Summary
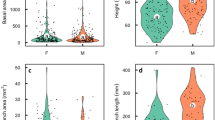
Flowering individuals of dwarf ginseng may be either male or hermaphroditic. I recorded the sex expression and size of individuals in three populations for three or four years in order to 1) determine whether this bimodal distribution of sex expression was due to sex changing or genetic dimorphism, and 2) test predictions about a) the relationship between size and gender, and b) the association of size change and sex change. Twenty five to 37% of the flowering individuals in each population changed gender from one year to the next. Of the plants I followed for four years, 83% changed sex and 57% changed more than once. In each of these populations as well as two others, hermaphrodites were significantly larger than males. Gender dynamics of the three populations differed, but hermaphrodites tended to become smaller and were more likely to change gender than remain hermaphroditic the following year, whereas males tended to grow larger and were more likely to remain male than to change gender. Dwarf ginseng is clearly a diphasic (sex changing) species in which sex expression is determined primarily by size. A difference between genders in the immediate resource costs of reproduction appears to be an important determinant of sex change and gender phase ratios in populations.
Similar content being viewed by others
References
Bawa KS, Beach JH (1981) Evolution of sexual systems in flowering plants. Ann M Bot Gard 68: 254–274
Bierzychudek P (1982) The demography of jack-in-the-pulpit, a forest perennial that changes sex. Ecol Monogr 52: 335–351
Bierzychudek P (1984) Determinants of gender in jack-in-the-pulpit: the influence of plant size and reproductive history. Oecologia 65: 14–18
Charlesworth B, Charlesworth D (1978) A model for the evolution of dioecy and gynodioecy. Am Natur 112: 975–997
Charlesworth D (1984) Androdioecy and the evolution of dioecy. Biol J Linn Soci 23: 333–348
Charnov EL (1982) The Theory of Sex Allocation. Princeton University Press, Princeton
Charnov EL, Bull JJ (1989) Non-fisherian sex ratios with sex change and environmental sex determination. Nature 338: 148–150
Condon MA, Gilbert LE (1988) Sex expression ofGurania andPsiguria (Cucurbitaceae): neotropical vines that change sex. Am J Botany 75: 875–884
Condon MA, Gilbert LE (1990) Reproductive biology and natural history of neotropical vinesGurania andPsiguria (=Anguria). In: Bates D, (ed) Biology and Chemistry of Cucurbitaceae. Cornell University Press, Ithaca
fienberg SE (1980) The Analysis of Cross-classified Categorical Data. 2nd ed. The MIT Press, Cambridge
Freeman DC, Harper KT, Charnov EL (1980) Sex change in plants: old observations and new hypotheses. Oecologia 47: 222–223
Freeman DC, McArthur ED (1984) The relative influences of mortality, nonflowering and sex change on the sex ratios of sixAtriplex species. Bot Gaz 145: 385–394
Freeman DH, Jr. (1987) Applied Categorical Data Analysis. Marcel Dekker, Inc., New York
Ghiselin MT (1969) The evolution of hermaphroditism among animals. Quart Rev Biol 44: 189–208
Gregg KB (1975) The effect of light intensity on sex expression in species ofCycnoches andCatasetum (Orchidaceae). Selbyana 1: 101–113
Gregg KB (1978) The interaction of light intensity, plant size, and nutrition on sex expression inCycnoches (Orchidaceae). Selbyana 2: 212–223
Gregg KB (1982) Sunlight-enhanced ethylene evolution by developing inflorescences ofCatasetum andCycnoches and its relation to female flower production. Bot Gaz 143: 466–475
Leigh EG, Charnov EL, Warner RR (1976) Sex ratio, sex change, and natural selection. Proceedings of the National Academy of Sciences 73: 3656–3660
Lloyd DG (1975) The maintenance of gynodioecy and androdioecy in angiosperms. Genetica 45: 325–339
Lloyd DG, Bawa KS (1984) Modification of the gender of seed plants in varying conditions. Evol Biol 17: 255–338
Lovett-Doust J, Cavers PB (1982) Sex and gender dynamics in jack-in-the-pulpit,Arisaema triphyllum (Araceae). Ecology 63: 797–808
Philbrick CT (1983) Contributions to the reproductive biology ofPanax trifolium L. (Araliaceae). Rhodora 84: 97–114
Policansky D (1982) Sex change in plants and animals. Ann Rev Ecol Syst 13: 471–495
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43: 223–225
Schaffner JH (1922) Control of the sexual state inArisaema triphyllum andArisaema dracontium. Am J Bot 9: 72–78
Schlessman MA (1986) Interpretation of evidence for gender choice in plants. Am Natur 128: 416–420
Schlessman MA (1987) Gender modification in North American ginsengs. BioScience 37: 469–475
Schlessman MA (1988) Gender diphasy (“sex choice”). In: Lovett-Doust J, Lovett-Doust L (eds) Plant Reproductive Ecology: Patterns and Strategies. Oxford University Press, New York, pp 139–153
Schlessman MA (1990) Phenotypic gender in sex-changing dwarf ginseng,Panax trifolium (Araliaceae). Am J Bot 77: 1125–1131
Sokal RR, Rohlf FJ (1981) Biometry. 2nd ed. W. H. Freeman and Co., New York
Zar JH (1974) Biostatistical analysis. Prentice-Hall, Inc. Englewood Cliffs
Zimmerman JK (1989) Evolutionary ecology ofCatasetum viridiflavum, an orchid that changes sex. Ph.D. dissertation, University of Utah, Salt Lake City
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schlessman, M.A. Size, gender, and sex change in dwarf ginseng,Panax trifolium (Araliaceae). Oecologia 87, 588–595 (1991). https://doi.org/10.1007/BF00320425
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00320425