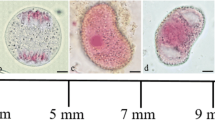
Abstract
Newly released zoospores fromEnteromorpha linza (L.) J. Ag. lack significant cellulose cell wall material and are suitable for treatment as protoplasts in a parasexual fusion process using high pH-Ca+ +, PEG and centrifugation. Treated zoospores settled on glass cover slips within 3 h and were examined microscopically at 1000 ×. Presumptive fusion products were identified by their larger size and presence of twin chloroplasts and eyespots. Unfused zoospores adjacent to fusion cells were killed by 2–3 min exposure to blue light (410–490 nm) from a high pressure mercury illuminator. Unexposed fusion cells developed into uniseriate germlings within 10 days at which stage they could be readily identified at 60 × with a dissecting microscope and isolated by micropipette. Ten-day germlings from both unfused zoospores and fusion cells were stained with the DNA-localizing fluorochrome hydroethidine and relative nuclear DNA content determined with epi-(incident) UV illumination. All germlings were found to be uninucleate. Germlings from unfused zoospores had haploid nuclei with 1N = 10 and 1C and 2C levels of DNA, while germlings from fusion cells had diploid nuclei with 2N = 20 and 2C and 4C levels of DNA. These result are interpreted as evidence of karyogamy following parasexual zoospore fusions. Isolated diploid germlings, cultured for 10 weeks were found to conserve their 2N chromosome complements and elevated levels of nuclear DNA. Although most diploid germlings were morphologically similar to haploid control plants, some exhibited ‘gigas’ characteristics, including larger cells, chloroplasts, and nuclei. These results are discussed in terms of unique phenotypes that result when nuclear and organellar genes are combined in different ways.
Similar content being viewed by others
References
Adamich M, Hemmingsen B (1980) Protoplast and spheroplast production. In Gantt E (ed.), Handbook of Phycological Methods. Cambridge Univ. Press, London, 153–157.
Afonso CL, Harkins KR, Thomas-Compton MA, Krejci AE, Galbraith DW (1985) Selection of somatic hybrid plants inNicotiana through fluorescence-activated sorting of protoplasts. Biotechnology 3: 811–816.
Ahuia MR (1982) Isolation, culture and fusion of protoplasts: problems and prospects. Silvae Genet. 31: 66–77.
Bajaj YPS (1977) Protoplast isolation, culture and somatic hybridization. In Reinert J, Bajaj YPS (eds.), Plant Cell, Tissue and Organ Culture. Springer-Verlag, Berlin, 467–580.
Berliner M (1981) Protoplasts of eukaryotic algae. Int. Rev. Cytol., Suppl. 16: 1–19.
Bliding C (1963) A critical survey of European taxa in Ulvales. Part 1.Capsosiphon, Percursaria, Blidingia, Enteromorpha. Opera Bot. 8: 1–160.
Chen LCM (1986) Cell development ofPorphyra miniata (Rhodophyceae) under axenic culture. Bot. Mar. 29: 435–439.
Cheney D (1984) Genetic modification in seaweeds: applications to commercial utilization and cultivation. In Colwell R, Pariser E, Sinskey A (eds), Biotechnology in the Marine Sciences. Wiley-Interscience, New York, 161–175.
Cheney DF (1986) Genetic engineering in seaweeds: applications and current status. Beih. Nova Hedwigia 81: 22–29.
Cheney DP, Mar E, Saga N, van der Meer J (1986) Protoplast isolation and cell division in the agar-producing seaweedGracilaria (Rhodophyta) J. Phycol. 22: 238–243.
Cocking EC (1960) A method for the isolation of plant protoplasts and vacuoles. Nature 187: 962–963.
Dangeard P (1959) L'Enteromorpha linza. Botaniste 43: 103–118.
Evans DA, Flick CE, Jensen RA (1981) Disease resistance: incorporation into sexually incompatible somatic hybrids of the genusNicotiana. Science 213: 907–909.
Evans DA, Bravo J, Gleba Y (1983) Somatic hybridization: fusion methods, recovery of hybrids and genetic analysis. Int. Rev. Cytol., Suppl. 16: 143–159.
Freshwater DW, Kapraun DF (1986) Field, culture and cytological studies ofPorphyra carolinensis Coll et Cox (Bangiales, Rhodophyta) from North Carolina. Jap. J. Phycol. 34: 251–262.
Fujita Y, Migita S (1987) Fusion of protoplasts from thalli of two different color types inPorphyra yezoensis Ueda and development of fusion products. Jap. J. Phycol. 35: 201–208.
Galbraith DW, Mauch TJ (1980) Identification of fusion of plant protoplasts. II. Conditions for the reproducible fluorescence labelling of protoplasts derived from mesophyll tissue. Z. Pflanzenphysiol. 98: 129–140.
Goff LJ, Coleman AW (1984) Elucidation of fertilization and development in a red alga by quantitative DNA microspectrofluorometry. Devel. Biol. 102: 173–194.
Harkins KR, Galbraith DW (1984) Flow sorting and culture of plant protoplasts. Physiol. Plant. 60: 43–52.
Harms C (1983) Somatic hybridization by plant protoplast fusion. Quart. Rev. Biol. 58: 325–353.
Kao, KN, Michayluk MR (1974) A method of high-frequency intergeneric fusion of plant prototypes. Planta 155: 355–367.
Kao KN, Constable MR, Gamborg OL (1974) Plant protoplast fusion and growth of intergeneric hybrid cells. Planta 120: 215–227.
Kapraun DF (1970) Field and culture studies ofUlva andEnteromorpha in the vicinity of Port Aransas, Texas. Contrib. Mar. Sci. 15: 205–285.
Kapraun DF (1974) Seasonal periodicity and spatial distribution of benthic marine algae in Louisiana. Contrib. Mar. Sci. 18: 139–167.
Kapraun DF, Flynn E (1973) Taxonomic and culture studies ofEnteromorpha linza (L.) J. Ag. AndUlvaria oxysperma (Kuetz.) Bliding (Chlorophyceae, Ulvales) from Central America. Phycologia 12: 145–152.
Kapraun DF, Freshwater DW (1987) Karyological studies of five species in the genusPorphyra (Bangiales, Rhodophyta) from the North Atlantic and Mediterranean. Phycologia 26: 82–87.
Kapraun DF, Martin DJ (1987) Karyological studies of three species ofCodium (Codiales, Chlorophyta) from coastal North Carolina. Phycologia 26: 228–234.
Kapraun DF, Sherman SG (1989) Strain selection and cell isolation ofUlvaria oxysperma (Kuetz.) Bliding for net cultivation. Hydrobiologia. 179: 53–60.
Kapraun DF, Gargiulo GM, Tripodi G (1988) Nuclear DNA and karyotype variation in species ofCodium (Codiales, Chlorophyta) from the North Atlantic. Phycologia 27: 273–282.
Kartha KK, Michayluk MR, Kao KN, Gamborg AL, Constabel F (1974) Callus formation and plant regeneration from mesophyll protoplasts of rape plants (Brassica napus L. cv. Zephyr) Plant Sci. Letters 3: 265–271.
Matagne R, Deltour D, Ledoux L (1979) Somatic fusion between cell wall mutants ofChlamydomonas reinhardtii. Nature (London) 278: 344–346.
Millner PA, Callow ME, Evans LV (1979) Preparation of protoplasts from the green algaEnteromorpha intestinalis (L.) Link. Planta (Berlin) 147: 174–177.
Polne-Fuller M, Gibor A (1987) Tissue culture of seaweeds. In Bird K, Benson PH (eds), Seaweed Cultivation for Renewable Resources. Gas Research Institute, Chicago, IL, 219–239.
Power JB, Cummins SE, Cocking EC (1970) Fusion of isolated plant protoplasts. Nature 225: 1016–1018.
Power JB, Frearson EM, Hayward C, George D, Evans PK, Berry SF, Cocking EC (1976) Somatic hybridization ofPetunia hybrida andPetunia parodii. Nature 263: 500–502.
Primke M, Berger S, Schweiger H (1978) Protoplasts fromAcetabularia: isolation and fusion. Cytobiologie 16: 375–380.
Robinson DG & Schlösser UG (1978) Cell wall regeneration by protoplasts ofChlamydomonas. Planta 141: 83–92.
Saga N, Polne-Fuller M, Gibor A (1986) Protoplasts from seaweeds: production and fusion. Beih. Nova Hedwigia 83: 37–43.
Schieder O, Vasil IK (1980) Protoplast fusion and somatic hybridization. Int. Rev. Cytol. Suppl. 11B: 21–46.
Schlösser UG, Sachs H, Robinson DG (1976) Isolation of protoplasts by means of a ‘species-specific’ autolysine inChlamydomonas. Protoplasma 88: 51–64.
Subrahmanyam NC, Kasha KJ (1973) Selective chromosomal elimination during haploid formation in barley following interspecific hybridization. Chromosoma (Berl.) 42: 111–125.
Torrey JG (1985) The development of plant biotechnology. Am. Scientist 73: 354–363.
Wallin A, Glimelius K, Eriksson T (1974) The induction of aggregation and fusion ofDaucus carota protoplasts by polyethylene glycol. Z. Pflanzenphysiol. 74: 64–80.
Zhang J (1983) Some experiments and observations on the tissue and cell culture ofUndaria pinnatifida. J. Shandong Coll. Oceanology 12: 29–38.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kapraun, D.F. Recovery and development of parasexual fusion products inEnteromorpha linza (L.) J. Ag. (Ulvales, Chlorophyta). J Appl Phycol 1, 239–246 (1989). https://doi.org/10.1007/BF00003649
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00003649