Abstract
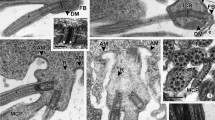
The meiotic spindle of spermatocytes of two wolf spiders contains a highly organized system of ER-like membranes. In cells observed ultrastructurally at early prometaphase, these membranes completely invest each bivalent and are present in the periphery of the spindle in association with the centrosomes. By metaphase each bivalent and its kinetochore fibers are completely encased in a tube of this membrane. We have treated living spermatocytes with the permeant, fiuorescent-chelate probe, chlorotetracycline (CTC) to determine whether or not the intraspindle membrane system is rich in associated Ca2+. Spider testes were dissected into PIPES-buffered saline containing 200 μM CTC and were kept in this solution for 10 min. Autofluorescence controls were prepared by incubation in saline without CTC, and nonspecific effects of CTC were assessed by incubation for 10 min in 200 μM oxytetracycline (OTC). Neither unstained nor OTC-treated spermatocytes emit significant fluorescence. In contrast, CTC treatment yields bright, punctate fluorescence, which coincides with the distribution of the mitochondria. The plasma membrane is only weakly fluorescent, while the nuclear envelope exhibits prominent fluorescence. The chromosomes are not fluorescent during prophase, but after nuclear envelope breakdown, they become outlined by dim, but distinct fluorescence. As spindle formation commences, the CTC signal from the intraspindle membrane system becomes strong. In some cells, thin lines of CTC fluorescence are apparent in the metaphase half spindle; this fluorescence pattern mimics the distribution of the intraspindle membrane system and suggests that it is rich in associated Ca2+. We suggest that the intraspindle membrane system functions in the regulation of cytosolic Ca2+during meiosis through sequestration of the cation.
Similar content being viewed by others
References
Blinks JR, Wier WG, Hess P, Prendergast FG (1982) Measurement of Ca2+ concentrations in living cells. Prog Biophys Molec Biol 40:1–114
Caswell AH (1979) Methods of measuring intracellular calcium. Int Rev Cytol 56:145–181
Chandler DE, Williams JA (1978) Intracellular divalent cation release in pancreatic acinar cells during stimulus-secretion coupling. I. Use of chlorotetracycline as fluorescent probe. J Cell Biol 76:371–385
Fabiato A, Fabiato F (1979) Use of chlorotetracycline fluorescence to demonstrate Ca2+-induced release of Ca2+from the sarcoplasmic reticulum of skinned cardiac cells. Nature 281:146–148
Forer A (1982) Crane fly spermatocytes and spermatids: a system for studing cytoskeletal components In: Wilson L (ed). Methods in cell biology. Academic Press, New York, pp 227–252
Forer A, Gupta L, Hall TA (1980) Electron probe X-ray micro analysis of calcium and other elements in meiotic spindles, in frozen sections of spermatocytes from crane fly testes. Exp Cell Res 126:217–226
Harris P (1978) Triggers, trigger waves, and mitosis: a new model. In: Buetow DE, Camerson IL, Padilla GM (ed). Monographs on cell biology. Academic Press, Inc., New York 75–104
Hepler, PK (1977) Membranes in the spindle apparatus: their possible role in the control of microtubule assembly. In: Rost TL, Gifford EM Jr (ed). Mechanisms and control of cell division. Dowden, Hutchinson and Ross, Inc., Stroudsburg, PA 212–232
Hepler PK (1980) Membranes in the mitotic apparatus of barley cells J Cell Biol 86:490–499
Hepler PK (1983) Restricting calcium prolongs metaphase in dividing stamen hairs of Tradescantia. J Cell Biol 97:40a
Hepler PK, Wick SM, Wolniak SM (1981) The structure and role of membranes in the mitotic apparatus. In: Schweiger, HG (ed). International Cell Biology 1980–1981 Springer, Berlin pp 673–686
Izant JG (1983) The role of calcium ions during mitosis. Calcium participates in the anaphase trigger. Chromosoma 88:1–10
Jackson WT, Doyle BG (1982) Membrane distribution in dividing endosperm cells of Haemanthus. J Cell Biol 94:637–643
Kiehart DP (1981) Studies on the in vivo sensitivity of spindle microtubules to calcium ions and evidence for a vesicular calcium-sequestering system. J Cell Biol 88:605–617
Paweletz N (1981) Membranes in the mitotic apparatus. Cell Biol Int Rep 5:323–336
Petzelt C (1979) Biochemistry of the mitotic spindle. Int Rev Cytol 60:53–92
Salmon ED, Segall RR (1980) Calcium-labile mitotic spindles isolated from sea urchin eggs (Lytechinus variegatus). J Cell Biol 86:355–365
Schatten G, Schatten H, Simerly C (1982) Detection of sequestered calcium during mitosis in mammalian cell cultures and in mitotic apparatus isolated from sea urchin zygotes. Cell Biol Int Rep 6:714–724
Silver RB, Cole RD, Cande WZ (1980) Isolation of mitotic apparatus containing vesicles with calcium sequestration activity. Cell 19:505–516
Sisken JE (1980) The significance and regulation of calcium during mitotic events. In: Nuclear-cytoplasmic interactions in cell cycle: Academic Press, N.Y.
Wick SM, Hepler PK (1980) Localization of Ca++-containing antimonate precipitates during mitosis. J Cell Biol 86:500–513
Wise, DA (1982) The ultrastructure of an intraspindle membrane system in spider spermatocytes. J Cell Biol 95:302a
Wolniak SM, Hepler PK, Jackson WT (1980) Detection of the membrane-calcium distribution during mitosis in Haemanthus endosperm with chlorotetracycline. J Cell Biol 87:27–32
Wolniak SM, Hepler PK, Jackson WT (1981) The coincident distribution of calcium-rich membranes and kinetochore fibers at metaphase in living endosperm cells of Haemanthus. Eur J Cell Biol 25:171–174
Wolniak SM, Hepler PK, Jackson WT (1983) Ionic changes in the mitotic apparatus at the metaphase/anaphase transition. J Cell Biol 96:598–605
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wise, D., Wolniak, S.M. A calcium-rich intraspindle membrane system in spermatocytes of wolf spiders. Chromosoma 90, 156–161 (1984). https://doi.org/10.1007/BF00292453
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00292453