Abstract
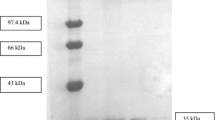
A thermophilic Bacillus stearothermophilus F1 that produced an extremely thermostable alkaline protease was isolated from decomposed oil palm branches. The isolated protease was purified to homogeneity by heat treatment, ultrafiltration and gel filtration chromatography with a 128-fold increase in specific activity and 75% recovery. The protease, which is a serine-type enzyme, has a relative molecular mass of 33 500 by sodium dodecyl sulphate-polyacrylamide gel electrophoresis but only 20 000 by gel-filtration chromatography. The enzyme was optimally active at pH 9.0 and was stable for 24 h at 70° C and in the pH range from 8.0 to 10.0. It was capable of hydrolysing many soluble and insoluble protein substrates but no esterase activity was detected. The enzyme activity was markedly inhibited by Co2+ and Hg2+, whereas Mg2+, Fe2+, Cu2+, Zn2+ and Sr2+ had little or no inhibitory effect. However, Mn2+ strongly activated the protease activity. The protease exhibited a high degree of thermostability [t 1/2 (85° C) = 4 h, (90° C) = 25 min]. The stability at higher temperatures (85° C and above) was shown to be dependent on the presence of Ca2+.
Similar content being viewed by others
References
Arvidson S, Wadstrom T (1973) Detection of proteolytic activity after isoelectric focusing on polyacrylamide gel. Biochim Biophys Acta 310:418–420
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of protein-dye binding. Anal Biochem 72:248–254
Brock FM, Frosberg CW, Buchanan-Smith JG (1982) Proteolytic activity of rumen microorganisms and effect of proteinase inhibitors. Appl Environ Microbiol 44:561–569
Cowan DA, Daniel RM (1982) Purification and some properties of an extracellular protease (caldolysin) from an extreme thermophile. Biochim Biophys Acta 705:239–305
Cowan DA, Smolenski KA, Daniel RM, Morgan HW (1987) An extremely thermostable extracellular proteinase from a strain of the archaebacterium Desulfurococcus growing at 88° C. Biochem J 247:121–123
Durham DR, Steward DB, Stellwag EJ (1987) Novel alkaline and heat-stable serine protease from alkalophilic Bacillus sp. strain GX6638. J Bacteriol 169:2762–2768
Kalebina TS, Rudenskaya GN, Selyalch IO, Khodova OM, Chestukhina GG, Stepanov VM, Kulaev IS (1988) Serine proteinases from Bacillus brevis: lytic action on intact yeast cells. Appl Microbiol Biotechnol 28:531–536
Gusek TW, Kinsella JE (1987) Purification and characterization of heat-stable serine proteinase from Thermomonospora fusca YX. Biochem J 246:511–517
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 277:680–685
Manachini PL, Fortina MG, Parini C (1988) Thermostable alkaline protease produced by Bacillus thermoruber — a new species of Bacillus. Appl Microbiol Biotechnol 28:409–413
Meloun B, Baudys M, Kostka V, Hausdorf G, Frommel C, Hohne WE (1985) Complete primary structure of thermitase from Thermoactinomyces vulgaris and its structural features related to subtilisin-type proteinases. FEBS Lett 183:195–200
Morihara K (1974) Comparative specificity of microbial proteinases. Adv Enzymol 14:179–243
North MJ (1982) Comparative biochemistry of the proteinases of eukaryotic microorganisms. Microbiol Rev 46:308–340
Ohta Y, Ogura Y, Wada A (1966) Thermostable protease from thermophilic bacteria. J Biol Chem 241:5919–5925
Peek K, Daniel RM, Monk C, Parker L, Coolbear T (1992) Purification and characterization of a thermostable proteinase isolated from Thermus sp. strain Rt 41A. Eur J Biochem 207:1035–1044
Shimogaki H, Takeuchi K, Nishino T, Ohdera M, Kudo T, Ohba K, Iwama M, Irie M (1991) Purification and properties of a novel surface-active agent and alkaline resistant protease from Bacillus sp. Y. Agric Biol Chem 55:2251–2258
Sidler W, Zuber H (1977) The production of extracellular thermostable neutral proteinase and α-amylase by Bacillus stearothermophilus. Eur J Appl Microbiol Biotechnol 4:255–266
Strongin AYA, Tzotova LS, Abramov ZT, Gorodetsky DI, Ermakova LM, Baratova LA, Belyanova LP, Stepanov VM (1978) Intracellular serine protease of Bacillus subtilis: sequence homology with extracellular subtilisin. J Bacteriol 133:1401–1411
Takami H, Akiba T, Horikoshi K (1989) Production of extremely thermostable alkaline protease from Bacillus sp. no. Ah-101. Appl Microbiol Biotechnol 30:120–124
Takii Y, Kuriyama N, Suzuki Y (1990) Alkaline serine protease produced from citric acid by Bacillus alcalophilus subsp. halodurans KP 1239. Appl Microbiol Biotechnol 34:57–62
Taguchi H, Hamaoki M, Matsuzawa H, Ohta T (1983) Heat-stable extacellular proteolytic enzymes produced by Thermus caldophilus strain GK24, an extremely thermophilic bacterium. J Biochem 93:7–13
Author information
Authors and Affiliations
Additional information
Correspondence to: A. B. Salleh
Rights and permissions
About this article
Cite this article
Rahman, R.N.Z.A., Razak, C.N., Ampon, K. et al. Purification and characterization of a heat-stable alkaline protease from Bacillus stearothermophilus F1. Appl Microbiol Biotechnol 40, 822–827 (1994). https://doi.org/10.1007/BF00173982
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00173982