Summary
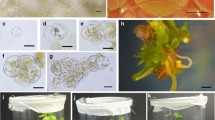
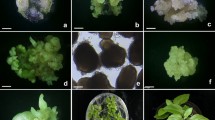
An adventitious shoot regeneration system for Pyrus communis was used to separate two chimeral pears into their component genotypes. The two cultivars were a variegated type, ‘Louise Bonne Panachée’ and a red fruited mutant, ‘Red Hardy’. Leaves of these cultivars were placed onto a regeneration medium consisting of Nitsch & Nitsch (1969) salts supplied with various levels of Thidiazuron (TDZ) and NAA. After two months the regenerants were moved onto a proliferation medium of Lepoivre salts. Later they were evaluated for their chimeral status. Among the regenerants of the variegated type, 100% segregation occurred, most shoots were green, a few were albino. Regeneration was more efficient for dissociating the variegated chimera than rapid shoot multiplication and physical injury. In ‘Red Hardy’, after two months on the regeneration medium, 20 to 33% of the regenerants were green, the rest were red. The stability of the red and the green regenerants were assessed on media supplemented with various levels of sucrose and by total anthocyanins measurement. Both types were stable.
Similar content being viewed by others
References
Broertjes, C. & A.M.Van Harten, 1988. Applied mutation breeding for vegetatively propagated crops. Developments in crop science (12). Elsevier, Amsterdam. pp 29.
Chevreau, E., L. Decourtye & R.M. Skirvin, 1989a. A review of pear chimeras: Their identification and separation into pure types. HortScience 24: 32–34.
Chevreau, E., R.M. Skirvin, H.A. Abu-Qaoud, S.S. Korban & J.G. Sullivan, 1989b. Adventitious shoot regeneration from leaf tissue of three pear (Pyrus sp.) cultivars in vitro. Plant Cell Repts. 7: 688–691.
Cordts, J.M., R. Scorza & R.L. Bell, 1987. Effects of carbohydrates and nitrogen on the development of anthocyanins of a red leaf peach (Prunus persica L. Batsch) in vitro. Plant Cell Tiss. Org. Cult. 9: 104–111.
Daley, L.S., R.A. Mendez & R.L. Stebbins, 1987. Identification of red-fruited pears by fourth derivative spectroscopy of intact lamina. J. Environ. Hort. 5: 25–28.
Dayton, D.F., 1966. The pattern and inheritance of anthocyanin distribution in red pears. Proc. Amer. Soc. Hort. Sci. 89: 110–116.
Decourtye, I., 1973. Hérédité de quelques caractères marqueurs et possibilités offertes par la mutagénèse. Angers 4–8 Sept., 1972. pp. 93–110.
Decourtye, I., 1987. L'origine ontogénique des racines, puis des bourgeons, néoformes sur des chimères de poirier (Pyrus communis 1.) et de pommier (Malus pumila Mill.). Agronomie 7: 27–32.
Dermen, H., 1960. Nature of plant sports. Amer. Hort. Mag. 39: 123–173.
Earle, E.D. & R.W. Langhans, 1974. Propagation of Chrysanthemum in vitro. I. Multiple plantlets from shoot tips and the establishment of tissue culture. J. Amer. Soc. Hort. Sci. 99: 128–132.
Edwin, F.G. & P.D. Sherrington, 1984. Plant propagation by tissue culture. Exegetics Ltd., London, England. pp. 73–87.
Francis, F.T., 1970. Anthocyanin in pears. Hort. Science 5: 42.
Johnson, R.T., 1980: Gamma irradiation and in vitro induced separation of chimeral genotypes in carnation. Hort. Science 15: 605–606.
Jones, J.B. & T. Murashige, 1974. Tissue culture propagation of Aechmea fasciata Baker and other bromeliads. Proc. Intern. Plant Prop. Soc. 24: 117–126.
Marcotrigiano, M., 1986. Origin of adventitious shoots regenerated from cultured tobacco leaf tissue. Amer. J. Bot. 73: 1541–1547.
Marcotrigiano, M., 1990. Genetic mosaics and chimeras: Implications in biotechnology. In: Y.P.S. Bajaj, (Ed.). Biotechnology in agriculture and forestry. Vol. 11. Springer-Verlag, Berlin. (in press)
McPheeters, K. & R.M. Skirvin, 1983. Histogenic layer manipulation in chimeral ‘Thornless Evergreen’ trailing blackberry. Euphytica 32: 351–360.
Nitsch, J.P., Asahira, T., Rossini, M.E. & C. Nitsch, 1970. Bases physiologiques de la production de chair de pomme et de poire in vitro. Bull. Soc. Bot. Fr. 117: 479.
Pierik, R.L.M. & H.H.M. Steegmans, 1983. Vegetative propagation of a chimeral Yucca elephantipes Regel in vitro. Sci. Hortic. 21: 267–272.
Pirie, A. & M.G. Mullins, 1976. Changes in anthocyanin and phenolics content of grapevine leaf and fruit tissue treated with sucrose, nitrate and abscisic acid. Plant Physiol. 58: 468–472.
Quoirin, M. & P. Lepoivre, 1977. Improved medium for in vitro culture of Prunus sp. Acta Hortic. 78: 437–442.
SAS Institute Inc., 1985. SAS user's guide: Statistics. (Ed.). SAS Institute Inc., Cary, NC.
Smith, R.H. & R.E. Norris, 1983. In vitro propagation of African violet chimeras. Hort. Science 18: 436–437.
Tilney-Bassett, R.A.E., 1986. Plant chimeras. 1st ed. Edward Arnold, London. pp. 19–62.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abu-Qaoud, H., Skirvin, R.M. & Chevreau, E. In vitro Separation of Chimeral Pears into their Component Genotypes. Euphytica 48, 189–196 (1990). https://doi.org/10.1007/BF00037199
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00037199