Summary
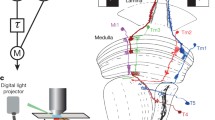
Intracellular recordings were made in the medullae of intact, restrained females ofCalliphora vicina that faced a hemispherical, minimum-distortion surface upon which moving patterns and spots were projected from the rear (Fig. 2). In the distal medulla, noisy hyperpolarizations to light, most likely recorded in terminals of laminar (L) cells, had flicker-like oscillations to moving gratings of 15° spatial wavelength but not of 2.5° spatial wavelength (Fig. 3). Medullary (M) cells penetrated distally responded to grating movements with similar but depolarizing oscillations, in one cell 180° out of phase with a nearby laminar response (Figs. 4–6).
A characteristic movement response recorded from most medullary cells consisted of abrupt, maintained nondirectional depolarizations in response to movements of gratings, often with directional ripple or spikes superimposed. When directions of movement reversed, there were brief repolarizations, but when movements stopped, depolarizations decayed away more slowly (Figs. 7 and 8). Magnitude of responses increased with increasing speeds of both 15° and 2.5° gratings (Figs. 9–11). In some cells, there were delayed decays of responses after stopping (Fig. 12). Still other cells seemed to receive inhibition from other, characteristically responding cells (Fig. 13).
Receptive fields tested were simple and usually large, with only a suggestion of surround inhibition (Fig. 14). In general, intensity and position were interchangeable over a cell's receptive field (Figs. 15 and 16). Moving edges and dark spots elicited responses primarily within receptive field centers (Figs. 18–20).
It is argued that waveforms of characteristic movement responses can be explained by multiplicative inputs from L- and M-cells to movement detectors (Figs. 21–26).
Similar content being viewed by others
Abbreviations
- L cells:
-
laminar (monopolar) cells
- M cells:
-
medullary cells
References
Barlow, H.B.: Visual resolution and the diffraction limit. Science149, 553–555 (1965)
Barlow, H.B., Levick, W.R.: The mechanism of directionally selective units in rabbit's retina. J. Physiol.178, 477–504 (1965)
Buchner, E.: Elementary movement detectors in an insect visual system. Biol. Cybern.24, 85–101 (1976)
Buchner, E., Götz, K.G., Straub, C.: Elementary detectors for vertical movement in the visual system ofDrosophila. Biol. Cybern.21, 235–242 (1978)
Buchner, E., Buchner, S., Hengstenberg, R.: 2-deoxy-D-glucose maps movement-specific nervous activity in the second visual ganglion ofDrosophila. Science205, 687–688 (1979)
Bülthoff, H., Götz, K.G.: Analogous motion illusion in man and fly. Nature278, 636–638 (1979)
Caldwell, J.H., Daw, N.W., Wyatt, HJ.: Effects of picrotoxin and strychnine on rabbit ganglion cells: lateral interactions for cells with more complex receptive fields. J. Physiol.276, 277–298 (1978)
Chen, A.C., Friedman, S.: An isotonic saline for the adult blowfly,Phormia regina, and its application to perfusion experiments. J. Insect Physiol.21, 529–536 (1975)
Collett, T.: Centripetal and centrifugal visual cells in medulla of the insect optic lobe. J. Neurophysiol.33, 239–255 (1970)
Collett, T., King, A.J.: Vision during flight. In: The compound eye and vision of insects. Horridge, G.A. (ed.), pp 437–466. Oxford: Clarendon Press 1975
DeVoe, R.D., Ockleford, E.M.: Intracellular responses from cells of the medulla of the fly,Calliphora erythrocephala. Biol. Cybern.23, 13–24 (1976)
Dvorak, D.R., Bishop, L.G., Eckert, H.E.: On the identification of movement detectors in the fly optic lobe. J. Comp. Physiol.100, 5–23 (1975)
Eckert, H.: Die spektrale Empfindlichkeit des Komplexauges vonMusca (Bestimmung aus Messungen der optomotorischen Reaktion). Kybernetik9, 145–156 (1971)
Eckert, H., Bishop, L.G.: Anatomical and physiological properties of the vertical cells in the third optic ganglion ofPhaenicia sericata (Diptera, Calliphoridae). J. Comp. Physiol.126, 57–86 (1978)
Franceschini, N.: Sampling of the visual environment by the compound eye of the fly: fundamentals and applications. In: Photoreceptor optics. Snyder, A.W., Menzel, R. (eds.), pp 98–125. Berlin, Heidelberg, New York: Springer 1975
French, A.S., Järvilehto, M.: The transmission of information by first and second order neurons in the fly visual system. J Comp. Physiol.126, 87–96 (1978)
Friedman, S.: Sustained flight inPhormia (by a new method) and its effect on blood pH. J. Insect Physiol.3, 118–119 (1959)
Geiger, G.: Optomotor responses of the flyMusca domestica to transient stimuli of edges and stripes. Kybernetik16, 37–43 (1974)
Gemperlein, R., Järvilehto, M.: Direkte Beobachtung der Rhabdomere beiCalliphora erythrocephala (Meig.). Z. Vergl. Physiol.65, 445–454 (1969)
Hardie, R.C.: Electrophysiological analysis of fly retina. I. Comparative properties of R1–6 and R7 and 8. J. Comp. Physiol.129, 19–33 (1979)
Hausen, K.: Functional characterization and anatomical identification of motion sensitive neurons in the lobula plate of the blowflyCalliphora erythrocephala. Z. Naturforsch.31c, 629–633 (1976)
Heisenberg, M., Buchner, E.: The role of retinula cell types in visual behavior ofDrosophila melanogaster. J. Comp. Physiol.117, 127–162 (1977)
Järvilehto, M., Zettler, F.: Electrophysiological-histological studies on some functional properties of visual cells and second order neurons of an insect retina. Z. Zellforsch.136, 291–306 (1973)
Kirschfeld, K.: The visual system ofMusca: studies on optics, structure and function. In: Information processing in the visual system of arthropods. Wehner, R. (ed.), pp 61–74. Berlin, Heidelberg, New York: Springer 1972
Kirschfeld, K., Lutz, B.: Lateral inhibition in the compound eye of the fly,Musca. Z. Naturforsch.29c, 95–97 (1974)
Laughlin, S.B., Hardie, R.C.: Common strategies for light adaptation in the peripheral visual systems of fly and dragonfly. J. Comp. Physiol.128, 319–340 (1978)
McCann, G.D.: Nonlinear identification theory models for successive stages of visual nervous systems of flies. J. Neurophysiol.37, 869–894 (1974)
McCann, G.D., Dill, J.C.: Fundamental properties of intensity, form and motion perception in the visual systems ofCalliphora phaenicia andMusca domestica. J. Gen. Physiol.53, 385–413 (1969)
McCann, G.D., MacGinitie, G.F.: Optomotor response studies of insect vision. Proc. R. Soc. (London) Biol.163, 369–401 (1965)
Marmarelis, P.Z., McCann, G.D.: Development and application of white-noise modeling techniques for studies of insect visual nervous system. Kybernetik12, 74–89 (1973)
Miller, R.F.: The neuronal basis of ganglion-cell receptive-field organization and the physiology of amacrine cells. In: Neurosciences, Fourth Study Program. Schmitt, F.O., Worden, F.G. (eds.), pp 227–245. Cambridge (Massachusetts). London: MIT Press 1979
Mimura, K.: Movement discrimination by the visual system of flies. Z. Vergl. Physiol.73, 105–138 (1971)
Mimura, K.: Analysis of visual information in lamina neurones of the fly. J. Comp. Physiol.88, 335–372 (1974)
Palka, J.: Diffraction and visual acuity in insects. Science149, 551–553 (1965)
Poggio, T., Reichardt, W.: Visual control of orientation behaviour in the fly. Part II. Towards the underlying neural interactions. Q. Rev. Biophys.9, 377–438 (1976)
Reichardt, W.: Autocorrelation, a principle for the evaluation of sensory information by the central nervous system. In: Sensory communication. Rosenblith, W.A. (ed.), pp 303–317. Massachusetts Institute of Technology: The M.I.T. Press; New York, London: John Wiley 1961
Reichardt, W., Poggio, T.: Visual control of orientation in the fly. Part I. A quantitative analysis. Q. Rev. Biophys.9, 311–375 (1976)
Rowell, C.H.F., O'Shea, M., Williams, J.L.D.: The neuronal basis of a sensory analyser, the acridid movement detector system IV. The preference for small field stimuli. J. Exp. Biol.68, 157–185 (1977)
Srinivasan, M.V., Bernard, G.D.: A proposed mechanism for multiplication of neural signals. Biol. Cybern.21, 227–236 1976
Srinivasan, M.V., Bernard, G.D.: The fly can discriminate movement at signal/noise ratios as low as one-eighth: Vision Res.17, 609–616 (1977)
Strausfeld, N.J.: Atlas of an insect brain. Berlin, Heidelberg, New York: Springer 1976
Torre, V., Poggio, T.: A synaptic mechanism possibly underlying directional sensitivity to motion. Proc. R. Soc. (London) Biol.202, 409–416 (1978)
Truxal, J.G.: Automatic feedback control system synthesis. New York, Toronto, London: McGraw-Hill 1955
Wyatt, H.J., Daw, N.W.: Directionally sensitive ganglion cells in the rabbit retina: specificity for stimulus direction, size and speed. J. Neurophysiol.38, 613–626 (1975)
Zaagman, W.H., Masterbroek, H.A.K., Buyse, T., Kuiper, J.W.: Receptive field characteristics of a directionally selective movement detector in the visual system of the blowfly. J. Comp. Physiol.116, 39–50 (1977)
Zaagman, W.H., Masterbroek, H.A.K., Kuiper, J.W.: On the correlation model: performance of a movement detecting neural element in the fly visual system. Biol. Cybern.31, 163–168 (1978)
Zettler, F., Weiler, R.: Neuronal processing in the first optic neuropile of the compound eye of the fly. In: Neural principles in vision. Zettler, F., Weiler, R. (eds.), pp 227–236. Berlin, Heidelberg, New York: Springer 1976
Zimmerman, R.P.: Field potential analysis and the physiology of second-order neurons in the visual system of the fly. J. Comp. Physiol.126, 297–316 (1978)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DeVoe, R.D. Movement sensitivities of cells in the fly's medulla. J. Comp. Physiol. 138, 93–119 (1980). https://doi.org/10.1007/BF00680435
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00680435