Abstract
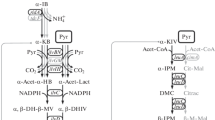
An isoleucine auxotroph of Salmonella typhimurium was derived from a merodiploid strain (containing the F′-14 episome from Escherichia coli) that contained two copies of the structural genes concerned with isoleucine and valine biosynthesis. A haploid derivative, strain TU6001, having the same growth properties as the original merodiploid mutant was found to have normal biosynthetic enzymes and an altered isoleucyl-tRNA synthetase. The K m for isoleucine was increased by about 200-fold over that for the wild-type enzyme. All five enzymes in the isoleucine and valine biosynthetic pathway were derepressed relative to wild-type enzyme levels. A partial revertant of strain TU6001 was isolated which had properties that were intermediate between those of the mutant and the wild type (i.e., intermediate growth dependence on exogenous isoleucine, intermediate activity of isoleucyl-tRNA synthetase, and intermediate derepression of biosynthetic enzymes). The properties of strain TU6001 were demonstrated to be simultaneously transferable by transduction (using PLT22 H4 bacteriophage) of a single genetic locus, linked to pyr A, which has been designated ilv S. It is concluded that some function of the isoleucyl-tRNA synthetase is important in repression of the isoleucine and valine biosynthetic enzymes.
Similar content being viewed by others
References
Arfin, S. M., Ratzkin, B., and Umbarger, H. E. (1969). The metabolism of valine and isoleucine in Escherichia coli. XVII. The role of induction in the derepression of acetohydroxy acid isomeroreductase. Biochem. Biophys. Res. Commun. 37902.
Bauerle, R. H., Freundlich, M., Størmer, F. C., and Umbarger, H. E. (1964). Control of isoleucine, valine, and leucine biosynthesis. II. Endproduct inhibition by valine of acetohydroxy acid synthetase in Salmonella typhimurium. Biochim. Biophys. Acta 92142.
Blatt, J. M., and Umbarger, H. E. (1970). The relation of isoleucyl-tRNA synthetase to repression of branched-chain amino acid biosynthetic enzymes in Salmonella typhimurium. Bacteriol. Proc., p. 136.
Brenner, M., De Lorenzo, F., and Ames, B. N. (1970). Histidyl-tRNA synthetase: Kinetics, genetics, physiology, and role in repression. Federation Proc. 29467.
Burns, R. O., Umbarger, H. E., and Gross, S. R. (1963). The biosynthesis of leucine. III. The conversion of α-hydroxy-β-carboxyisocaproate to α-ketoisocaproate. Biochemistry 21053.
Calvo, J., and Umbarger, H. E. (1963). Feedback control of leucine biosynthesis in Salmonella typhimurium. Bacteriol. Proc., p. 126.
Cantoni, G. L., and Richards, H. H. (1966). Preparation and properties of sRNA from rabbit liver. in Cantoni, G. L., and Richards, D. R. (eds.), Procedures in Nucleic Acid Research, Harper and Row, New York, pp. 617–623.
Cleland, W. W. (1963a). The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim. Biophys. Acta 67104.
Cleland, W. W. (1963b). Computer programmes for processing enzyme kinetic data. Nature 198463.
Coker, M., and Umbarger, H. E. (1970). Genetic lesions leading to thiaisoleucine resistance in Escherichia coli. Bacteriol. Proc., p. 135.
Davis, B. D., and Mingioli, E. S. (1950). Mutants of Escherichia coli requiring methionine of vitamin B12. J. Bacteriol. 6017.
Demerec, M., and Ohta, N. (1964). Genetic analysis of Salmonella typhimurium × Escherichia coli hybrids. Proc. Natl. Acad. Sci. 52317.
Dwyer, S. B., and Umbarger, H. E. (1968). Isoleucine and valine metabolism of Escherichia coli. XVI. Pattern of multivalent repression in strain K-12. J. Bacteriol. 951680.
Eidlic, L., and Neidhardt, F. N. (1965). Role of valyl-sRNA synthetase in enzyme repression. Proc. Natl. Acad. Sci. 53539.
Freundlich, M. (1963). Multivalent repressionof the biosynthesis of threonine in Salmonella typhimurium and Escherichia coli. Biochem. Biophys. Res. Commun. 10277.
Hatfield, G. W., and Burns R. O. (1970). Specific binding of leucyl transfer RNA to an immature form of l-threonine deaminase: Its implications in repression. Proc. Natl. Acad. Sci. 661027.
Hiraga, S., Ito, K., Hamada, K., and Yura. T. (1967). A new regulatory gene for the tryptophan operon of Escherichia coli. Biochem. Biophys. Res. Commun. 26522.
Hirota, Y. (1960). The effect of acridine dyes on mating type factors in Escherichia coli. Proc. Natl. Acad. Sci. 4657.
Iaccarino, M., and Berg, P. (1971). Isoleucine auxotrophy as a consequence of a mutationally altered isoleucyl tRNA synthetase. J. Bacteriol. 105527.
Jacob, F., and Monod, J. (1961). Genetic regulatory mechanisms in the synthesis of proteins. J. Mol. Biol. 3318.
Lane, E. (1957). Spectrophotometric and turbidimetric methods for measuring proteins. In Colowick, S. P., and Kaplan, N. O. (eds.), Methods in Enzymology, Vol. 3, Academic Press, Inc., New York, pp. 447–454.
Langdon, R. G. (1966). Glucose 6-phosphate dehydrogenase from erythrocytes. In Colowick, S. P., and Kaplan, N. O. (eds.), Methods in Enzymology, Vol. 9, Academic Press, Inc., New York, pp. 216–231.
Lennox, E. S. (1955). Transduction of linked genetic characters of the host by bacteriophage P1. Virology 1190.
Mans, R. J., and Novelli, G. D. (1961). Measurement of the incorporation of radioactive amino acids into protein by a filter-paper disk method. Arch. Biochem. Biophys. 9448.
Nass, G., and Neidhardt, F. C. (1967). Regulation of formation of aminoacyl-ribonucleic acid synthetases in Escherichia coli. Biochim. Biophys. Acta. 134347.
National Research Council (1967). Specifications and Criteria for Biochemical Compounds, 2nd ed., Natl. Acad. Sci. Publ. No. 1344.
Novelli, G. D. (1967). Amino acid activation for protein synthesis. In Boyer, P. D. (ed.), Annual Review of Biochemistry, vol. 36, Annual Reviews, Inc., Palo Alto, Calif., p. 449–484.
Power, J. (1967). The l-rhamnose genetic system in Escherichia coli K-12. Genetics 55557.
Ramakrishnan, T., and Adelberg, E. A. (1964). Regulatory mechanisms in the biosynthesis of isoleucine and valine. I. Genetic derepression of enzyme formation. J. Bacteriol. 87566.
Roth, J. R., and Ames, B. N. (1966). Histidine regulatory mutants having altered histidyl-tRNA synthetase. J. Mol. Biol. 22326.
Sanderson, K. E. (1970). Current linkage map of Salmonella typhimurium. Bacteriol. Rev. 34176.
Silbert, D. F., Fink, G. R., and Ames, B. N. (1966). Histidine regulatory mutants in Salmonella typhimurium. III. A class of regulatory mutants deficient in tRNA for histidine. J. Mol. Biol. 22335.
Stetson, H. A. (1969). Ph.D. thesis, Purdue University.
Szentirmai, A., and Umbarger, H. E. (1968). Isoleucine and valine metabolism of Escherichia coli. XIV. Effect of thiaisoleucine. J. Bacteriol. 951666.
Szentirmai, A., Szentirmai, M., and Umbarger, H. E. (1968). Isoleucine and valine metabolism of Escherichia coli. XV. Biochemical properties of mutants resistant to thiaisoleucine. J. Bacteriol. 951672.
Taylor, R. T., and Jenkins, W. T. (1966). Leucine aminotransferase. I. Colorimetric assays. J. Biol. Chem. 2414391.
Truffa-Bachi, P., LeBras, G., and Cohen, G. (1966). The threonine-sensitive homoserine dehydrogenase and aspartokinase activities of Escherichia coli. II. The effects of p-mercuribenzoic acid. Biochim. Biophys. Acta 128440.
Umbarger, H. E. (1956). Evidence for a negative feedback mechanism in the biosynthesis of isoleucine. Science 123848.
Vogel, H. J., and Bonner, D. M. (1956). Acetylornithinase of Escherichia coli. Partial purification and some properties. J. Biol. Chem. 21897.
Yegian, C. D., and Stent, G. S. (1969). Differential aminoacylation of three species of isoleucine transfer RNA from Escherichia coli. J. Mol. Biol. 3959.
Author information
Authors and Affiliations
Additional information
Supported by grant GM 12522 from the National Institute of General Medical Sciences, U.S. Public Health Service. J. M. B. received a U.S. Public Health Service Postdoctoral Fellowship 1-F02-GM-30, 650-02.
Rights and permissions
About this article
Cite this article
Blatt, J.M., Umbarger, H.E. On the role of isoleucyl-tRNA synthetase in multivalent repression. Biochem Genet 6, 99–118 (1972). https://doi.org/10.1007/BF00486395
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00486395