Abstract
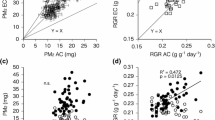
CO2 compenstation point (Γ), the concentration of CO2 at which photosynthesis and respiration are at equilibrium, is a commonly used diagnostic for the C4 photosynthetic pathway, since it reflects the reduced photorespiration that is a property of C4 photosynthesis. Geographic variation for Γ was examined within Flaveria linearis, a C3−C4 intermediate species. Collections from four widely separated Floridian populations were propagated in a greenhouse and measured for Γ. Little differentiation among populations was found, but significant within-population variation was present. Temperature is a hypothesized selective agent for the C4 photosynthetic pathway. To test this hypothesis, plants exhibiting a range of Γ were cloned and placed in growth chambers at 25°C and 40°C. After 7 weeks, Γ valves were remeasured and plants were harvested and weighed. There was a poor correlation between initial and final measures of Γ for a given genotype (r=0.38, P>0.1). Broad sense heritability for Γ was computed to be 0.10. At 25°C, there was no relationship between final size and Γ. At 40°C, more C4-like plants, as indicated by their low Γ, had grown larger. Differences in relative growth rate were attributable more to differences in net assimilation rate than in leaf area ratio. Taken together, these results demonstrate that although significant plasticity exists in the amount of photorespiration in this C3−C4 species, high temperature appears to be an effective selective agent for the reduction of photorespiration and the enhancement of C4-like traits.
Similar content being viewed by others
References
Araus JL, Brown RH, Byrd GT, Serret MD (1991) Comparative effects of growth irradiance on photosynthesis and leaf anatomy of Flaveria brownii (C4-like), Flaveria linearis (C3−C4) and their F1 hybrid. Planta 183:497–504
Barton NH, Turelli M (1989) Evolutionary quantitative genetics: how little do we know? Annu Rev Genet 23:337–370
Brown RH, Hattersley PW (1989) Leaf anatomy of C3−C4 species as related to evolution of C4 photosynthesis. Plant Physiol 91: 1543–1550
Brown RH, Bassett CL, Cameron RG, Evans PT, Bouton JH, Black CCJ, Sternberg LO, DeNiro MJ (1986) Photosynthesis of F1 hybrids between C4 and C3−C4 species of Flaveria. Plant Physiol 82:211–217
Caemmerer S von (1989) A model of photosynthetic CO2 assimilation and carbon-isotope discrimination in leaves of certain C3−C4 intermediates. Planta 178:463–474
Clegg MD, Sullivan CY, Eastin JD (1978) A sensitive technique for the rapid measurement of carbon dioxide concentration. Plant Physiol 62:924–926
Dijkstra P, Lambers H (1989) A physiological analysis of genetic variation in relative growth rate within Plantago major L. Funct Ecol 3:577–587
Doliner LH, Jolliffe PA (1979) Ecological evidence concerning the adaptive significance of the C4 dicarboxylic acid pathway of photosynthesis. Oecologia 38:23–34
Edwards GE, Ku MSB (1987) Biochemistry of C3−C4 intermediates. In Stumpf PK, Conn EE (eds) The biochemistry of plants: a comprehensive treatise, vol 10. Academic Press, New York, pp 275–325
Ehleringer J, Monson R (1993) Evolutionary and ecological aspects of photosynthetic pathway variation. Annu Rev Ecol Syst 24: 411–440
Ehleringer J, Pearcy R (1983) Variation in quantum yield for CO2 uptake among C3 and C4 plants. Plant Physiol 73:555–559
Ehleringer J, Sage R, Flanagan L, Pearcy R (1991) Climate change and the evolution of C4 photosynthesis. Trends Ecol Evol 6:95–99
Ehrlich PR, Raven PH (1969) Differentiation of populations. Science 165:1228–1232
Falconer DS (1981) Introduction to quantitative genetics. Longman Scientific and Technical, Harlow, England
Hatch MD (1987) C4 photosynthesis: a unique blend of modified biochemistry, anatomy, and ultrastructure. Biochim Biophys Acta 895:81–106
Hattersley PW (1986) Variations in photosynthetic pathway. In: Soderstrom TR (ed) Grass systematics and evolution. Smithsonian Institution, Washington DC, pp 49–64
Holaday AS, Lee KW, Chollet R (1984) C3−C4 intermediate species in the genus Flaveria: leaf anatomy, ultrastructure, and the effect of O2 on the CO2 compensation concentration. Planta 160:25–32
Ku MSB, Monson RK, Littlejohn RO, Nakamoto H, Fisher DB, Edwards GE (1983) Photosynthetic characteristics of C3−C4 intermediate Flaveria species. I. Leaf anatomy, photosynthetic responses to O2 and CO2, and activities of key enzymes in the C3 and C4 pathways. Plant Physiol 71:944–948
Ku MSB, Wu J, Dai Z, Scott RA, Chu C, Edwards GE (1991) Photosynthetic and photorespiratory characteristics of Flaveria species. Plant Physiol 96:518–528
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261
Lande R, Arnold SJ (1983) The measurement of selection on correlated characters. Evolution 37:1210–1226
Long SP (1983) C4 photosynthesis at low temperatures. Plant Cell Environ 6:345–363
Mitchell-Olds T, Rutledge JJ (1986) Quantitative genetics in natural plant populations: a review of the theory. Am Nat 127: 379–402
Monson RK (1989) On the evolutionary pathways resulting in C4 photosynthesis and Crassulacean acid metabolism (CAM). Adv Ecol Res 19:57–109
Monson RK, Jaeger CJ (1991) Photosynthetic characteristics of C3−C4 intermediate Flaveria floridana (Asteraceae) in natural habitats: evidence of advantages to C3−C4 photosynthesis in high leaf temperatures. Am J Bot 78:795–800
Monson RK, Moore BD (1989) On the significance of C3−C4 intermediate photosynthesis to the evolution of C4 photosynthesis. Plant Cell Environ 12:689–699
Monson RK, Edwards GE, Ku MSB (1984) C3−C4 intermediate photosynthesis in plants. BioScience 34:563–574
Monson RK, Moore BD, Ku MSB, Edwards GE (1986) Co-function of C3- and C4-photosynthetic pathways in C3, C4 and C3−C4 intermediate Flaveria species. Planta 168:493–502
Pearcy RW, Tumosa N, Williams K (1981) Relationships between growth, photosynthesis and competitive interactions for a C3 and a C4 plant. Oecologia 48:371–376
Pearcy RW, Björkman O, Caldwell MM, Keeley JE, Monson RK, Strain BR (1987) Carbon gain by plants in natural environments. BioScience 37:21–29
Peisker M (1986) Models of carbon metabolism in C3−C4 intermediate plants as applied to the evolution of C4 photosynthesis. Plant Cell Environ 9:627–635
Peisker M, Apel P, Ticha I, Hak R (1988) CO2 compensation concentration in relation to leaf insertion level in three Flaveria species with different photosynthetic pathways. Photosynthetica 22:1–8
Poorter H, Remkes C (1990) Leaf area ratio and net assimilation rate of 24 wild species differing in relative growth rate. Oecologia 83:553–559
Potvin C, Tardif S (1988) Sources of variability and experimental design in growth chambers. Funct Ecol 2:123–130
Powell AM (1978) Systematics of Flaveria (Flaveriinac-Asteraceae). Ann Mo Bot Gard 65:590–636
SAS Institute (1990) SAS/STAT user's guide. SAS Institute, Cary, NC
Sayre RT, Kennedy RA (1977) Ecotypic differences in the C3 and C4 photosynthetic activity in Mollugo verticillata, a C3−C4 intermediate. Planta 134:257–262
Schuster WS, Monson RK (1990) An examination of the advantages of C3−C4 intermediate photosynthesis in warm environments. Plant Cell Environ 13:903–912
Sokal RR, Rohlf FJ (1981) Biometry. Freeman, New York
Teeri JA, Stowe LG (1976) Climatic patterns and the distribution of C4 grasses in North America. Oecologia 23:1–12
US Department of Commerce (1989) World weather records. US Dept Commerce, Washington, DC
Wade MJ, Kalisz S (1990) The causes of natural selection. Evolution 44:1947–1955
Winsberg MD (1990) Florida weather. University of Central Florida Press, Orlando, FL
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Teese, P. Intraspecific variation for CO2 compensation point and differential growth among variants in a C3−C4 intermediate plant. Oecologia 102, 371–376 (1995). https://doi.org/10.1007/BF00329804
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00329804