Abstract
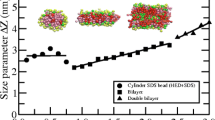
Using data on heat capacity of sodium decyl sulfate solutions near the third critical micelle concentration, obtained earlier via high-precision scanning calorimetry, the temperature dependences are calculated for the thermodynamic functions of the corresponding micellar transition and the components of its Gibbs free energy. It is shown that the considered intermicellar transition, interpreted as the micellar shape transforming from cylindrical to lamellar as the concentration grows, occurs at an equilibrium molality of 0.33 and a temperature of 323 K. Correlations between the thermodynamic functions of the transition with structural changes in the micelles and the key micellar parameters are discussed.



Similar content being viewed by others
REFERENCES
S. Lang, Curr. Opin. Colloid Interface Sci, No. 7, 12 (2002).
V. S. Kuznetsov, N. V. Usol’tseva, V. V. Bykova, V. P. Zherdev, and G. A. Anan’eva, Colloid. J. 67, 581 (2005).
V. S. Kuznetsov, N. V. Usol’tseva, V. P. Zherdev, and V. V. Bykova, Colloid. J. 72, 216 (2010).
V. S. Kuznetsov, V. G. Badelin, E. A. Tyunina, and V. P. Zherdev, Russ. J. Phys. Chem. A 93 (4) (2019, in press).
A. I. Serdyuk and R. V. Kucher, Micellar Transitions in Solutions of Surfactants (Naukova Dumka, Kiev, 1987) [in Russian].
N. V. Usol’tseva, Lyotropic Liquid Crystals: Chemical and Supramolecular Structure (Ivan. Gos. Univ., Ivanovo, 1994) [in Russian].
P. L. Privalov, FEBS Lett. 40 (Suppl.), 140 (1974).
A. A. Senin, S. A. Potekhin, E. J. Tiktopulo, and V. V. Filimonov, J. Therm. Anal. Calorim. 62, 153 (2000).
Y. N. Israelachvili, D. Y. Mitchell, and B. W. Ninham, J. Chem. Soc., Faraday Trans. II 72, 1525 (1976).
Ya. B. Zeldovich and A. D. Myshkis, Elements of Applied Mathematics (Khimiya, Moscow, 1967) [in Russian].
A. I. Rusanov, Micellization in Surfactant Solutions (Khimiya, St. Petersburg, 1992) [in Russian].
K. Holmberg, B. Jonsson, B. Kronberg, and B. Lindman, Surfactants and Polymers in Aqueous Solution (Wiley, New York, 2002; BINOM, Moscow, 2007).
A. I. Rusanov, A. P. Grinin, F. M. Kuni, and A. K. Shchekin, Russ. J. Gen. Chem. 72, 607 (2002).
Y. N. Israelachvili, Intermolecular and Surface Forces, 2nd ed. (Academic, London, 1992).
O. G. Us’yarov, Colloid. J. 69, 95 (2007).
V. P. Vasil’ev, Thermodynamic Properties of Electrolyte Solutions (Vysshaya Shkola, Moscow, 1982) [in Russian].
I. E. Tamm, Foundations of Electricity (Nauka, Moscow, 1976) [in Russian].
V. S. Kuznetsov, N. V. Usol’tseva, and V. P. Zherdev, Russ. J. Inorg. Chem. 59, 637 (2014).
ACKNOWLEDGMENTS
This work was supported by the Russian Foundation for Basic Research, project no. 15-29-01068 ofi_m.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by D. Terpilovskaya
Rights and permissions
About this article
Cite this article
Kuznetsov, V.S., Zherdev, V.P., Badelin, V.G. et al. Thermodynamics and Structure of Micelles in Aqueous Solutions of Sodium Decyl Sulfate in the Region of the Third Critical Micelle Concentration. Russ. J. Phys. Chem. 93, 1471–1477 (2019). https://doi.org/10.1134/S0036024419080156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024419080156