Abstract
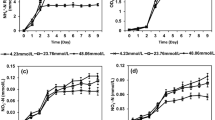
The marine green sulfur bacterium Chlorobium vibrioforme strain 1930 produced H2 and elemental sulfur from sulfide or thiosulfate under N limitation in the light. H2 production depended on nitrogenase and occurred only in the absence of ammonia. Methionine sulfoximine, an inhibitor of glutamine synthetase, prevented the switch-off by ammonia. In defined syntrophic cocultures of the acetate-oxidizing, sulfur-reducing bacterium Desulfuromonas acetoxidans with green sulfur bacteria, H2 was produced from acetate via a light-driven sulfur cycle. The sulfur-reducing bacterium could not be replaced by sulfate-reducing bacteria in these experiments. In a coculture of the marine Chlorobium vibrioforme strain 1930 and the sulfur-reducing bacterium Desulfuromonas acetoxidans strain 5071, optimum long-term H2 production from acetate was obtained with molecular nitrogen as N source, at low light intensity (110 μmol · m-2 · s-1), in sulfide-reduced mineral medium (2 mM Na2S) at pH 6.8. Traces of sulfide (10 μM) were sufficient to keep the sulfur cycle running. The coculture formed no poly-β-hydroxyalkanoates (PHA), but 20%–40% polysaccharide per cell dry mass. Per mol acetate added, the coculture formed 3.1 mol of H2 (78% of the theoretical maximum). Only 8% of the reducing equivalents was incorporated into biomass. The maximum rate of H2 production was 1300 ml H2 per day and g cell dry mass.
Similar content being viewed by others
Abbreviations
- MOPS:
-
2-(N-morpholino) propane sulfonic acid
- MSX:
-
Methionine sulfoximine
- PHA:
-
poly-β-hydroxyalkanoates
References
Biebl H, Pfennig N (1978) Growth yields of green sulfur bacteria in mixed cultures with sulfur and sulfate reducing bacteria. Arch Microbiol 117: 9–16
Burris R H (1991) Nitrogenases. J Biol Chem 266: 9339–9342
Chadwick L J, Irgens R L (1991) Hydrogen gas production by an Ectothiorhodospira vacuolata strain. Appl Environ Microbiol 57: 594–596
Chaney A L, Marbach E P (1962) Modified reagents for the determination of urea and ammonia. Clin Chem 8: 130–132
Cypionka H, Pfennig N (1986) Growth yields of Desulfotomaculum orientis with hydrogen in chemostat culture. Arch Microbiol 143: 296–399
Gest H, Ormerod J G, Ormerod K S (1962) Photometabolism of Rhodospirillum rubrum: light-dependent dissimilation of organic compounds to carbon dioxide and molecular hydrogen by an anaerobic citric acid cycle. Arch Biochem Biophys 97: 21–33
Gotto J W, Yoch D C (1985) Regulation of nitrogenase activity by covalent modification in Chromatium vinosum. Arch Microbiol 141: 40–43
Gray B H, Fowler C F, Nugent N A, Rigopoulos N, Fuller R C (1973) Reevaluation of Chloropseudomonas ethylica strain 2-K. Int J Syst Bacteriol 23: 256–264
Hansen T A, Veldkamp H (1973) Rhodopseudomonas sulfidophila, nov. spec., a new species of the purple nonsulfur bacteria. Arch Microbiol 92: 45–58
Heda G D, Madigan M T (1986) Aspects of nitrogen fixation in Chlorobium. Arch Microbiol 143: 330–336
Herbert D, Phipps P J, Strange R E (1971) Chemical analysis of microbial cells. In: Norris J R, Ribbons D W (eds) Methods in microbiology, vol. 5B, Academic Press, London
Ivanovskii R N, Fedorova E A (1984) Ammonium assimilation by the green sulfur bacterium Chlorobium limicola forma thiosulfatophilum. Microbiologiya 55: 14–19 (Engl. transl.)
Kanemoto R H, Ludden P W (1987) Amino acid concentration in Rhodospirillum rubrum during expression and switch-off of nitrogenase activity. J Bacteriol 169: 3035–3042
Keppen O I, Lebedeva N V, Pethukov S, Rodionov Yu V (1985) Nitrogenase activity in the green bacterium Chlorobium limicola. Microbiologiya 54: 28–32 (Engl. transl.)
Klein G, Klipp W, Jahn A, Steinborn B, Oelze J (1991) The relationship of biomass, polysaccharide and H2 formation in the wild-type and nifA/nifB mutants of Rhodobacter capsulatus. Arch Microbiol 155: 477–482
Liebergesell M, Hustede E, Timm A, Steinbüchel A, Fuller R C, Lenz R W, Schlegel H G (1991) Formation of poly(3-hyxdroxyalkanoates) by phototrophic and chemolithotrophic bacteria. Arch Microbiol 155: 415–421
Macler B A, Pelroy R A, Bassham J A (1979) Hydrogen formation in nearly stoichiometric amounts from glucose by a Rhodopseudomonas sphaeroides mutant. J Bacteriol 138: 446–452
Matheron R, Baulaigue R (1983) Photoproduction d'hydrogène sur soufre et sulfure par des Chromatiaceae. Arch Microbiol 135: 211–214
Merric J M (1978) Metabolism of reserve material. In: Clayton R K, Sistrom W R (eds) The photosynthetic bacteria. Plenum Press, New York London, pp 199–219
Nesbakken T, Kolsaker P, Ormerod J (1988) Mechanism of biosynthesis of 2-oxo-3-methylvalerate in Chlorobium vibrioforme. J Bacteriol 170: 3287–3290
Nitschke W, Rutherford A W (1991) Photosynthetic reaction centres: variations on a common structural theme? TIBS 16: 241–245
Otha Y, Mitsui A (1981) Enhancement of hydrogen photoproduction by marine Chromatium sp. Miamy PBS 1071 grown in molecular nitrogen. In: Moo-Young M, Robinson C W (eds) Advances in biotechnology, Vol. 2, Pergamon Press, Toronto, pp 303–307
Overmann J, Cypionka H, Pfennig N (1991) An extremely low light adapted phototrophic sulfur bacterium from the Black Sea. Limnol Oceanogr (in press)
Pfennig N (1967) Photosynthetic bacteria. Ann Rev Microbiol 21: 285–324
Pfennig N (1975) The photolithotrophic bacteria and their role in the sulfur cycle. Plant Soil 43: 1–16
Pfennig N (1978) Rhodocyclus purpureus, gen. nov. and sp. nov., a ring-shaped vitamin B12-requiring member of the family Rhodospirillaceae. Int J Syst Bacteriol 28: 283–288
Pfennig N, Biebl H (1976) Desulfuromonas acetoxidans, gen. nov. and sp. nov., a new anaerobic, sulfur-reducing, acetate-oxidizing bacterium. Arch Microbiol 110: 3–12
Schink B, Pfennig N (1982) Fermentation of trihydroxybenzenes by Pelobacter acidigallici gen. nov. sp. nov., a new strictly anaerobic, non-sporeforming bacterium. Arch Microbiol 133: 195–201
Schlegel H G, Lafferty R L, Krauss I (1970) The isolation of mutants not accumulating poly-β-hydroxybutyric acid. Arch Microbiol 71: 283–294
Schmidt K, Liaaen-Jensen S, Schlegel H G (1963) Die Carotinoide der Thiorhodaceae. 1. Okenon als Hauptcarotenoid von Chromatium okenii Perty. Arch Microbiol 46: 117–126
Seitz H J, Schink B, Conrad R (1988) Thermodynamics of hydrogen metabolism in methanogenic cocultures degrading ethanol or lactate. FEMS Microbiol Lett 55: 119–124
Shaposhnikov V V, Kondrat'eva E N, Fedorov V D (1960) A new species of green sulphur bacteria. Nature 187: 167–168
Steinborn B, Oelze J (1989) Nitrogenase and photosynthetic activities of chemostat cultures of Rhodobacter capsulatus 37b4 grown under different illuminations. Arch Microbiol 152: 100–104
Stevens P, Plovie N, DeVos P, DeLey J (1986) Photoproduction of molecular hydrogen by Rhodobacter sulfidophilus. Syst Appl Microbiol 8: 19–23
Stewart W D P, Fitzgerald G P, Burris R H (1968) Acetylene reduction bu nitrogen-fixing blue green algae. Arch Microbiol 62: 336–348
Vignais P M, Colbeau A, Willison J C, Jouanneau Y (1985) Hydrogenase, nitrogenase, and hydrogen metabolism in the photosynthetic bacteria. In: Rose A H, Tempest D W (eds) Advances in microbiol physiology, vol. 26. Academic Press, London, pp 155–234
Vijayaragavan R (1987) Photoproduction of hydrogen by Rhodobacter capsulatus B100. Diploma thesis, Tamil Nadu Agricultural University, Coimbatore, India
Widdel F (1983) Methods for enrichment and pure culture isolation of filamentous gliding sulfate-reducing bacteria. Arch Microbiol 134: 282–285
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Warthmann, R., Cypionka, H. & Pfennig, N. Photoproduction of H2 from acetate by syntrophic cocultures of green sulfur bacteria and sulfur-reducing bacteria. Arch. Microbiol. 157, 343–348 (1992). https://doi.org/10.1007/BF00248679
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00248679