Abstract
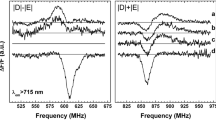
The Photosystem I reaction centre protein CP1, isolated from barley using polyacrylamide gel electrophoresis showed an EPR (Electron Paramgnetic Resonance) spectrum with the polarisation pattern AEEAAE, typical of the primary donor triplet state 3P700, created via radical pair formation and recombination. 3P700 could also be detected by Fluorescence Detected Magnetic Resonance (FDMR) at λf > 700 nm even in the presence of a large number of chlorophyll antennae. Its zero field splitting parameters, D=282.5×10-4 cm-1 and E=38.5×10-4 cm-1, were independent of the detection wavelength, and agreed with ADMR (Absorption Detected Magnetic Resonance) and EPR values. The signs of the 3P700 D+E and D-E transitions were positive (increase in fluorescence intensity on applying a resonance microwave field). In contrast, in the emission band 685 < λf < 700 nm FDMR spectra with negative D+E and D-E transitions were detected, and the D value was wavelength-dependent. These FDMR results support an excitation energy transfer model for CP1, derived from time-resolved fluorescence studies, in which two chlorophyll antenna forms are distinguished, with fluorescence at 685 < λf < 700 nm (inner core antennae, F690), and λf > 700 nm (low energy antenna sites, F720), in addition to the P700. The FDMR spectrum in F690 emission can be interpreted as that of 3P700, observed via reverse singlet excitation energy transfer and added to the FDMR spectrum of the antenna triplet states generated via intramolecular intersystem crossing. This would indicate that reversible energy transfer between F690 and P700 occurs even at 4.2 K.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- CP1:
-
core chlorophyll protein of Photosystem I
- EPR:
-
electron paramagnetic resonance
- F690, F720:
-
chlorophyll forms having fluorescence maximum at 690–695 and 720 nm, respectively
- F(A)(O)DMR:
-
fluorescence (absorption) (optical) detected magnetic resonance
- FF:
-
fluorescence fading
- ISC:
-
intramolecular intersystem crossing
- λf :
-
fluorescence emission wave-length
- LHC I:
-
light harvesting chlorophyll a/b protein of Photosystem I
- P700:
-
primary donor of Photosystem I
- PS I:
-
Photosystem I
- RC:
-
reaction centre
- RP:
-
radical pair
- SDS:
-
sodium dodecyl sulphate
- ZFS:
-
zero field splitting
References
AngerhoferA, Von SchützJU and WolfHC (1984) Fluorescence-ODMR of reaction centers of Rhodopseudomonas viridis. Z Naturforsch 39c: 1085–1090
BonnerjeaJ and EvansMCW (1982) Identification of multiple components in the intermediary electron carrier complex of Photosystem I. FEBS Lett 148: 313–316
BottinH and SetifP (1991) Inhibition of electron transfer from A0 to A1 in Photosystem I after treatment in darkness at low redox potential. Biophys Biochim Acta 1057: 331–336
BrettelK and SetifP (1987) Magnetic-field effects on primary reactions in PS I. Biochim Biophys Acta 893: 109–114
BudilDE and ThurnauerMC (1991) The chlorophyll triplet state as a probe of structure and function in photosynthesis. Biophys Biochim Acta 1057: 1–41
CausgroveTP, YangS and StruveWS (1989) Polarised Pump-probe spectroscopy of Photosystem I antenna excitation transport. J Phys Chem 93: 6844–6850
ChidseyCED, TakiffG, GoldsteinRA and BoxerSG (1985) Effect of magnetic fields on the triplet state lifetime in photosynthetic reaction centers: evidence for thermal repopulation of the initial radical pair. Proc Natl Acad Sci USA 82: 6850–6854
Den BlankenHJ and HoffAJ (1983) High-resolution absorbance difference spectra of the triplet state of the primary donor P700 in Photosystem I subchloroplast particles measured with absorbance-detected magnetic resonance at 1.2 K. Evidence that P700 is a dimeric chlorophyll-complex. Biochim Biophys Acta 724: 52–61
Den BlankenHJ, Van Den ZwetGP and HoffAJ (1982) Study of the long-wavelength fluorescence band at 920 nm of isolated reaction centers of the photosynthetic bacterium Rhodopseudomonas spaeroides R-26 with fluorescence-detected magnetic resonance in zero field. Biochim Biophys Acta 681: 375–382
FrankHA, MacLeanMB and SauerK (1979) Triplet states in Photosystem I of spinach chloroplasts and subchloroplast particles. Proc Natl Acad Sci USA 76: 5124–5128
GolbeckJH (1988) Structure, function and organisation of the Photosystem I reaction center complex. Biochim Biophys Acta 895: 167–204
HalaJ, SearleGFW, SchaafsmaTJ, Van HoekA, PancoskaP, BlahaK and VacekK (1986) Picosecond laser spectroscopy and optically detected magnetic resonance studies on a model photosynthetic system. Photochem Photobiol 44: 527–534
HøjPB and MøllerBL (1986) The 110-kDa reaction center protein of Photosystem I. P700-Chlorophyll a-Protein 1, is an iron-sulfur protein. J Biol Chem 261: 14292–14300
IkegamiI and ItohS (1986) Chlorophyll organisation in P-700-enriched particles isolated from spinach chloroplasts. CD and absorption spectroscopy. Biochim Biophys Acta 851: 75–85
IkegamiI, SetifP and MathisP (1987) Absorption studies of Photosystem I photochemistry in the absence of vit K1. Biochim Biophys Acta 894: 414–422
KuangTY, Argyroudi-AkoyunoglouJH, NakataniHY, WatsonJ and ArntzenCJ (1984) The origin of the long-wavelength fluorescence emission band (77°K) from Photosystem 1. Arch Biochem Biophys 235: 618–627
LemaistreJP and ZewailAH (1979) Fluorescence, phosphorescence and ODMR line narrowing of molecules in solids. Chem Phys Lett 68: 302–308
LutzM and RobertB (1988) Chlorophylls and the photosynthetic membrane. In: SpiroTG (ed) Biological Applications of Raman Spectroscopy, pp 347–411. Wiley, New York
MacholdO, SimpsonDJ and MöllerBL (1979) Chlorophyll-proteins of thylakoids from wild type and mutants of barley (Hordeum vulgare L.) Carlsberg Res Commun 44: 235–254
MathisP and SetifP (1981) Near infra-red absorption spectra of the chlorophyll a cations and triplet state in vitro and in vivo. Isr J Chem 21: 316–320
MimuroM, TamaiN, YamazakiT and YamazakiI (1987a) Excitation energy transfer in spinach chloroplasts. Analysis by the time-resolved fluorescence spectrum at −196°C in the picosecond time range. FEBS Lett 213: 119–122
MimuroM, YamazakiI, TamaiN, YamazakiT and FujitaY (1987b) Analysis of pigment system I Chl a fluorescence at room temperature by the steady state spectrum and the time resolved spectrum in picosecond time range. In: BigginsJ (ed) Progress in Photosynthesis Research, Vol 1, pp 91–94. Martinus Nijhoff Publishers, Dordrecht
MulletJE, BurkeJJ and ArntzenCJ (1980) Chlorophyll proteins of Photosystem I. Plant Physiol 65: 814–822
NechushtaiR, NourizadehSD and ThornberJP (1986) A re-evaluation of the fluorescence of the core chlorophylls of Photosystem I. Biochim Biophys Acta 848: 193–200
RegevA, NechushtaiR, LevanonH and ThornberJP (1989) Time-resolved electron paramagnetic resonance study of the higher plant Photosystem I core complex. J Phys Chem 93: 2421–2426
RutherfordAW and MulletJE (1981) Reaction center triplet states in Photosystem I and Photosystem II. Biochim Biophys Acta 635: 225–235
SchaafsmaTJ (1982) ODMR spectroscopy in photosynthesis. The chlorophyll triplet state in vitro and in vivo. In: ClarkeRH (ed) Triplet State ODMR Spectroscopy, pp 291–365. Wiley, New York
SchellerHV, SvendsenI and MøllerBL (1989) Subunit composition of Photosystem I and identification of center X as a [4Fe-4S] iron-sulfur cluster. J Biol Chem 264: 6929–6934
SearleGFW, KoehorstRBM, SchaafsmaTJ, MøllerBL and Von WettsteinD (1981) Fluorescence detected magnetic resonance (FDMR) spectroscopy of chlorophyll-proteins from barley. Carlsberg Res Commun 46: 183–194
SearleGFW, AVan Hoek and TJ Schaafsma (1983) Picosecond laser spectroscopy and magnetic resonance studies on isolated chlorophyll-proteins. In: DoustTAM and WestMA (eds) Picosecond Chemistry and Biology, pp 35–67. Science Reviews Ltd, Northwood, Middlesex, England
SearleGFW, TamkiviR, Van HoekA and SchaafsmaTJ (1988) Temperature dependence of antennae chlorophyll fluorescence kinetics in Photosystem I reaction centre protein. J Chem Soc Faraday Trans 2: 315–327
SearleGFW, SchaafsmaTJ and AVan Hoek (1990) Picosecond laser spectroscopy and optically detected magnetic resonance on model photosynthetic systems in biopolymers. In: FouassierJ-P and RabekJF (eds) Lasers in Polymer science and Technology, Vol 4 Applications, pp 219–245. CRC Press, Boca Raton, USA
SetifP and BottinH (1989) Identification of electron transfer reactions involving the acceptor A1 of Photosystem I at room temperature. Biochemistry 28: 2689–2697
SetifP, QuaegebeurJ-P and MathisP (1982) Primary processes in Photosystem I. Identification and decay kinetics of the P-700 triplet state. Biochim Biophys Acta 681: 345–353
SetifP, BottinH and MathisP (1985) Absorption studies of primary reaction centers in Photosystem I. Yield and rate of formation of the P-700 triplet state. Biochim Biophys Acta 808: 112–122
ShubinVV, MurphySDS, KarapetyanNV and MohantyP (1991) Origin of the 77 K variable fluorescence at 758 nm in the cyanobacterium Spirulina platensis. Biochim Biophys Acta 1060: 28–36
StanishevskyIV, SchaafsmaTJ and KoehorstRBM (1991) Energy transfer in a rhodamine-porphyrin mixture monitored by optically detected magnetic resonance. Chem Phys Lett 178: 141–146
TelferA, BarberJ, HeathcoteP and EvansMCW (1978) Variable chlorophyll a fluorescence from P-700 enriched Photosystem I particles dependent on the redox state of the reaction centre. Biochim Biophys Acta 504: 153–164
Van Der WaalsJH and De GrootMS (1967) Magnetic interactions related to phosphorescence. In: ZahlanAB (ed) The Triplet State, pp 101–133. Cambridge University Press, Cambridge, England.
Van DorpWG, SchaafsmaTJ, SomaM and Van Der WaalsJH (1973) Investigation of the lowest triplet state of free base porphin by microwave induced changes in its fluorescence. Chem Phys Lett 21: 221–225
Van DorpWG, SchoemakerWH, SomaM and Van Der WaalsJH (1975) The lowest triplet state of free base porphin. Determination of its kinetics of populating and depopulating from microwave-induced transients in the fluorescence intensity. Mol Phys 30: 1701–1721
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Searle, G.F.W., Schaafsma, T.J. Fluorescence detected magnetic resonance of the primary donor and inner core antenna chlorophyll in Photosystem I reaction centre protein: Sign inversion and energy transfer. Photosynth Res 32, 193–206 (1992). https://doi.org/10.1007/BF00034795
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00034795