Abstract
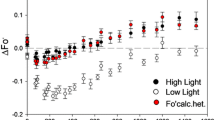
Photoinhibition of PSII and turnover of the D1 reaction-centre protein in vivo were studied in pumpkin leaves (Cucurbita pepo L.) acclimated to different growth irradiances and in low-light-grown moss, (Ceratodon purpureus) (Hedw.) Brid. The low-light-acclimated pumpkins were most susceptible to photoinhibition. The production rate of photoinhibited PSII centres (kPI), determined in the presence of a chloroplast-encoded protein-synthesis inhibitor, showed no marked difference between the high- and low-light-grown pumpkin leaves. On the other hand, the rate constant for the repair cycle (kREC) of PSII was nearly three times higher in the high-light-grown pumpkin when compared to low-light-grown pumpkin. The slower degradation rate of the damaged D1 protein in the low-light-acclimated leaves, determined by pulsechase experiments with [35S]methionine suggested that the degradation of the Dl protein retards the repair cycle of PSII under photoinhibitory light. Slow degradation of the D1 protein in low-light-grown pumpkin was accompanied by accumulation of a phosphorylated form of the D1 protein, which we postulate as being involved in the regulation of D1-protein degradation and therefore the whole PSII repair cycle. In spite of low growth irradiance the repair cycle of PSII in the moss Ceratodon was rapid under high irradiance. When compared to the high- or low-light-acclimated pumpkin leaves, Ceratodon had the highest rate of D1-protein degradation at 1000 μmol photons m−2 s−1. In contrast to the higher plants, the D1 protein of Ceratodon was not phosphorylated either under high irradiance in vivo or under in-vitro conditions, which readily phosphorylate the D1 protein of higher plants. This is consistent with the rapid degradation of the D1 protein in Ceratodon. Screening experiments indicated that D1 protein can be phosphorylated in the thylakoid membranes of angiosperms and conifers but not in lower plants. The postulated regulation mechanism of D1-protein degradation involving phosphorylation and the role of thylakoid organization in the function of PSII repair cycle are discussed.
Similar content being viewed by others
Abbreviations
- Chl:
-
Chlorophyll
- D1* :
-
phosphorylated form of D1 protein
- Fmax and Fv :
-
maximal and variable fluorescence respectively
- kPJ and kREC :
-
rate constants of photoinhibition and concurrent recovery respectively
- LHCII:
-
lightharvesting chlorophyll a/bprotein of PSII
- PFD:
-
photon flux density
References
Allen, J.F. (1992) How does protein phosphorylation regulate photosynthesis? Trends Biochem. Sci. 17, 12–17
Aro, E.-M. (1982) A comparison of the chlorophyll-protein composition and chloroplast ultrastructure in two bryophytes and two higher plants. Z. Pflanzenphysiol. 108, 97–105
Aro, E.-M., Kettunen, R., Tyystjärvi, E. (1992) ATP and light regulate D1 protein modification and degradation. Role of D1* in photoinhibition. FEBS Lett. 297, 29–33
Aro, E.-M., McCaffery, S., Anderson, J. (1993a) Photoinhibition and D1 protein degradation in peas acclimated to different growth irradiances. Plant Physiol. 103, 835–843
Aro, E.-M., Virgin, L, Andersson, B. (1993b) Photoinhibition of Photosystem II. Inactivation, protein damage and turnover. Biochim. Biophys. Acta 1143, 113–134
Aro, E-M., Rintamäki, E., Korhonen, P., Mäenpää, P. (1986) Relationship between chloroplast structure and O2 evolution rate of leaf discs in plants from different biotopes in South Finland. Plant Cell Environ. 9, 87–94.
Barbato, R., Shipton, A., Giacometti, G.M., Barber, J. (1991) New evidence suggests that the initial photoinduced cleavage of the D1 protein may not occur near the PEST sequence. FEBS Lett. 290, 162–166
Bennett, J. (1991) Protein phosphorylation in green plant chloroplasts. Annu. Rev. Plant Physiol. Plant Mol. Biol. 42, 281–311
Clarke, A.K., Soitamo, A., Gustafsson, P., Öquist, G. (1993) Rapid interchange between two distinct forms of cyanobacterial D1 photosystem II reaction-center protein in response to photoinhibition. Proc. Natl. Acad. Sci. USA 90, 9973–9977
Cleland, R.E., Melis, A., Neale, P.J. (1986) Mechanism of photoinhibition: photochemical reaction center inactivation in system II of chloroplasts. Photosynth. Res. 9, 79–88
Demmig, B., Björkman, O. (1987) Comparison of the effect of excessive light on chlorophyll fluorescence (77 K) and photon yield of O2 evolution in leaves of higher plants. Planta 171, 171–184
Demmig-Adams, B., Adams III, W.W. (1992) Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol. 43, 599–626
de Vitry, C., Diner, B.A., Popot, J.-L. (1991) Photosystem II particles from Chlamydomonas reinhardtii. Purification, molecular weight, small subunit composition, and protein phosphorylation. J. Biol. Chem. 266, 16614–16621
Elich, T.D., Edelman, M., Mattoo, A.K. (1992) Identification, characterization, and resolution of the in vivo phosphorylated form of the D1 photosystem II reaction center protein. J. Biol. Chem. 267, 3523–3529
Giardi, M.T., Rigoni, F., Barbato, R. (1992) Photosystem II core phosphorylation heterogeneity, differential herbicide binding, and regulation of electron transfer in photosystem II preparations from spinach. Plant Physiol. 100, 1948–1954
Godde, D., Schmitz, H., Weidner, M. (1991) Turnover of the D-1 reaction centre polypeptide from photosystem II in intact spruce needles and spinach leaves. Z. Naturforsch. 46c, 245–251
Gong, H., Ohad, I. (1991) The PQ/PQH2 ratio and occupancy of photosystem II-QB site by plastoquinone control the degradation of D1 protein during photoinhibition in vivo. J. Biol. Chem. 286, 21293–21299
Greenberg, B.M., Gaba, V., Mattoo, A.K., Edelman, M. (1987) Identification of a primary in vivo degradation product of the rapidly-turning-over 32 kd protein of photosystem II. EMBO J. 6, 2865–2869
Greer, D.H., Berry, J.A., Björkman, O. (1986) Photoinhibition of photosynthesis in intact bean leaves: Role of light and temperature, and requirement for chloroplast-protein synthesis during recovery. Planta 168, 253–260
Guenther, J.E., Melis, A. (1990) The physiological significance of photosystem II heterogeneity in chloroplasts. Photosynth. Res. 23, 105–109
Herrmann, G., Thiel, A., Böger, P. (1985) Antiserum against 33-kDa herbicide-binding protein. Z. Naturforsch. 40c, 814–818
Jansen, M.A.K., Depka, B., Trebst, A., Edelman, M. (1993) Engagement of specific sites in the plastoquinone niche regulates degradation of the D1 protein in photosystem II. J. Biol. Chem. 268, 21246–21252
Kanervo, E., Mäenpää, P., Aro, E.-M. (1993) D1 protein degradation and psbA transcript levels in Synechocystis PCC 6803 during photoinhibition in vivo. J. Plant Physiol. 142, 669–675
Kettunen, R., Lehtonen, E., Tyystjärvi, E., Aro, E-M. (1992) Degradation products of the D1 protein are located in the non-appressed regions of the thylakoid membrane in vivo. In: Research in photosynthesis, vol. IV, pp. 309–316, Murata, N., ed. Kluwer Acad. Publ., Dordrecht
Kettunen, R., Tyystjärvi, E., Aro, E.-M. (1991) D1 protein degradation during photoinhibition of intact leaves. A modification of the D1 protein precedes degradation. FEBS Lett. 290, 153–156
Kim, J.H., Nemson, J.A., Melis, A. (1993) Photosystem II reaction center damage and repair in Dunaliella salina (green alga). Plant Physiol. 103, 181–189
Krause, G.H., Weis, E. (1991) Chlorophyll fluorescence and photosynthesis: The basics. Annu. Rev. Plant Physiol. Plant Mol. Biol. 42, 313–349
Kyle, D.J., Ohad, I., Arntzen, C.J. (1984) Membrane protein damage and repair: Selective loss of a quinone-protein function in chloroplast membranes. Proc. Natl. Acad. Sci. USA 81, 4070–4074
Mattoo, A.K., Edelman, M. (1987) Intramembrane translocation and posttranslational palmitoylation of the chloroplast 32-kDa herbicide-binding protein. Proc. Natl. Acad. Sci. USA 84, 1497–1501
Michel, H., Hunt, D.F., Shabanowitz, J., Bennett, J. (1988) Tandem mass spectrometry reveals that three photosystem II proteins of spinach chloroplasts contain N-acetyl-O-phosphothreonine at their NH2 termini. J. Biol. Chem. 263, 1123–1130
Mullet, J.E., Klein, P.G., Klein, R.R. (1990) Chlorophyll regulates accumulation of the plastid-encoded chlorophyll apoproteins CP43 and D1 by increasing apoprotein stability. Proc. Natl. Acad. Sci. USA 87, 4038–4042
Ohad, I., Kyle, D.J., Hirschberg, J. (1985) Light-dependent degradation of the QB-protein in isolated pea thylakoids. EMBO J. 4, 1655–1659
Oquist, G., Anderson, J.M., McCaffery, S., Chow, W.S. (1992) Mechanistic differences in photoinhibition of sun and shade plants. Planta 188, 422–431
Porra, R.J., Thompson, W.A., Kriedemann, P.E. (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophyll a and b with four different solvents: verification of the concentration of chlorophyll by atomic absorption spectroscopy. Biochim. Biophys. Acta 975, 384–394
Powles, S.B. (1984) Photoinhibition of photosynthesis induced by visible light. Annu. Rev. Plant Physiol. 35, 15–44
Prasil, O., Adir, N., Ohad, I. (1992a) Dynamics of photosystem II:Mechanism of photoinhibition and recovery processes. In: Topics in photosynthesis, vol. 11, pp. 293–348, Barber, J., ed. Elsevier, Amsterdam
Prasil, O., Zer, H., Godde, D., Ohad, I. (1992b) Role of the PSII acceptor site in the light induced degradation of D1. In: Research in photosynthesis, vol. IV, pp. 501–504, Murata, N., ed. Kluwer Acad. Publ., Dordrecht
Rintamäki, E., Salo, R., Aro, E.-M. (1994) Rapid turnover of the D1 reaction-center protein of photosystem II as a protection mechanism against photoinhibition in a moss, Ceratodon purpureus (Hedw.) Brid. Planta 193, 520–529
Samuelsson, G., Lönneborg, A., Gustafsson, P., Öquist, G. (1987) The susceptibility of photosynthesis to photoinhibition and the capacity of recovery in high and low light-grown cyanobacteria, Anacystis nidulans. Plant Physiol. 83, 438–441
Schnettger, B., Critchley, C., Santore, U.J., Graf, M., Krause, G.H. (1994) Relationship between photoinhibition of photosynthesis, D1 protein turnover and chloroplast structure. Effects of protein synthesis inhibitors. Plant Cell Environ. 17, 55–64
Svensson, B., Vass, I., Styring, S. (1991) Sequence analysis of the D1 and D2 reaction center proteins of photosystem II. Z. Naturforsch. 46c, 765–776
Syme, A.J., Bolhar-Nordenkampf, H.R., Critchley, C. (1993) The role of D1* in light-induced D1 protein turnover in leaves. Z. Naturforsch. 48c, 246–250
Trebst, A., Draber, W. (1986) Inhibitors of photosystem II and the topology of the herbicide and QB binding polypeptide in the thylakoid membrane. Photosynth. Res. 10, 381–392
Trebst, A., Depka, B., Kraft, B., Johanningmeier, U. (1988) The QB site modulates the conformation of the photosystem II reaction center polypeptides. Photosynth. Res. 18, 163–177
Tyystjärvi, E., Ali-Yrkkö, K., Kettunen, R., Aro, E.-M. (1992) Slow degradation of the D1 protein is related to the susceptibility of low-light-grown pumpkin plants to photoinhibition. Plant Physiol. 100, 1310–1317
Author information
Authors and Affiliations
Additional information
Dr. R. Barbato (Dipartimento di Biologia, Universita di Padova, Padova, Italy), Prof. P. Böger (Lehrstuhl fur Physiologie und Biochemie der Pflanzen, Universität Konstanz, Konstanz, Germany), Prof. A. Melis (Department of Plant Biology, University of California, Berkeley, USA), Prof. I. Ohad (Department of Biological Chemistry, Hebrew University, Jerusalem, Israel) and Mr. A. Soitamo (Department of Biology, University of Turku, Turku, Finland) are gratefully acknowledged for the D1-protein-specific antibodies. The authors thank Ms. Virpi Paakkarinen for excellent technical assistance. This work was supported by the Academy of Finland and the Foundation of the University of Turku.
Rights and permissions
About this article
Cite this article
Rintamäki, E., Salo, R., Lehtonen, E. et al. Regulation of D1-protein degradation during photoinhibition of photosystem II in vivo: Phosphorylation of the D1 protein in various plant groups. Planta 195, 379–386 (1995). https://doi.org/10.1007/BF00202595
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202595