Abstract
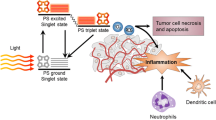
Photodynamic therapy (PDT) is a clinically established modality for the treatment of cancerous and other diseased tissue by localized activation of a photoreactive drug with light to generate cytotoxic reactive oxygen species in targeted lesions. The nature of insult inflicted on tissue by PDT classifies it with cancer therapies including hyperthermia, cryoablation, and electric field ablation, which produce a prompt trauma at the treated site and rapid dramatic changes in tumor microenvironment. This type of insult (regardless that it is induced in the tumor) is sensed as a threat to tissue integrity and homeostasis at the affected site, and provokes canonical responses evolved for dealing with localized injury. Thus the principal protective effector process engaged by the host following tumor PDT is a strong acute inflammatory reaction tasked with isolating the affected area, neutralizing focal source of danger by eliminating injured cells and debris, and securing local healing with the restoration of tissue function. The key event in this response, removal of dead and dying cancer cells, has a critical influence on the subsequent process of the resolution of inflammatory reaction and healing. Even more importantly for therapy outcome, the presence of an overwhelming number of dead cancer cells can overcome the capacity of sequestered professional phagocytes to remove cellular corpses fast enough to avoid breaking immune tolerance and can lead to the development of adaptive immune response against PDT-treated tumor.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
von Tappeiner H (1909) Die photodynamische Erscheinung (Sensibilisierung durch fluoreszierende Stoffe). Monatsschr Kinderheilkd 8:698–741
Dougherty TJ, Grindey GB, Fiel R, Weishaupt KR, Boyle DG (1975) Photodynamic therapy. II. Cure of animal tumors with hematoporphyrin and light. J Natl Cancer Inst 55:115–121
Weishaupt KR, Gomer CJ, Dougherty TJ (1976) Identification of singlet oxygen as the cytotoxic agent in photo-inactivation of a murine tumor. Cancer Res 36:2326–2329
Kelly JF, Snell ME (1976) Hematoporphyrin derivative: a possible aid in the diagnosis and therapy of carcinoma of the bladder. J Urol 115:150–151
Dougherty TJ, Kaufman JE, Goldfarb A, Weishaupt KR, Boyle D, Mittelman A (1978) Photoradiation therapy for the treatment of malignant tumors. Cancer Res 38:2628–2635
Agostinis P, Berg K, Cengel KA, Foster TH, Girotti AW, Gollnick SO, Hahn SM, Hamblin MR, Juzeniene A, Kessel D, Korbelik M, Moan J, Mroz P, Nowis D, Piette J, Wilson BC, Golab J (2011) Photodynamic therapy of cancer; an update. CA Cancer J Clin 61:250–281
Hunag Z (2005) A review of progress in clinical photodynamic therapy. Technol Cancer Res Treat 4:283–294
Brown SB, Brown EA, Walker I (2004) The present and future role of photodynamic therapy in cancer treatment. Lancer Oncol 5:497–508
Palumbo G (2007) Photodynamic therapy and cancer: a brief sightseeing tour. Expert Opin Drug Deliv 4:131–148
Gilbert A, Baggott J (1991) Molecular photophysics. In: Gilbert A (ed) Essentials of molecular photochemistry, CRC , Boca Raton, pp 91–144
Gomer CJ, Razum NJ (1984) Acute skin response in albino mice following porphyrin photosensitization under oxic and anoxic conditions. Photochem Photobiol 40:435–439
Feix JB, Bachowski GJ, Girotti AW (1991) Photodynamic action of merocyanine 540 on erythrocyte membranes: structural perturbation of lipid and protein constituents. Biochim Biophys Acta 1075:28–35
Dougherty TJ, Gomer CJ, Henderson BW, Jori G, Kessel D, Korbelik M, Moan J, Peng Q (1998) Photodynamic therapy. J Natl Cancer Inst 90:889–905
Kessel D, Reiners JJ Jr (2007) Apoptosis and autophagy after mitochondrial or endoplasmic reticulum photodamage. Photochem Photobiol 83:1024–1028
Henderson BW, Dougherty TJ (1992) How does photodynamic therapy work? Photochem Photobiol 55:145–157
Fingar VH (1996) Vascular effects of photodynamic therapy. J Clin Laser Med Surg 14:323–328
Korbelik M (2006) PDT-associated host response and its role in the therapy outcome. Lasers Surg Med 38:500–508
Korbelik M (2008) PDT and inflammation. In: Hamblin MR, Mroz P (eds) Advances in photodynamic therapy: basic, translational and clinical, Artech House, Boston, pp 255–266
Garg AD, Krysko DV, Vandenbeele P, Agostinis P (2011) DAMPs and PDT-mediated photo-oxidative stress; exploring the unknown. Photochem Photobiol Sci 10:670–680
Korbelik M, Merchant S (2011) Involvement of damage-associated molecular patterns in tumor response to photodynamic therapy: surface expression of calreticulin and high-mobility group box-1 release. Cancer Immunol Immunother 60:1431–1437
Korbelik M, Zhang W, Separovic D (2012) Monitoring ceramide and sphingosine-1-phosphate levels in cancer cells and macrophages from tumors treated by photodynamic therapy. Photochem Photobiol Sci 11:779–784
Srikrishna G, Freeza HH (2009) Endogenous damage-associated molecular pattern molecules at the crossroads of inflammation and cancer. Neoplasia 11:615–628
Garg AD, Nowis D, Golab J, Vandenbeele P, Krysko DV, Agostinis P (2010) Immunogenic cell death, DAMPs, and anticancer therapeutics: an emerging amalgamation. Biochim Biophys Acta 1805:53–71
Pober JS, Sessa WC (2007) Evolving functions of endothelial cells in inflammation. Nat Rev Immunol 7:803–815
Krosl G, Korbelik M, Dougherty GJ (1995) Induction of immune cell infiltration into murine SCCVII tumor by photofrin-based photodynamic therapy. Br J Cancer 71:549–555
Sun J, Cecic I, Parkins CS, Korbelik M (2002) Neutrophils as inflammatory and immune effectors in photodynamic therapy-treated mouse SCCVII tumors. Photochem Photobiol Sci 1:690–695
Korbelik M, Cecic I (2003) Mechanism of tumor destruction by photodynamic therapy. In: Nalwa HS (ed) Handbook of photochemistry and photobiology, vol 4, American Scientific Publishers, Stevenson Ranch, pp 39–77
Gollnick SO, Evans SS, Bauman H, Owczarczak B, Maier P, Vaughan L, Wang WC, Unger E, Henderson BW (2003) Role of cytokines in photodynamic therapy-induced local and systemic inflammation. Br J Cancer 88:1772–1779
Castano AP, Mroz P, Hamblin MR (2006) Photodynamic therapy and anti-tumor immunity. Nat Rev Cancer 6:535–545
Krysko DV, D’Herde K, Vandenbeele P (2006) Clearance of apoptotic and necrotic cells and its immunological consequences. Apoptosis 11:1709–1726
deChatelineau AM, Henson PM (2003) The final step in programmed cell death: phagocytes carry apoptotic cells to the grave. Essays Biochem 39:105–117
Peter C, Wesselborg S, Herrmann M, Lauber K (2010) Dangerous attraction: phagocyte recruitment and danger signals of apoptotic and necrotic cells. Apoptosis 15:1008–1028
Ravichandran KS (2011) Beginnings of a good apoptotic meal: the find-me and eat-me signaling pathways. Immunity 35:445–455
Majai G, Petrovski G, Fesüs L (2006) Inflammation and the apopto-phagocytic system. Immunol Lett 104:94–101
Henson PM (2005) Dampening inflammation. Nat. Immunology 6:1179–1181
Golpon HA, Fadok VA, Yaraseviciene-Stewart L, Scerbavicius R, Sauer C, Welte T, Henson PM, Voekel NF (2004) Life after corpse engulfment: phagocytosis of apoptotic cells leads to VEGF secretion and cell growth. FASEB J 18:1716–1718
Gollnick SO, Liu X, Owczarczak B, Musser DA, Henderson BW (1997) Altered expression of interleukin 6 and interleukin 10 as a result of photodynamic therapy in vivo. Cancer Res 57:3904–3909
Gomer CJ, Ferrario A, Luna M, Rucker N, Wong S (2006) Photodynamic therapy: combined modality approaches targeting the tumor microenvironment. Lasers Surg Med 38:516–521
Xiao YQ, Malcolm K, Worthen GS, Gardai S, Schiemann WP, Fadok VA, Bratton DL, Henson PM (2002) Cross-talk between ERK and p38 MAPK mediates selective suppression of pro-inflammatory cytokines by transforming growth factor-β. J Biol Chem 277:14884–14893
Lentsch AB, Shanley TP, Sarma V, Ward PA (1997) In vivo suppression of NF-kappa B and preservation of I kappa B alpha by interleukin-10 and interleukin-13. J Clin Invest 100:2443–2448
Shi Y, Massague J (2003) Mechanisms of TGF-β signaling from cell membrane to nucleus. Cell 113:685–700
Ferrario A, Rucker N, Wong S, Luna M, Gomer CJ (2007) Survivin, a member of the Inhibitor of apoptosis family, is induced by photodynamic therapy and is a target for improving treatment response. Cancer Res 67:4989–4995
Ferrario A, Fisher AM, Rucker N, Gomer CJ (2005) Celecoxib and NS-398 enhance photodynamic therapy by increasing in vitro apoptosis and decreasing in vivo inflammatory and angiogenic factors. Cancer Res 65:9473–9478
Makowski M, Grzela T, Nidrla J, Azarczyk M, Mroz P, Kopee M, Nowis D, Mrowka P, Wasik M, Jakobisiak M, Golab J (2003) Inhibitor of cyclooxygenase-2 indirectly potentiates antitumor effects of photodynamic therapy in mice. Clin Cancer Res 9:5417–5422
Ferrario A, Gomer CJ (2006) Avastin enhances photodynamic therapy treatment of Kaposi’s sarcoma in a mouse tumor model. J Environ Path Tox Oncol 25:251–259
Ferrario A Chantrain CF, von Tiehl K, Buckley S, Rucker N, Shalinsky DR, Shimada H, DeClerck YA, Gomer CJ (2004) The matrix metalloproteinase inhibitor prinomastat enhances photodynamic therapy responsiveness in a mouse tumor model. Cancer Res 64:2328–2332
Merchant S, Huang N, Korbelik M (2010) Expression of complement and pentraxin proteins in acute phase response elicited by tumor photodynamic therapy: The engagement of adrenal hormones. Int Immunopharmacol 10:1595–1601
Gabay C, Kushner I (1999) Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 340:448–454
Korbelik M, Cecic I, Merchant S, Sun J (2008) Acute phase response induction by cancer treatment with photodynamic therapy. Int J Cancer 122:1411–1417
Merchant S, Sun J, Korbelik M (2011) Heat shock protein 70 is acute phase reactant: response elicited by tumor photodynamic therapy. Cell Stress Chaperones 16:153–162
Gallowitsch-Puerta M, Pavlov VA (2007) Neuro-immune interactions via the cholinergic anti-inflammatory pathway. Life Sci 80:2325–2329
Tracey KJ (2009) Reflex control of immunity. Nat Rev Immunol 9:418–428
Yeager MP, Guyre PM, Munck AU (2004) Glucocorticoid regulation of the inflammatory response to injury. Acta Anaestesiol Scand 48:799–813
Yamamoto N, Ngwenya BZ (1987) Activation of mouse peritoneal macrophages by lysophospholipids and ether derivatives of neutral lipids and phospholipids. Cancer Res 47:2008–2013
Korbelik M (2011) Cancer vaccines generated by photodynamic therapy. Photochem Photobiol Sci 10:664–669
Castano AP, Mroz P, Hamblin MR (2006) Photodynamic therapy and anti-tumor immunity. Nat Rev Cancer 6:535–545
Korbelik M, Dougherty GJ (1999) Photodynamic therapy-mediated immune response against subcutaneous mouse tumors. Cancer Res 59:1941–1946
Mroz P, Szokalska A, Wu MX, Hamblin MR (2010) Photodynamic therapy of tumors can lead to development of systemic antigen-specific immune response. PLoS One 5:e15194
Castano AP, Liu Q, Hamblin MR (2006) A green fluorescent protein-expressing murine tumour but not its wild-type counterpart is cured by photodynamic therapy. Br J Cancer 94:391–397
Kabingu E, Oseroff AR, Wilding GE, Gollnick SO (2009) Enhanced systemic immune reactivity to a basal cell carcinoma associated antigen following photodynamic therapy. Clin Cancer Res 15:4460–4466
Korbelik M, Stott B, Sun J (2207) Photodynamic therapy-generated vaccines: relevance of tumour cell death expression. Br J Cancer 97:1381–1387
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Korbelik, M. (2013). Tumor-Localized Insult Delivered by Photodynamic Therapy and the Breakdown of Tumor Immunotolerance. In: Keisari, Y. (eds) Tumor Ablation. The Tumor Microenvironment, vol 5. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-4694-7_7
Download citation
DOI: https://doi.org/10.1007/978-94-007-4694-7_7
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-4693-0
Online ISBN: 978-94-007-4694-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)