Summary
-
1.
The close association of muscle and neurons in Ascaris suum makes it difficult to determine whether spikes recorded from nerve cords originate in muscle or neurons. We have developed criteria that distinguish muscle and neuronal activity. There are two categories of extracellular spikes.
-
2.
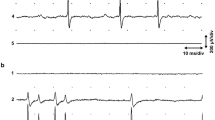
The first category consists of spikes with a wide range of amplitudes, marked by large spikes. These spikes, which can be recorded over lateral muscle and over the dorsal and ventral nerve cords, are abolished when muscle is disrupted or removed, or when curare is applied. Large spikes are relatively infrequent, are correlated with intracellularly recorded muscle events, and respond to polarizations of motor neurons, implying that they originate in muscle.
-
3.
The second spike category, small amplitude spikes, is exclusive to the ventral nerve cord, occurs more frequently than large spikes and displays patterned firing. Small spikes are not affected by muscle removal or by curare, and are correlated with motor neuronal postsynaptic potentials, but not with intracellularly recorded muscle events. We infer that they originate in neurons.
-
4.
Low level activity recorded extracellularly over nerve cords may represent muscle activity due to tonic motor neuronal synaptic transmission. It responds to motor neuronal polarization and is suppressed by curare or muscle removal.
Similar content being viewed by others
Abbreviations
- VNC :
-
ventral nerve cord
- DNC :
-
dorsal nerve cord
- MOPS :
-
3-(N-Morpholino)propane-sulfonic acid
- RVG :
-
retrovesicular ganglion
- DE1 :
-
dorsal excitatory type 1 motor neuron
- DE2 :
-
dorsal excitatory type 2 motor neuron
- DI :
-
dorsal inhibitory motor neuron
- VI :
-
ventral inhibitory motor neuron
- SE :
-
suction electrode
- d-TC :
-
d-tubocurarine
- TTX :
-
tetrodotoxin
References
Anderson PAV, Schwab WE (1984) An epithelial cell-free preparation of the motor nerve net of Cyanea (Coelenterata). Biol Bull 166:396–408
Anderson PAV, Spencer AN (1989) The importance of cnidarian synapses for neurobiology. J Neurobiol 20:435–457
Angstadt JD, Stretton AOW (1989) Slow active potentials in ventral inhibitory motor neurons of the nematode Ascaris. J Comp Physiol A 166:165–177
Angstadt JD, Donmoyer JE, Stretton AOW (1989) The retrovesicular ganglion of Ascaris. J Comp Neurol 284:374–388
deBell JT, del Castillo J, Sanchez V (1963) Electrophysiology of the somatic muscle cells of Ascaris lumbricoides. J Cell Comp Physiol 62:159–178
Davis RE (1984) Membrane properties and synaptic interactions of motor neurons in the nematode Ascaris. Ph.D. Thesis, University of Wisconsin, Madison
Davis RE, Stretton AOW (1989a) Passive membrane properties of motor neurons and their role in long distance signaling in the nematode Ascaris. J Neurosci 9:403–414
Davis RE, Stretton AOW (1989b) Signaling properties of Ascaris motor neurons: graded active responses, graded synaptic transmission, and tonic transmitter release. J Neurosci 9:415–425
Delcomyn F (1974) A simple system for suction electrodes. J Electrophysiol Tech 3:22–25
Delcomyn F (1980) Neural basis of rhythmic behavior in animals. Science 210:492–498
Dickinson P (1989) Modulation of simple motor patterns. Seminars in Neurosci 1:15–24
Friesen WO (1989) Neuronal control of leech swimming movements. In: Jacklet JW (ed) Neuronal and cellular oscillators. Marcel Dekker, New York, 269–316
Getting PA (1989) A network oscillator underlying swimming in Tritonia. In: Jacklet JW (ed) Neuronal and cellular oscillators. Marcel Dekker, New York, pp 215–236
Hesse R (1892) Über das Nervensystem von Ascaris megalocephala. Z Wiss Zool 54: 548–568
Hoyle G (1983) Muscles and their neural control. John Wiley and Sons, New York, p 158
Jarman M, Ellory JC (1969) Effect of tetrodotoxin on Ascaris somatic muscle. Experientia 25/5:507
Kass IS, Stretton AOW, Wang CC (1984) The effects of avermectin and drugs related to acetylcholine and 4-aminobutyric acid on neurotransmission in Ascaris suum. Mol Biochem Parasitol 13:213–225
Keenan L, Koopowitz H (1982) Physiology and in situ identification of putative aminergic neurotransmitters in the nervous system of Gyrocotyle fimbriata, a parasitic flatworm. J Neurobiol 13:9–21
Keenan L, Koopowitz H, Solon MH (1984) Primitive nervous systems: electrical activity in the nerve cords of the parasitic flatworm, Gyrocotyle fimbriata. J Parasitol 70:131–138
Koopowitz H (1989) Polyclad neurobiology and the evolution of central nervous systems. In: Anderson PAV (ed) Evolution of the first nervous systems. Plenum Press, New York, 315–328
Koopowitz H, Keenan L (1982) The primitive brains of platyhelminths. Trends Neurosci 5:77–79
Mackie GO (1984) Fast pathways and escape behavior in Cnidaria. In: Eaton RC (ed) Neural mechanisms of startle behavior. Plenum Press, New York, pp 15–42
Meade JA (1991) Intracellular recordings from neurons and muscle cells in a semi-intact preparation of the nematode Ascaris suum: implications for Ascaris locomotion. Thesis, University of Wisconsin, Madison
Meech RW (1989) The electrophysiology of swimming in jellyfish Aglantha digitale. In: Anderson PAV (ed) Evolution of the first nervous systems. Plenum Press, New York, pp 281–298
Pax RA, Bennett JL (1991) Neurobiology of parasitic platyhelminths: possible solutions to the problems of correlating structure with function. Parasitology 102:S31–39
Robertson RM, Pearson KG (1985) Neural networks controlling locomotion in locusts. In: Selverston AI (ed) Model neural networks and behavior. Plenum Press, New York, pp 21–35
Rosenbluth J (1965) Ultrastructure of somatic muscle cells in Ascaris lumbricoides. J Cell Biol 26:579–591
Satterlie RA (1989) Reciprocal inhibition and rhythmicity: swimming in a pteropod mollusk. In: Jacklet JW (ed) Neuronal and cellular oscillators. Marcel Dekker, New York, pp 151–171
Stretton AOW (1976) Anatomy and development of the somatic musculature of the nematode Ascaris. J Exp Biol 64:773–788
Stretton AOW, Fishpool RM, Southgate E, Donmoyer JE, Walrond JP, Moses JER, Kass IS (1978) Structure and physiological activity of the motor neurons of the nematode Ascaris. Proc Natl Acad Sci USA 75:3493–3497
Stretton AOW, Davis RE, Angstadt JD, Donmoyer JE, Johnson CD (1985) Neural control of behaviour in Ascaris. Trends in Neurosci 8:294–300
Walrond JP, Stretton AOW (1985a) Reciprocal inhibition in the motor nervous system of the nematode Ascaris: direct control of inhibitory motoneurons by dorsal excitatory motoneurons. J Neurosci 5:9–15
Walrond JP, Stretton AOW (1985b) Excitatory and inhibitory activity in the dorsal musculature of the nematode Ascaris evoked by single dorsal excitatory motoneurons. J Neurosci 5:16–22
Walrond JP, Kass IS, Stretton AOW, Donmoyer JE (1985) Identification of excitatory and inhibitory motoneurons in the nematode Ascaris by electrophysiological techniques. J Neurosci 5:1–8
Weisblat DA, Byerly L, Russell RL (1976) Ionic mechanisms of electrical activity in somatic muscle of the nematode Ascaris lumbricoides. J Comp Physiol 111:93–113
Willmer P (1990) Invertebrate relationships: patterns in animal evolution. Cambridge University Press, Cambridge New York, pp 152–153
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davis, R.E., Stretton, A.O.W. Extracellular recordings from the motor nervous system of the nematode, Ascaris suum . J Comp Physiol A 171, 17–28 (1992). https://doi.org/10.1007/BF00195957
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00195957