Summary
-
1.
The common response properties to simple visual stimuli (light impulses, light steps, and movement of simple patterns at different speeds) has been investigated by intracellular recording from Giant Vertical Cells (VS) in the lobula plate of the blowflyCalliphora erythrocephala.
-
2.
The impulse response begins < 10ms after onset of the photoreceptor signal (Fig. 6), and shows several phases which gradually subside within about 0.5 s. Very late events, which would hint at recurrent or far-reaching sidepaths, were not observed.
-
3.
The step response is highly non-linear in that both, the increase and decrease of brightness elicit transient depolarization. The excitatory transients are followed by inhibitory waves (Figs. 7, 8), similar to those observed in impulse responses. The possible significance of this succession of excitation and inhibition is discussed.
-
4.
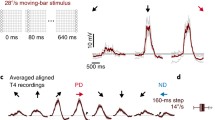
Vertical movement of arbitrary patterns (dot, edges, bar, and gratings) elicit, invariably and irrespective of contrast polarity, depolarizing responses with downward movement, and hyperpolarizing responses with upward movement (Fig. 10). Both responses increase nonlinearly with contour length (Fig. 11). Possible mechanisms, and the functional significance of such nonlinear summation are discussed.
-
5.
The velocity dependence of movement responses to periodic gratings was investigated at both high and low pattern luminance and contrast. Under these conditions VS-cells respond best at a contrast frequency of ≈ 2 Hz, which corresponds with that of velocity dependent optomotor reactions.
-
6.
These results confirm earlier findings that giant vertical cells have many response properties in common. They are best suited to perceive widefield motion, which occurs when a fly performs rotatory and translatory movements in a resting environment. VS-cells are therefore most likely involved in the visual control of such movements.
-
7.
The present results are not sufficient to indicate which of the VS-cells contribute to which of the optomotor reactions. A subsequent publication will be addressed to these questions.
Similar content being viewed by others
Abbreviations
- HS :
-
horizontal system
- VS :
-
vertical system
References
Blondeau J, Heisenberg M (1982) The threedimensional optomotor torque system ofDrosophila melanogaster. Studies on wildtype and the mutantoptomotor-blind H31. J Comp Physiol 145:321–329
Buchner E (1976) Elementary movement detectors in an insect visual system. Biol Cybern 24:85–101
Buchner E, Götz K, Straub C (1978) Elementary detectors for vertical movement in the visual system ofDrosophila. Biol Cybern 31:235–242
Dvorak DR, Bishop LG, Eckert HE (1975) On the identification of movement detectors in the fly optic lobe. J Comp Physiol 100:5–23
Eckert H (1979) Anatomie, Elektrophysiologie and funktionelle Bedeutung bewegungssensitiver Neurone in der Sehbahn von Dipteren (Phaenicia). Habilitationsschrift Abt Biologie, Ruhr-Universität Bochum
Eckert H, Bishop LG (1978) Anatomical and physiological properties of the vertical cells in the third optic ganglion ofPhaenicia sericata (Diptera, Calliphoridae). J Comp Physiol 126:57–86
Eckert H, Hamdorf K (1980) Excitatory and inhibitory response components in the landing response of the blowflyCalliphora erythrocephala. J Comp Physiol 138:253–264
Eckert H, Hamdorf K (1981) Action potentials in “nonspiking” visual interneurons. Z Naturforsch 36c:470–474
Fermi G, Reichardt W (1963) Optomotorische Reaktionen der FliegeMusca domestica. Kybernetik 2:15–28
Götz KG (1964) Optomotorische Untersuchung des visuellen Systems einiger Augenmutanten der FruchtfliegeDrosophila. Kybernetik 2:77–92
Götz KG (1965) Die optischen Übertragungseigenschaften der Komplexaugen vonDrosophila. Kybernetik 2:215–221
Götz KG (1968) Flight control inDrosophila by visual perception of motion. Kybernetik 4:199–208
Götz KG, Buchner E (1978) Evidence for one-way movement detection in the visual system ofDrosophila. Biol Cybern 31:243–248
Götz KG, Wenking H (1973) Visual control of locomotion in the walking fruitflyDrosophila. J Comp Physiol 85:235–266
Götz KG, Hengstenberg B, Biesinger R (1979) Optomotor control of wing beat and body posture inDrosophila. Biol Cybern 35:101–112
Hassenstein B (1958b) Die Stärke von optokinetischen Reaktionen auf verschiedene Mustergeschwindigkeiten. Z Naturforsch 13b:1–6
Hassenstein B, Reichardt W (1951) Funktionsanalyse der Bewe- gungsperzeption eines Käfers. Naturwissenschaften 21:507–508
Hassenstein B, Reichardt W (1956) Systemtheoretische Analyse der Zeit-, Reihenfolgen- und Vorzeichenauswertung bei der Bewegungsperzeption des RüsselkäfersChlorophanus. Z Naturforsch 11b: 513–524
Hausen K (1976a) Struktur, Funktion und Konnektivität bewegungsempfindlicher Interneurone im dritten optischen Neuropil der SchmeißfliegeCalliphora erythrocephala. Thesis, Universität Tübingen
Hausen K (1976b) Functional characterization and anatomical identification of motion sensitive neurons in the lobula plate of the blowflyCalliphora erythrocephala. Z Naturforsch 31 c:629–633
Hausen K (1981) Monocular and binoculor computation of motion in the lobula plate of the fly. Verh Dtsch Zool Ges 1981:49–70
Hengstenberg R (1971) Das Augenmuskelsystem der StubenfliegeMusca domestica. I. Analyse der “clock-spikes” und ihrer Quellen. Kybernetik 2:56–77
Hengstenberg R (1973) The effect of pattern movement on the impulse activity of the cervical connective ofDrosophila melanogaster. Z Naturforsch 28c: 593–596
Hengstenberg R (1977) Spike responses of “non-spiking” visual interneurone. Nature 270:338–340
Hengstenberg R (1981a) A piezoelectric device to aid penetration of small nerve fibres with microelectrodes. J Neurosci Methods 4:249–255
Hengstenberg R (1981b) Visuelle Drehreaktionen von Vertikalzellen in der Lobula Platte vonCalliphora. Verh Dtsch Zool Ges 1981:180
Hengstenberg R, Hengstenberg B (1980) Intracellular staining of insect neurons with procion yellow. In: Strausfeld NJ, Miller TA (eds) Neuroanatomical techniques. Springer, Berlin Heidelberg New York, pp 307–324
Hengstenberg R, Sandeman DC (1982) Kompensatorische Kopf-Rollbewegungen bei Fliegen. Verh Dtsch Zool Ges 1982 (in press)
Hengstenberg R, Hausen K, Hengstenberg B (1982) The number and structure of giant vertical cells (VS) in the lobula plate of the blowflyCalliphora erythrocephala. J Comp Physiol 149:163–177
Hengstenberg R, Bülthoff H, Hengstenberg B (in press) Three- dimensional reconstruction and stereoscopic display of nerve cells. In: Strausfeld NJ (ed) Neuroanatomical techniques. Springer, Berlin Heidelberg New Nork
Kirschfeld K (1979) The visual system of the fly: Physiological optics, and functional anatomy, as related to behaviour. In: Schmitt FO, Worden FG (eds) The neurosciences, IV. Study program. MIT Press, Cambridge, Massachusetts, pp 297–310
Payne R, Howard J (1981) Response of an insect photoreceptor: a simple log-normal model. Nature 290:415–416
Pierantoni R (1976) A look into the cock-pit of the fly: the architecture of the lobular plate. Cell Tissue Res 171:101–122
Poggio T, Reichardt W (1973) Considerations on models of movement detection. Kybernetik 3:223–227
Poggio T, Reichardt W (1976) Visual control of orientation behaviour in the fly. Part II. Toward the underlying neural interactions. Q Rev Biophys 9:377–438
Poggio T, Reichardt W, Hausen K (1981) A neuronal circuitry for relative movement discrimination by the visual system of the fly. Naturwissenschaften 68:443–446
Rowell CHF, O'Shea M, Williams JLD (1977) Neuronal basis of a sensory analyzer, the acridiid movement detector system. IV. The preference for small field stimuli. J Exp Biol 68:157–185
Soohoo SL, Bishop LG (1980) Intensity and motion responses of giant vertical neurons in the fly eye. J Neurobiol 11:159–177
Srinivasan MV (1977) A visually evoked roll response in the house fly. Open-loop and closed-loop studies. J Comp Physiol 119:1–14
Srinivasan MV, Dvorak DR (1980) Spatial processing of visual information in the movement detecting pathway of the fly. J Comp Physiol 140:1–23
Strausfeld NJ (1976) Atlas of an insect brain. Springer, Berlin Heidelberg New York
Thorson J (1966) Small signal analysis of a visual reflex in the locust. Kybernetik 3:53–66
Torre V, Poggio T (1978) A synaptic mechanism possibly underlying directional selectivity to motion. Proc R Soc Lond [Biol] 202:409–416
Wehrhahn C (1978a) Flight torque and lift responses of the housefly (Musca domestica) to a single stripe moving in different parts of the visual field. Biol Cybern 29:237–247
Wehrhahn C (1978b) The angular orientation of the movement detectors acting on the flight lift response in flies. Biol Cybern 31:169–173
Wehrhahn C, Reichardt W (1975) Visually induced height orientation of the flight lift response in the flyMusca. Biol Cybern 20:37–50
Wehrhahn C, Hausen K, Zanker J (1981) Is the landing response of the housefly (Musca) driven by motion of a flow field? Biol Cybern 41:91–99
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hengstenberg, R. Common visual response properties of giant vertical cells in the lobula plate of the blowflyCalliphora . J. Comp. Physiol. 149, 179–193 (1982). https://doi.org/10.1007/BF00619212
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00619212