Abstract
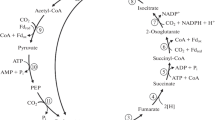
The strict anaerobe Desulfobacter hydrogenophilus is able to grow autotrophically with CO2, H2, and sulfate as sole carbon and energy sources. The generation time at 30°C under autotrophic conditions in a pure mineral medium was 15 h, the growth yield was 8 g cell dry mass per mol sulfate reduced to H2S. Enzymes of the autotrophic CO2 assimilation pathway were investigated. Key enzymes of the Calvin cycle and of the acetyl CoA pathway could not be found. All enzymes of a reductive citric acid cycle were present at specific activities sufficient to account for the observed growth rate. Notably, an ATP-citrate lyase (1.3 μmol · min-1 · mg cell protein-1) was present both in autotrophically and in heterotrophically grown cells, which was rapidly inactivated in the absence of ATP. The data indicate that in D. hydrogenophilus a reductive citric acid cycle is operating in autotrophic CO2 fixation. Since other autotrophic sulfate reducers possess an acetyl CoA pathway for CO2 fixation, two different autotrophic pathways occur in the same physiological group.
Similar content being viewed by others
References
Alvarez M, Barton LL (1977) Evidence for the presence of phosphoriboisomerase and ribulose-1,5-diphosphate carboxylase in extracts of Desulfovibrio vulgaris. J Bacteriol 131:133–135
Anfinsen CB (1955) Aconitase from pig heart muscle, In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 1. Academic Press, New York, pp 695–698
Badziong W, Thauer RK (1978) Growth yields and growth rates of Desulfovibrio vulgaris (Marburg) growing on hydrogen plus sulfate and hydrogen plus thiosulfate as the sole energy sources. Arch Microbiol 117:209–214
Bergmeyer HU (1974) Methoden der enzymatischen Analyse, 3rd ed. Verlag Chemie, Weinheim
Bernt E, Bergmeyer HU (1979) Isocitrate-dehydrogenase. In: Bergmeyer HU (ed) Methoden der enzymatischen Analyse, vol 1. Verlag Chemie, Weinheim, pp 664–667
Brandis-Heep A, Gebhardt NA, Thauer RK, Widdel F, Pfennig N (1983) Anaerobic acetate oxidation to CO2 by Desulfobacter postgatei. 1. Demonstration of all enzymes required for the operation of the citric acid cycle. Arch Microbiol 136:222–229
Brysch K, Schneider C, Fuchs G, Widdel F (1987) Lithoautotrophic growth of sulfate-reducing bacteria, and description of Desulfobacterium autotrophicum. Arch Microbiol (in press)
Buchanan BB (1979) Ferredoxin-linked carbon dioxide fixation in photosynthetic bacteria. In: Gibbs M, Latzko E (ed) Photosynthesis II. Encyclopedia of plant physiology, New Series 6. Springer, Berlin Heidelberg New York, pp 416–424
Cánovas JL, Kornberg HL (1969) Phosphoenolpyruvate carboxylase from Escherichia coli. In: Lowenstein JM (ed) Methods in enzymology, vol XIII. Academic Press, New York London, pp 288–296
Dorn M, Andreesen JR, Gottschalk G (1978) Fermentation of fumarate and l-malate by Clostridium formicoaceticum. J Bacteriol 133:26–32
Evans MCW, Buchanan BB, Arnon DI (1966) A new ferredoxin dependent carbon reduction cycle in a photosynthetic bacteria. Proc Natl Acad Sci USA 55:928–934
Eyzaguirre J, Jansen K, Fuchs G (1982) Phosphoenolpyruvate synthetase in Methanobacterium thermoautotrophicum. Arch Microbiol 132:67–74
Fuchs G (1986) CO2 fixation in acetogenic bacteria: variations on a theme. FEMS Microbiol Rev 39:181–213
Fuchs G, Stupperich E (1986) Carbon assimilation pathways in archaebacteria. System Appl Microbiol 7:365–369
Gebhardt NA, Linder D, Thauer RK (1983) Anaerobic acetate oxidation to CO2 by Desulfobacter postgatei. 2. Evidence from 14C-labelling studies for the operation of the citric acid cycle. Arch Microbiol 136:230–233
Goa J (1953) A microbiuret method for protein determination: determination of total protein in cerebrospinal fluid. Scand J Clin Lab Invest 5:218
Ivanovsky RN, Sintsov NV, Kondratieva EN (1980) ATP-linked citrate lyase activity in the green sulfur bacterium Chlorobium limicola forma thiosulfatophilum. Arch Microbiol 128:239–241
Jansen K, Thauer RK, Widdel F, Fuchs G (1984) Carbon assimilation pathways in sulfate reducing bacteria. Formate, carbon dioxide, carbon monoxide, and acetate assimilation by Desulfovibrio baarsii. Arch Microbiol 138:257–262
Jansen K, Fuchs G, Thauer RK (1985) Autotrophic CO2 fixation by Desulfovibrio baarsii: demonstration of enzyme activities characteristic for the acetyl-CoA pathway. FEMS Microbiol Lett 28:311–315
Kuenen JG, Veldkamp H (1972) Thiomicrospira elophila gen. nov. sp. nov., a new obligatory chemolithotrophic coulourless sulfur bacterium. Antonie van Leeuwenhoek Microbiol Serol 38: 241–256
Möller D, Thauer RK (1987) Acetate oxidation to CO2 via a citric acid cycle involving an ATP-citrate lyase: mechanism for the synthesis of ATP via substrate level phosphorylation in Desulfobacter postgatei growing on acetate and sulfate. Arch Microbiol (in press)
Oberlies G, Fuchs G, Thauer RK (1980) Acetate thiokinase and assimilation of acetate in Methanobacterium thermoautotrophicum. Arch Microbiol 128:248–252
Quayle JR, Keech DB (1959) Carbon assimilation by Pseudomonas oxalaticus (OX1). 2. Formate and carbon dioxide utilization by cell free extracts of the organism grown on formate. Biochem J 72:631–637
Schauder R, Eikmanns B, Thauer RK, Widdel F, Fuchs G (1986) Acetate oxidation to CO2 in anaerobic bacteria via a novel pathway not involving reactions of the citric acid cycle. Arch Microbiol 145:162–172
Schlegel HG, Kaltwasser H, Gottschalk G (1961) Ein Submersverfahren zur Kultur wasserstoffoxidierender Bakterien: Wachstumsphysiologische Untersuchungen. Arch Mikrobiol 38:209–222
Shiba H, Kawasumi T, Igarashi Y, Kodama T, Minoda Y (1985) The CO2 assimilation via the reductive tricarboxylic acid cycle in an obligatory autotrophic, aerobic hydrogen-oxidizing bacterium Hydrogenobacter thermophilus. Arch Microbiol 141:198–203
Simon H, Floss HG (ed) (1967) Anwendung von Isotopen in der organischen Chemie und Biochemie, Bd. I. Springer, Berlin Heidelberg New York
Sirevåg R (1974) Further studies on carbon dioxide fixation in Chlorobium. Arch Microbiol 98:3–18
Sorokin Y (1966a) Sources of energy and carbon for biosynthesis in sulfate-reducing bacteria. Microbiology USSR 35:643–647
Sorokin Y (1966b) Investigations of the structural metabolism of sulfate-reducing bacteria with 14C. Microbiology USSR 35: 806–814
Stams AJM, Kremer DR, Nicolay K, Weenk GH, Hansen TA (1984) Pathway of propionate formation in Desulfobulbus propionicus. Arch Microbiol 139:167–173
Stupperich E, Fuchs G (1984) Autotrophic synthesis of activated acetic acid from two CO2 in Methanobacterium thermoautotrophicum. I. Properties of in vitro system. Arch Microbiol 139:8–13
Tabatabai MA (1974) Determination of sulfate in water sample. Sulphur Inst J 10:11–13
Widdel F (1987a) New types of the acetate-oxidizing, sulfate-reducing Desulfobacter species, D. hydrogenophilus sp. nov., D. latus sp. nov., and D. curvatus sp. nov. Arch Microbiol (in press)
Widdel F (1987b) Microbiology and ecology of sulfate- and sulfurreducing bacteria. In: Zehnder AJB (ed) Environmental microbiology of anaerobes, chapt 10. John Wiley & Sons, New York London (in press)
Widdel F, Pfennig N (1984) Dissimilatory sulfate- or sulfur-reducing bacteria. In: Krieg NR, Holt JG (eds) Bergey's manual of systematic bacteriology, IX ed, vol 1. Williams and Wilkins, Baltimore, pp 663–679
Williamson JR, Corkey BE (1969) Assays of intermediates of the citric acid cycle and related compounds by fluorometric enzyme methods. In: Lowenstein JM (ed) Methods in enzymology, vol XIII. Academic Press, New York, pp 434–513
Wood HG, Ragsdale SW, Pezacka E (1986) The acetyl-CoA pathway of autotrophic growth. FEMS Microbiol Rev 39:345–362
Zeikus JG, Fuchs G, Kenealy W, Thauer RK (1977) Oxidoreductase involved in cell carbon synthesis of Methanobacterium thermoautotrophicum. J Bacteriol 132:604–613
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. H. G. Wood on the occasion of his 80th birthday
Rights and permissions
About this article
Cite this article
Schauder, R., Widdel, F. & Fuchs, G. Carbon assimilation pathways in sulfate-reducing bacteria II. Enzymes of a reductive citric acid cycle in the autotrophic Desulfobacter hydrogenophilus . Arch. Microbiol. 148, 218–225 (1987). https://doi.org/10.1007/BF00414815
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00414815