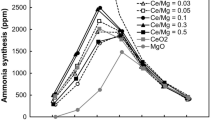
Abstract
To promote deployment of ammonia production system using hydrogen generated by renewable energy, it is necessary for operation of catalytic reactor to vary process parameters, such as temperature, pressure, space velocity and ratio for flow rate of hydrogen to nitrogen, according to the situation of hydrogen supply and forecast of renewable energy supply. Therefore, development of kinetic model that can be applied to various operating conditions is important for design of the ammonia production system. In this chapter, kinetic analysis of ammonia synthesis on ruthenium-based catalysts under high pressure conditions for simulating the reactor situation under varying hydrogen supply is discussed. Various kinetic analysis on iron-based catalysts is considered, and then the reaction rate equation for ruthenium-based catalysts is estimated. In analysis of kinetics of ammonia synthesis over Ru/Cs/MgO catalyst, ammonia production on this catalyst is well explained by the Langmuir–Hinshelwood model. In kinetic analysis for Ru/CeO2, an optimized kinetic model on the catalyst is showed to be different from that on the above-mentioned Ru/Cs/MgO catalyst. And it could be estimated that Ru/CeO2 performs favorable activity as an ammonia synthesis catalyst under high pressure conditions. Additionally, the intermediate surface species (NHx) on Ru/CeO2 is suggested to be contribute to the total rate of ammonia synthesis under high pressure conditions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aika K (2017) Role of alkali promoter in ammonia synthesis over ruthenium catalysts-Effect on reaction mechanism. Catal Today 286:14–20
Reese M, Marquart C, Malmali M, Wagner K, Buchanan E, McCormick A, Cusslerv EL (2016) Performance of a small-scale Haber process. Ind Eng Chem Res 55:3742–3750
Aika K, Ozaki A (1969) Kinetics and isotope effect of ammonia synthesis over an unpromoted iron catalyst. J Catal 13:232–237
Kubota H, Shindo M (1959) The rate expression of high pressure ammonia synthesis in which industrial iron catalyst is employed. Kagakukogaku 23:242–249
Araújo A, Skogestad S (2008) Control structure design for the ammonia synthesis process. Comput Chem Eng 32:2920–2932
Murase A, Roberts HL, Converse AO (1970) Optimal thermal design of an autothermal ammonia synthesis reactor. Ind Eng Chem Proc Des Dev 9:503–513
Nanba T, Javaid R, Matsumoto H (2019) Ammonia synthesis catalyst for storage of renewable hydrogen (in Japanese). Shokubai 61:66–71
Siporin SE, Davis RJ (2004) Use of kinetic models to explore the role of base promoters on Ru/MgO ammonia synthesis catalysts. J Catal 225:359–368
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Javaid, R., Nanba, T., Matsumoto, H. (2023). Kinetic Analysis of Ammonia Production on Ru Catalyst Under High Pressure Conditions. In: Aika, Ki., Kobayashi, H. (eds) CO2 Free Ammonia as an Energy Carrier. Springer, Singapore. https://doi.org/10.1007/978-981-19-4767-4_18
Download citation
DOI: https://doi.org/10.1007/978-981-19-4767-4_18
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-4766-7
Online ISBN: 978-981-19-4767-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)