Abstract
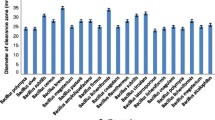
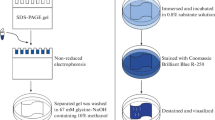
An ammonium sulfate precipitation of fermentation broth produced by Bacillus subtilis FBL-1 resulted in 2.9-fold increase of specific protease activity. An eluted protein fraction from the column chromatographies using DEAE-Cellulose and Sephadex G-75 had 94.2- and 94.9-fold higher specific protease activity, respectively. An SDS-PAGE revealed a band of purified protease at approximately 37.6 kDa. Although purified protease showed the highest activity at 45°C and pH 9.0, the activity remained stable in temperature range from 30 to 50°C and pH range from 7.0 to 9.0. Protease activity was activated by metal ions such as Ca2+, Mg2+, Mn2+, Fe2+, Ca2+ and K+, but 10 mM Fe3+ significantly inhibited enzyme activity (53%). Protease activity was inhibited by 2 mM EDTA as a metalloprotease inhibitor, but it showed good stability against surfactants and organic solvents. The preferred substrates for protease activity were found to be casein (100%) and soybean flour (71.6%).
Similar content being viewed by others
References
Hartley, B. S. (1960) Proteolytic enzymes. Annu. Rev. Biochem. 29: 45–72.
Liao, C. H. and D. E. McCallus (1998) Biochemical and genetic characterization of an extracellular protease from Pseudomonas fluorescens CY091. Appl. Environ. Microbiol. 64: 914–921.
Rao, M. B., A. M. Tanksale, M. S. Ghatge, and V. V. Deshpande (1998) Molecular and biotechnological aspects of microbial proteases. Microbiol. Mol. Biol. Rev. 62: 597–635.
Deng, A., J. Wu, Y. Zhang, G. Zhang, and T. Wen (2010) Purification and characterization of a surfactant-stable highalkaline protease from Bacillus sp. B001. Bioresour. Technol. 101: 7111–7117.
Godfrey, T. and J. Reichelt (1984) Industrial enzymology: The application of enzymes in industry. The Nature Press, NY, USA.
Li, Q., L. Yi, P. Marek, and B. L. Iverson (2013) Commercial proteases: Present and future. FEBS Lett. 587: 1155–1163.
Kim, M., J. B. Si, and Y. J. Wee (2016) Identification of a newly isolated protease-producing bacterium, Bacillus subtilis FBL-1, from soil. Microbiol. Biotechnol. Lett. 44: 185–193.
Kim, M., J. B. Si, L. V. Reddy, and Y. J. Wee (2016) Enhanced production of extracellular proteolytic enzyme excreted by a newly isolated Bacillus subtilis FBL-1 through combined utilization of statistical design and response surface methodology. RSC Adv. 6: 51270–51278.
Folin, O. and V. Ciocalteu (1927) On tyrosine and tryptophane determinations in proteins. J. Biol. Chem. 73: 627–650.
Smith, P. K., R. I. Krohn, G. T. Hermanson, A. K. Mallia, F. H. Gartner, M. D. Provenzano, E. K. Fujimoto, N. M. Goeke, B. J. Olson, and D. C. Klenk (1985) Measurement of protein using bicinchoninic acid. Anal. Biochem. 150: 76–85.
Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Reddy, L. V. A., Y. J. Wee, and H. W. Ryu (2008) Purification and characterization of an organic solvent and detergent-tolerant novel protease produced by Bacillus sp RKY3. J. Chem. Technol. Biotechnol. 83: 1526–1533.
Yang, J. K., I. L. Shih, Y. M. Tzeng, and S. L. Wang (2000) Production and purification of protease from a Bacillus subtilis that can deproteinize crustacean wastes. Enz. Microb. Technol. 26: 406–413.
Bose, A., V. Chawdhary, H. Keharia, and R. B. Subramanian (2014) Production and characterization of a solvent-tolerant protease from a novel marine isolate Bacillus tequilensis P15. Ann. Microbiol. 64: 343–354.
Manni, L., K. Jellouli, R. Agrebi, A. Bayoudh, and M. Nasri (2008) Biochemical and molecular characterization of a novel calcium-dependent metalloprotease from Bacillus cereus SV1. Proc. Biochem. 43: 522–530.
Shah, K., K. Mody, J. Keshri, and B. Jha (2010) Purification and characterization of a solvent, detergent and oxidizing agent tolerant protease from Bacillus cereus isolated from the Gulf of Khambhat. J. Mol. Catal. B-Enz. 67: 85–91.
McConn, J. D., D. Tsuru, and K. T. Yasunobu (1964) Bacillus subtilis neutral proteinase. I. A zinc enzyme of high specific activity. J. Biol. Chem. 239: 3706–3715.
Haddar, A., R. Agrebi, A. Bougatef, N. Hmidet, A. Sellami-Kamoun, and M. Nasri (2009) Two detergent stable alkaline serine-proteases from Bacillus mojavensis A21: Purification, characterization and potential application as a laundry detergent additive. Bioresour. Technol. 100: 3366–3373.
Smith, C. A., H. S. Toogood, H. M. Baker, R. M. Daniel, and E. N. Baker (1999) Calcium-mediated thermostability in the subtilisin superfamily: The crystal structure of Bacillus Ak.1 protease at 1.8 å resolution. J. Mol. Biol. 294: 1027–1040.
Sellami-Kamoun, A., A. Haddar, H. Ali Nel, B. Ghorbel-Frikha, S. Kanoun, and M. Nasri (2008) Stability of thermostable alkaline protease from Bacillus licheniformis RP1 in commercial solid laundry detergent formulations. Microbiol. Res. 163: 299–306.
Beg, Q. K. and R. Gupta (2003) Purification and characterization of an oxidation-stable, thiol-dependent serine alkaline protease from Bacillus mojavensis. Enz. Microb. Technol. 32: 294–304.
Divakar, K., J. D. A. Priya, and P. Gautam (2010) Purification and characterization of thermostable organic solvent-stable protease from Aeromonas veronii PG01. J. Mol. Catal. B-Enz. 66: 311–318.
Abd Rahman, R. N. Z. R., S. Mahamad, A. B. Salleh, and M. Basri (2007) A new organic solvent tolerant protease from Bacillus pumilus 115b. J. Ind. Microbiol. Biotechnol. 34: 509–517.
Jaouadi, B., B. Abdelmalek, D. Fodil, F. Z. Ferradji, H. Rekik, N. Zaraî, and S. Bejar (2010) Purification and characterization of a thermostable keratinolytic serine alkaline proteinase from Streptomyces sp. Strain AB1 with high stability in organic solvents. Bioresour. Technol. 101: 8361–8369.
Powers, J. C., J. L. Asgian, O. D. Ekici, and K. E. James (2002) Irreversible inhibitors of serine, cysteine, and threonine proteases. Chem. Rev. 102: 4639–4750.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Si, JB., Jang, EJ., Charalampopoulos, D. et al. Purification and Characterization of Microbial Protease Produced Extracellularly from Bacillus subtilis FBL-1. Biotechnol Bioproc E 23, 176–182 (2018). https://doi.org/10.1007/s12257-017-0495-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0495-3