Abstract
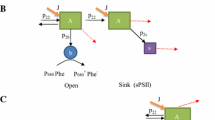
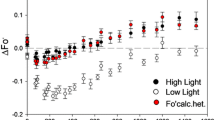
Based on the electron-transport properties on the reducing side of the reaction center, photosystem II (PS II) in green plants and algae occurs in two distinct forms. Centers with efficient electron-transport from QA to plastoquinone (QB-reducing) account for 75% of the total PS II in the thylakoid membrane. Centers that are photochemically competent but unable to transfer electrons from QA to QB (QB-nonreducing) account for the remaining 25% of total PS II and do not participate in plastoquinone reduction. In Dunaliella salina, the pool size of QB-nonreducing centers changes transiently when the light regime is perturbed during cell growth. In cells grown under moderate illumination intensity (500 μE m-2s-1), dark incubation induces an increase (half-time 45 min) in the QB-nonreducing pool size from 25% to 35% of the total PS II. Subsequent illumination of these cells restores the steady-state concentration of QB-nonreducing centers to 25%. In cells grown under low illumination intensity (30 µE m−2s−1), dark incubation elicits no change in the relative concentration of QB-nonreducing centers. However, a transfer of low-light grown cells to moderate light induces a rapid (half-time 10 min) decrease in the QB-nonreducing pool size and a concomitant increase in the QB-reducing pool size. These and other results are explained in terms of a pool of QB-nonreducing centers existing in a steady-state relationship with QB-reducing centers and with a photochemically silent form of PS II in the thylakoid membrane of D. salina. It is proposed that QB-nonreducing centers are an intermediate stage in the process of damage and repair of PS II. It is further proposed that cells regulate the inflow and outflow of centers from the QB-nonreducing pool to maintain a constant pool size of QB-nonreducing centers in the thylakoid membrane.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- PS:
-
photosystem
- QA :
-
primary quinone electron acceptor of PS II
- QB :
-
secondary quinone electron acceptor of PS II
- LHC:
-
light harvesting complex
- Fo :
-
non-variable fluorescence yield
- Fpl :
-
intermediate fluorescence yield plateau level
- Fmax :
-
maximum fluorescence yield
- Fi :
-
mitial fluorescence yield increase from Fo to Fpl(Fpl-Fo)
- Fv :
-
total variable fluorescence yield (Fmax-Fo)
- DCMU:
-
dichlorophenyl-dimethylurea
References
Anderson JM and Melis A (1983) Localization of different photosystems in separate regions of chloroplast membranes. Proc Natl Acad Sci USA 80: 745–749
Andersson B and Anderson JM (1980) Lateral heterogeneity in the distribution of chlorophyll-protein complexes of the thylakoid membranes of spinach chloroplasts. Biochim Biophys Acta 593: 427–440
Andersson B and Haehnel W (1982) Location of photosystem I and photosystem II reaction centers in different thylakoid regions of stacked chloroplasts. FEBS Lett 146: 13–17
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoxidases in Beta vulgaris. Plant Physiol 24: 1–15
Baker NR and Webber AN (1987) Interactions between photosystems. Adv in Bot Res 13: 2–66
Black MT, Brearley TH and Horton P (1986) Heterogeneity in chloroplast photosystem II. Photosynth Res 89: 193–207
Forbush B and Kok B (1968) Reaction between primary and secondary electron acceptors of photosystem II of photosynthesis. Biochim Biophys Acta 162: 243–253
Graan T and Ort DR (1986) Detection of oxygen-evolving photosystem II centers inactive in plastoquinone reduction. Biochim Biophys Acta 852: 320–330
Greene BA, Staehelin LA and Melis A (1988) Compensatory alterations in the photochemical apparatus of a photoregulatory, chlorophyll b-deficient mutant of maize. Plant Physiol 87: 365–370
Greer DM, Berry JA and Bjorkman O (1986) Photoinhibition of photosynthesis in intact bean leaves: role of light and temperature and requirement for chloroplast protein synthesis during recovery. Planta 168: 253–260
Guenther JE and Melis A (1989) The physiological significance of photosystem II heterogeneity in chloroplast. Photosynth Res 23: 105–109.
Guenther JE, Nemson JA and Melis A (1988) Photosystem stoichiometry and chlorophyll antenna size in Dunaliella salina (green algae). Biochim Biophys Acta 934: 108–117
Hiyama T and Ke B (1972) Difference spectra and extinction coefficients of P700. Biochim Biophys Acta 267: 160–171
Kautsky H and Hirsch A (1934) Das Fluoreszenzverhalten grüner Pflanzen. Biochem Z 274: 422–434
Lavergne J (1982) Two types of primary acceptors in chloroplasts photosystem II. Photobiochem Photobiophys 3: 257–285
Melis A (1985) Functional properties of PS IIβ in spinach chloroplasts. Biochim Biophys Acta 808: 334–342
Melis A and Anderson JM (1983) Structural and functional organization of the photosystems in spinach chloroplasts. Antenna size, relative electron-transport capacity and chlorophyll composition. Biochim Biophys Acta 724: 473–484
Melis A, Guenther JE, Morrissey PJ and Ghirardi ML (1988) Photosystem II heterogeneity in chloroplasts. In: Lichtenthaler HK (ed) Applications of Chlorophyll Fluorescence, pp 33–43. Dordrecht, The Netherlands: Kluwer Academic Publishers
Melis A, Spangfort M and Andersson B (1987) Light-absorption and electron-transport balance between photosystem II and photosystem I in spinach chloroplasts. Photochem Photobiol 45: 129–136
Morrissey PJ, Glick RE and Melis A (1989) Supramolecular assembly and function of subunits associated with the chlorophyll ab light-harvesting complex II (LHC II) in soybean chloroplasts. Plant & Cell Physiol 30: 335–344
Pick U, Karni L and Avron M (1986) Determination of ion content and ion fluxes in halotolerant alga Dunaliella salina. Plant Physiol 81: 92–96
Powles SB (1984) Photoinhibition of photosynthesis induced by visible light. Ann Rev Plant Physiol 35: 15–44
Pulles MPJ, VanGorkom HJ and Verschoor GAM (1976) Primary reactions of photosystem II at low pH 2. Light-induced changes of absorbance and electron-spin resonance in spinach chloroplasts. Biochim Biophys Acta 440: 98–104
Schreiber U and Neubauer C (1987) The polyphasic rise of chlorophyll fluorescence upon onset of strong continuous illumination: II. Partial control by the photosystem II donor side and possible ways of interpretation. Z Naturforsch 42: 1255–1264
Shibata K (1958) Spectrophotometry of biological material. J Biochem (Tokyo) 45: 599–604
Thielen APGM and VanGorkom HJ (1981) Electron transport properties of photosystems IIα and IIβ. In: Akoyunoglou G (ed) Photosynthesis, Proceedings of the 5th International Congress Vol. II. pp 57–64. Balaban International Science Services, Philadelphia, PA
VanGorkom HJ (1974) Identification of the reduced primary electron acceptor of photosystem II as a bound semiquinone anion. Biochim Biophys Acta 347: 439–442
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Guenther, J.E., Melis, A. Dynamics of photosystem II heterogeneity in Dunaliella salina (green algae). Photosynth Res 23, 195–203 (1990). https://doi.org/10.1007/BF00035010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00035010