Abstract
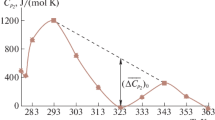
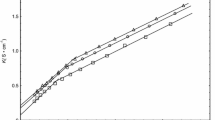
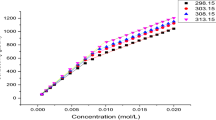
The enthalpy-entropy compensation in micellization of sodium dodecyl sulphate (SDS) in binary mixtures of water/methanol (MeOH), water/ethylene glycol (EG) and water/glycerol (GL) over a temperature range of 10–60°C was examined. When the cosolvent concentration was low, the critical micelle concentration (CMC) depended only on the total amount of the hydroxyl group added. When the cosolvent concentration was high, the increase in CMC followed the sequence: MeOH>EG>GL. Enthalpy and entropy changes were evaluated from which the compensation temperature was determined. Both enthalpy and entropy changes decreased on the addition of the cosolvents, indicating a lowering of solution hydrophobicity. The compensation temperature was found as a constant over the cosolvent concentration range, as a result, was not a good index for characterizing the solute/solvent interactions. The two reference temperatures at which the enthalpy-entropy change respectively became zero were strongly influenced by the cosolvent addition, therefore could serve as a proper index for solution hydrophobicity.
Similar content being viewed by others
References
Tanford C (1980) The Hydrophobic Effect, 2nd Edn., John-Willy, NY
Hunter RJ (1989) Foundations of Colloid Science, Vol. I, Oxford press, NY
Jolicoeur C, Philip PR (1974) Can J Chem 52:1834–1839
Goto A, Takemoto, M, Endo F (1985) Bull Chem Soc Jpn 58:247–251
Bedo Zs, Berecz E, Lakatos I (1992) Colloid Polym Sci 270: 799–805
Krishnan CV, Friedman HL (1973) J Soln Chem 2:37
Krishnan CV, Friedman HL (1973) J Soln Chem 2:119
Baldwins RL (1986) Proc Natl Acad Sci 83:8069–8072
Muller N (1988) J Soln Chem 17:661–672
Muller N (1990) Acc Chem Res 23:23–28
Privalov PL, Gill SJ (1989) Pure Appl Chem 61:1097–1104
Murphy KP, Privalov PL, Gill SJ (1990) Science 247:559–561
Gilli, P, Ferretti, V, Gilli G, Borea PA (1993) J Phys Chem 98:1515–1518
Madan B, Lee B (1994) Biophys Chem 51:279–289
Makhatadze GI, Privalov PL (1994) Biophys Chem 51:291–309
Murphy, KP (1994) Biophys Chem 51:311–326
Lee DJ (1995) Colloids Polym Sci 273:539–543
Mukerjee PM, Ray A (1963) J Phys Chem 67:190–192
Schick MJ (1964) J Phys Chem 68:3585–3592
Shirahama K, Matuura R (1965) Bull Chem Soc Jpn 38:373–378
Emerson MF, Holtzer A (1967) J Phys Chem 71:3320–3330
Shirahama K, Hayashi M, Matuura R (1969) Bull Chem Soc Jpn 42:1206–1212
Gratzer WB, Beaven GH (1969) J Phys Chem 73:2270–2273
Ray A, Nemethy G (1971) J Phys Chem 75:809–815
Hishikido N, Moroi Y, Uehara H, Matuura R (1974) Bull Chem Soc Jpn 47:2634–2638
Niyagishi S (1975) Bull Chem Soc Jpn 48:2349–2352
Kodama M, Boku R, Ishida T, Miura M (1977) Bull Chem Soc Jpn 50:751–752
Hayase K, Hayano S (1978) J Colloid Interf Sci 63:446
Hayase K, Hayano S (1978) Bull Chem Soc Jpn 51:933–934
Manabe M, Koda M, (1978) Bull Chem Soc Jpn 51:1534–1538
Singh HN, Swarup S (1978) Bull Chem Soc Jpn 51:1534–1538
Ionescu LG, Tokuhiro T, Czerniawski BJ (1979) Bull Chem Soc Jpn 52:922–924
Treiner C (1982) J Colloid Interf Sci 90:444–453
Almgren M, Swarup S (1982) J Phys Chem 86:4212–4216
Almgren M, Swarup S (1983) J Phys Chem 87:876–881
Kanungo SK, Sinha BK (1984) J Indian Chem Soc LXI:964–966
Moroi Y, Suggi R, Matuura R (1984) J Colloid Interf Sci 98:184–191
Panda L, Behera GB (1985) J Indian Chem Soc LXII:44–49
Almgren M, Swarup S, Lofroth JE (1985) J Phys Chem 89:4621–4626
Ramadan MSh, Evans DF, Lumry R, Philson S (1985) J Phys Chem 89:3405–3408
Pradhan P, Sinha BK (1987) Indian J Chem 26A:691
Deardon LV, Woolley EM (1987) J Phys Chem 91:4123–4127
Singh PK, Ahluwalia JC (1989) In Mittal KL (ed) Surfactants in Solution. Plenum Press, New York
Causi S, De Lisi R, Milioto S, Tirone N (1991) J Phys Chem 95:5664–5673
Nishikawa S, Matsuo F (1991) J Phys Chem 95:437–444
Moroi Y, Takagi H, Nagadome S, Hirata T, Sugihara G (1992) J Colloid Interf Sci 149:252–255
Challaghan A, Ronald D, Alexander E Palepu R (1993) Langmuir 9:3422–3426
Flockhart BD (1961) J Colloid Sci 16:484–492
La Mesa C (1990) J Phys Chem 94:323–326
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, D.J., Huang, W.H. Enthalpy-entropy compensation in micellization of sodium dodecyl sulphate in water/methanol, water/ethylene glycol and water/glycerol binary mixtures. Colloid Polym Sci 274, 160–165 (1996). https://doi.org/10.1007/BF00663448
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00663448