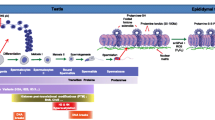
Abstract
The specialized structure of the sperm chromatin has a dual function – first to protect the DNA from damage during storage and transport to the oocyte, and then to enable a rapid and complete unpacking of the undamaged paternal genome in the ooplasm. It is evident that zinc has a pivotal role in maintaining the structural stability and in enabling a rapid decondensation at the appropriate time. It is important for the sperm chromatin structure that the spermatozoa are ejaculated together with the zinc-rich prostatic secretion. Early exposure to zinc-binding seminal vesicular fluid can deplete the sperm chromatin of zinc and most likely induce surplus formation of disulfide bridges, likely to cause incomplete and delayed decondensation of the sperm chromatin in the oocyte. A premature decrease in sperm chromatin structure stability is likely to increase the risk for damage to the DNA due to increased access to the genome for DNA damaging compounds. The status of the sperm chromatin structure can vary in vitro depending on the exposure to zinc-depleting conditions when spermatozoa are stored in semen after ejaculation. When sperm DNA damage tests are evaluated and validated, it is therefore essential to also take into account the dynamics of zinc-dependent and zinc-independent sperm chromatin stability.
An erratum to this chapter is available at http://dx.doi.org/10.1007/978-1-4614-7783-9_12
An erratum to this chapter can be found at http://dx.doi.org/10.1007/978-1-4614-7783-9_12
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Arver S (1982a) Studies on zinc and calcium in human seminal plasma. Acta Physiol Scand 507:1–21
Arver S (1982b) Zinc and zinc ligands in human seminal plasma. III. The principal low molecular weight zinc ligand in prostatic secretion and seminal plasma. Acta Physiol Scand 116(1):67–73
Arver S, Eliasson R (1982) Zinc and zinc ligands in human seminal plasma. II. Contribution by ligands of different origin to the zinc binding properties of human seminal plasma. Acta Physiol Scand 115(2):217–224
Bal W, Dyba M, Szewczuk Z, Jezowska-Bojczuk M et al (2001) Differential zinc and DNA binding by partial peptides of human protamine HP2. Mol Cell Biochem 222(1–2):97–106
Balhorn R (2007) The protamine family of sperm nuclear proteins. Genome Biol 8(9):227
Barney GH, Orgebin-Crist MC, Macapinalac MP (1968) Genesis of esophageal parakeratosis and histologic changes in the testes of the zinc-deficient rat and their reversal by zinc repletion. J Nutr 95(4):526–534
Bench G, Corzett MH, Kramer CE et al (2000) Zinc is sufficiently abundant within mammalian sperm nuclei to bind stoichiometrically with protamine 2. Mol Reprod Dev 56(4):512–519
Björndahl L, Kvist U (1985) Loss of an intrinsic capacity for human sperm chromatin decondensation. Acta Physiol Scand 124(2):189–194
Björndahl L, Kvist U (1990) Influence of seminal vesicular fluid on the zinc content of human sperm chromatin. Int J Androl 13(3):232–237
Björndahl L, Kvist U (2003) Sequence of ejaculation affects the spermatozoon as a carrier and its message. Reprod Biomed Online 7(4):440–448
Björndahl L, Kvist U (2010) Human sperm chromatin stabilization: a proposed model including zinc bridges. Mol Hum Reprod 16(1):23–29
Björndahl L, Kjellberg S, Kvist U (1991) Ejaculatory sequence in men with low sperm chromatin-zinc. Int J Androl 14(3):174–178
Calvin HI, Bedford JM (1971) Formation of disulphide bonds in the nucleus and accessory structures of mammalian spermatozoa during maturation in the epididymis. J Reprod Fertil Suppl 13(Suppl 13):65–75
Calvin HI, Bleau G (1974) Zinc-thiol complexes in keratin-like structures of rat spermatozoa. Exp Cell Res 86(2):280–284
Calvin HI, Yu CC, Bedford JM (1973) Effects of epididymal maturation, zinc (II) and copper (II) on the reactive sulfhydryl content of structural elements in rat spermatozoa. Exp Cell Res 81(2):333–341
Fisch H, Lambert SM, Goluboff ET (2006) Management of ejaculatory duct obstruction: etiology, diagnosis, and treatment. World J Urol 24(6):604–610. doi:10.1007/s00345-006-0129-4
From Björk M, Björndahl L, Zakeri A et al (2009) Can sperm chromatin packaging influence results of sperm DNA integrity measures? Results of the TUNEL assay. J Androl March/April Supplement: 34th annual meeting of the American Society of Andrology, Philadelphia, p 40
Gusse M, Sautiere P, Belaiche D et al (1986) Purification and characterization of nuclear basic proteins of human sperm. Biochim Biophys Acta 884(1):124–134. doi:0304-4165(86)90235-7 [pii]
Kjellberg S (1993) Zinc and human sperm chromatin. Ph.D. thesis, University of Linköping, Linköping
Kvist U (1980a) Importance of spermatozoal zinc as temporary inhibitor of sperm nuclear chromatin decondensation ability in man. Acta Physiol Scand 109(1):79–84
Kvist U (1980b) Sperm nuclear chromatin decondensation ability. An in vitro study on ejaculated human spermatozoa. Acta Physiol Scand 486:1–24
Kvist U, Björndahl L (1985) Zinc preserves an inherent capacity for human sperm chromatin decondensation. Acta Physiol Scand 124(2):195–200
Kvist U, Eliasson R (1978) Zinc dependent chromatin stability in human ejaculated spermatozoa. Int J Androl Suppl 1:178
Kvist U, Afzelius BA, Nilsson L (1980) The intrinsic mechanism of chromatin decondensation ability and its activation in human spermatozoa. Dev Growth Differ 22:543–554
Kvist U, Björndahl L, Roomans GM et al (1985) Nuclear zinc in human epididymal and ejaculated spermatozoa. Acta Physiol Scand 125(2):297–303
Lundquist F (1949) Aspects of the biochemistry of human semen. Acta Physiol Scand 19(Suppl 66):1–108
Pettersson G, From Björk M, Björndahl L et al (2009) Can sperm chromatin packaging influence results of sperm DNA integrity measures? Results from acridin orange flow cytometry. J Androl March/April Supplement: 9th International Congress of Andrology, Barcelona: pp 108–109
Roomans GM, Lundevall E, Björndahl L et al (1982) Removal of zinc from subcellular regions of human spermatozoa by EDTA treatment studied by X-ray microanalysis. Int J Androl 5(5):478–486
Tu J, Björndahl L, Kvist U (2009) Can sperm chromatin packaging influence results of sperm DNA integrity measures? Results of the Comet assay. J Androl March/April Supplement: 34th annual meeting of the American Society of Andrology, Philadelphia:42
Ward WS (2010) Function of sperm chromatin structural elements in fertilization and development. Mol Hum Reprod 16(1):30–36
World Health Organization (2010) WHO laboratory manual for the examination and processing of human semen, 5th edn. World Health Organization, Geneva
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Björndahl, L., Kvist, U. (2014). Structure of Chromatin in Spermatozoa. In: Baldi, E., Muratori, M. (eds) Genetic Damage in Human Spermatozoa. Advances in Experimental Medicine and Biology, vol 791. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-7783-9_1
Download citation
DOI: https://doi.org/10.1007/978-1-4614-7783-9_1
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-7782-2
Online ISBN: 978-1-4614-7783-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)