Takeuchi M, Yokokura T, Sawada S, Miysaka T, Mukai M (1987) Antitumor activity of 7-ethyl-10-[4-(1-piperidino)-1-piperidino]-carbonyloxy-camptothecin, a novel water-soluble derivative of camptothecin, against murine tumors. Cancer Res 47:5944–5947
Liu LF, Miller KG (1981) Eukaryotic DNA topoisomerases: two forms of type I DNA topoisomerases from HeLa cell nuclei. Proc Natl Acad Sci USA 78:3487–3491
Marikage T, Ohmori T, Nishio K, Fujiwara Y, Takeda Y, Saijo N (1993) Modulation of cisplatin sensitivity and accumulation by amphotericin B in cisplatin resistant human lung cance cell line. Cancer Res 53:3302–3307
Minato K, Kanzawa F, Nakagawa K, Sugimoto Y, Fujiwara Y, Nishio K, Saijo N (1990) Characterization of an etoposide-resistant human small cell lung cancer cell line. Cancer Chemother Pharmacol 26:313–317
Muggia FM, Creaven PJ, Hansen HH, Cohen, MH, Selawry OS (1972) Phase I clinical trial of weekly and daily treatment with camptothecin (NSC-100880): correlation with preclinical studies. Cancer Chemother Rep 56:515–521
Nakagawa K, Yokota J, Wada M, Sasaki Y, Fujiwara T, Tsunokawa Y, Terada M, Saijo N (1988) Levels of glutathione S transferase π mRNA in human lung cancer cell lines correlate with the resistance to cisplatin and carboplatin. Jpn J Cancer Res 79:301–304
Nakagawa K, Saijo N, Tsuchida S, Sakai M, Tsunokawa Y, Yokota J, Muramatsu M, Sato K, Terada M, Tew KD (1990) Glutathione-S-transferase as a determinant of drug resistance in transfectant cell line. J Biol Chem 265:4296–4301
Nakagawa K, Fukuoka M, Niitani H (1993) Phase II study of CPT-11 and cisplatin in patients with advanced non-small cell lung cancer. Proc Amer Soc Clin Oncol 12:1104
Negoro S, Fukuoka M, Masuda N, Takada M, Kusunoki Y, Matsui K, Takafuji N, Kudoh S, Niitani H, Taguchi T (1991) Phase I study of weekly intravenous infusions of CPT-11, a new derivative of camptothecin, in a treatment of advanced non-small cell lung cancer. J Natl Cancer Inst 83:1164–1168
Niimi S, Nakagawa K, Sugimoto Y, Nishio K, Fujiwara Y, Yokoyama S, Terashima Y, Saijo N (1992) Mechanism of cross-resistance to camptothecin analogue (CPT-11) in a human ovarian cancer cell line selected by cisplatin. Cancer Res 52:328–333
Nishio M, Arioka H, Nishio K, Kubota N, Heike Y, Ohata M, Saijo N (1993) Overexpression ofcis-diamminedichloroplatinum (II)-inducible proteins that are recognized by anti-HMG antibody in cisplatin resistant human lung cancer cell lines. Proc Jpn Cancer Assoc 52:2105
Ohe Y, Sasaki Y, Shinkai T, Eguchi K, Tamura T, Kojima K, Kunikane H, Okamoto H, Karato A, Ohmatsu H, Kanzawa F, Saijo N (1992) Phase I study and pharmacokinetics of CPT-11 with 5-day continuous infusion. J Natl Cancer Inst 84:972–974
Ohno R, Okada K, Masaoka T, Kuramoto A, Arima T, Yoshida Y, Ariyoshi H, Ichimaru M, Sakai Y, Ito Y, Morishima Y, Yokomaku S, Ota K (1990) An early phase II study of CPT-11; a new derivative of camptothecin, for the treatment of leukemia and lymphoma. J Clin Oncol 8:1907–1912
Pommier Y (1993) DNA topoisomerase I and II in cancer chemotherapy: update and perspectives. Cancer Chemother Pharmacol 32:103–108
Shimada Y, Yoshino M, Wakui A, Nakao K, Futatsuki K, Sakata Y, Kambe M, Taguchi T (1993) Phase II study of CPT-11, a new camptothecin derivative, in patient with metastatic colorectal cancer. J Clin Oncol 11:909–913
Shinkai T, Arioka H, Kunikane H, Eguchi K, Sasaki Y, Tamura T, Ohe Y, Nishio M, Karato A, Okamoto H, Nakashima H, Ohmatsu H, Shiraishi J, Saijo N (1994) A phase I clinical trial of CPT-11, 7-ethyl-10-[4-(1-piperidino)-1-piperidino]-carbonyloxy camptothecin, and cisplatin in combination with fixed dose of vindesine in advanced non-small cell lung cancer. Cancer Res (in press)
Wall ME, Wani MC, Cook CE, Palmar KH, Macphail AT, Simm GA (1966) Plant antitumor agents. 1. The isolation and structure of camptothecin, a novel alkaloid leukemia and tumor inhibition fromCamptotheca acuminata. J Am Chem Soc 88:388–3890
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saijo, N., Nishio, K., Kubota, N. et al. 7-Ethyl-10-[4-(1-piperidino)-1-piperidino] carbonyloxy camptothecin: mechanism of resistance and clinical trials. Cancer Chemother. Pharmacol. 34 (Suppl 1), S112–S117 (1994). https://doi.org/10.1007/BF00684874
Issue Date:
DOI: https://doi.org/10.1007/BF00684874