Abstract
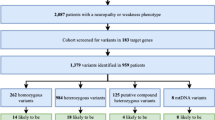
The aim of the study was to develop a panel for detecting heterozygous carriage of frequent mutations associated with phenylketonuria (PKU), and to determine their allelic frequencies in one of the regions of Russia. PKU is one of the most common monogenic diseases with autosomal recessive inheritance. Carrier screening leads to a reduction in the number of children born with PKU. The diagnostic panel included 23 pathogenic variants of the PAH gene nucleotide sequence. Participants of the study (n = 642) were randomly selected from an ESSE-Vologda population-based study. Genotyping was performed using real-time polymerase chain reaction on a QuantStudio 12K Flex Real-Time PCR System (Thermo Fisher Scientific, United States). The data were analyzed using the TaqMan Genotyper Software (Thermo Fisher Scientific, United States). Seventeen heterozygous carriers of six variants of the nucleotide sequence associated with the development of PKU were identified: R408W (rs5030858), A403V (rs5030857), I306V (rs62642934), L48S (rs5030841), IVS12+1G>A (rs5030861), R261Q (rs5030849_C_T). The frequency of heterogyzotes in the Russian population was 2.65% (95% CI 1.55–4.21), or 1 : 38. The data obtained indicate the prospects of mass screening for the carriage of frequent mutations associated with PKU in people with an uncomplicated family history. The developed diagnostic panel allows one to rapidly obtain the result of genetic analysis and can be used for PKU carrier screening.

Similar content being viewed by others
REFERENCES
Blau, N., van Spronsen, F.J., and Levy, H.L., Phenylketonuria, Lancet, 2010, vol. 376, no. 9750, pp. 1417–1427. https://doi.org/10.1016/S0140-6736(10)60961-0
Volgina, S.Ya., Yafarova, S.Sh., and Kletenkova, G.R., Phenylketonuria in children: Modern aspects of pathogenesis, clinic, treatment, Ross. Vestn. Perinatol. Pediatr., 2017, vol. 62, no. 5, pp. 111–118. https://doi.org/10.21508/1027-4065-2017-62-5-111-118
PAHvdb: Phenylalanine Hydroxylase Gene Locus-Specific Database. http://www.biopku.org/home/ pah.asp. Accessed February 5, 2020.
Zschocke, J., Phenylketonuria mutations in Europe, Hum. Mutat., 2003, vol. 21, no. 4, pp. 345–356. https://doi.org/10.1002/humu.10192
Gundorova, P., Kuznetsova, I.A., Kutsev, S.I., Golikhina, T.A., Aksyanova, Kh.F., Nenasheva, S.A., et al., Results of genotyping program of PKU and HPA patients, Med. Genet., 2018, vol. 17, no. 12, pp. 14–24.
Gundorova, P., Stepanova, A.A., Shchagina, O.A., and Polyakov, A.V., Results of the new medical technologies “Detection of major PAH gene point mutations by multuplex ligase reaction” and “Detection of ten additional mutations in PAH gene by multuplex ligase reaction” application in the DNA-diagnostics of phenylketonuria, Med. Genet., 2016, vol. 15, no. 2, pp. 29–36.
Amelina, M.A., Stepanova, A.A., Polyakov, A.V., Amelina, S.S., and Zinchenko, R.A., Spectrum and frequency of mutations in RAH gene in patients with phenylketonuria from Rostov region, Med. Genet., 2015, vol. 14, no. 8, pp. 30–36.
Amelina, M.A., Zinchenko, R.A., Stepanova, A.A., Gundorova, P., Polyakov, A.V., Amelina, S.S., et al., Examine the relationship genotypes (PAH) and phenotype in patients with phenylketonuria Rostov region, Med. Genet., 2016, vol. 15, no. 6, pp. 3–10.
Nikiforova, A.I., Abramov, D.D., Kadochnikova, V.V., Zobkova, G.U., Ogurtsova, K.A., Bryukhanova, N.O., et al., Determining the frequency of PAH mutations in Moscow region residents with phenylketonuria using a combination of Real-time PCR and Next-Generation sequencing, Bull. Russ. State Med. Univ., 2017, no. 4, pp. 38–44.
Abramov, D.D., Kadochnikova, V.V., Yakimova, E.G., Belousova, M.V., Maerle, A.V., Sergeev, I.V., et al., High carrier frequency of CFTR gene mutations associated with cystic fibrosis, and PAH gene mutations associated with phenylketonuria in Russian population, Bull. Russ. State Med. Univ., 2015, no. 4, pp. 32–35.
Zobkova, G.Yu., Kadochnikova, V.V., Abramov, D.D., Donnikov, A.E., and Demikova, N.S., Determination of the carrier frequency of mutations in the CFTR, PAH, GALT and GJB2 genes among 2168 individuals without clinical signs of hereditary diseases, Med. Genet., 2019, vol. 18, no. 10, pp. 30–35. https://doi.org/10.25557/2073-7998.2019.10.30-35
Broccanello, C., Gerace, L., and Stevanato, P., QuantStudio 12K Flex OpenArray System as a tool for high-throughput genotyping and gene expression analysis, Methods Mol. Biol., 2020, vol. 2065, pp. 199–208. https://doi.org/10.1007/978-1-4939-9833-3_15
Anichkina, A.A., Gavrilyuk, A.P., Tverskaya, S.M., and Polyakov, A.V., Analysis of most prevalent mutations of PAH gene in phenylketonuria patients, Med. Genet., 2003, vol. 2, no. 4, pp. 175–181.
Stepanova, A.A., Tverskaya, S.M., Zinchenko, R.A., and Polyakov, A.V., Molecular genetic study of phenylalanine hydroxylase gene in a group of Russian PKU-patients, Med. Genet., 2006, vol. 5, no. 2 (44), pp. 32–39.
Ross, J.P., Mohtashami, S., Leveille, E., Johnson, A.M., Xiong, L., Dion, P.A., et al., Association study of essential tremor genetic loci in Parkinson’s disease, Neurobiol. Aging, 2018, vol. 66, pp. 178-e13–178-e15. https://doi.org/10.1016/j.neurobiolaging.2018.01.001
Gnani, D., Romito, I., Artuso, S., Chierici, M., De Stefanis, C., Panera, N., et al., Focal adhesion kinase depletion reduces human hepatocellular carcinoma growth by repressing enhancer of zeste homolog 2, Cell Death Differ., 2017, vol. 24, no. 5, pp. 889–902. https://doi.org/10.1038/cdd.2017.34
Martins, F.T., Ramos, P.Z., Svidnicki, M.C., Castilho, A.M., and Sartorato, E.L., Optimization of simultaneous screening of the main mutations involved in non-syndromic deafness using the TaqMan OpenArray Genotyping platform, BMC Med. Genet., 2013, vol. 14, no. 1, p. 112. https://doi.org/10.1186/1471-2350-14-112
Blau, N., Shen, N., and Carducci, C., Molecular genetics and diagnosis of phenylketonuria: State of the art, Expert Rev. Mol. Diagn., 2014, vol. 14, no. 6, pp. 655–671. https://doi.org/10.1586/14737159.2014.923760
Tsybakova, N.Yu., Sokolenko, A.P., Ievleva, A.G., Suspitsin, E.N., and Imyanitov, E.N., BRCA1, CHEK2, NBS1, CFTR, PAH, and CX26 founder mutations in healthy female residents of St. Petersburg, Transfusiology, 2011, vol. 12, pp. 1329–1341.
Bercovich, D., Elimelech, A., Zlotogora, J., Korem, S., Yardeni, T., Gal, N., et al., Genotype–phenotype correlations analysis of mutations in the phenylalanine hydroxylase (PAH) gene, J. Hum. Genet., 2008, vol. 53, no. 5, pp. 407–418. https://doi.org/10.1007/s10038-008-0264-4
Dordević, M., Klaassen, K., Sarajlija, A., Tosic, N., Zukic, B., Kecman, B., et al., Molecular genetics and genotype-Ba sed estimation of BH4-responsiveness in Serbian PKU patients: Spotlight on phenotypic implications of p. L48S, JIMD Rep., 2013, vol. 9, pp. 49–58. https://doi.org/10.1007/8904_2012_178
Eiken, H.G., Knappskog, P.M., Motzfeldt, K., Boman, H., and Apold, J., Phenylketonuria genotypes correlated to metabolic phenotype groups in Norway, Eur. J. Pediatr., 1996, vol. 155, no. 7, pp. 554–560. https://doi.org/10.1007/BF01957904
Kulikov, A.Yu. and Rybchenko, Yu.V., Pharmacoeconomic analysis of the use of the kuvan drug product in phenylketonuric patients, Pharmacoecon.: Theory Pract., 2015, vol. 3, no. 1, pp. 17–19.
Klinicheskie rekomendatsii “Fenilketonuriya i narusheniya obmena tetragidroboipterina u detei” (Clinical Recommendations “Phenylketonuria and Metabolic Imbalance of Tetrahydrobiopterin in Children”), Moscow: Ministry of Healthcare of the Russian Federation, 2017. http://www.pediatr-russia.ru/sites/default/files/ file/kr_fen.pdf.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflicts of interest.
Statement of compliance with standards of research involving humans as subjects. This article does not contain any studies involving animals as subjects. All procedures in studies involving human participants were carried out in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all participants involved in the study.
Additional information
Translated by D. Novikova
ADDITIONAL INFORMATION
Kurilova O.V.: https://orcid.org/0000-0003-3082-5161; e‑mail: olga_kurilova81@mail.ru
Klimushina M.V.: https://orcid.org/0000-0002-7876-9325; e-mail: mklimushina@gmail.com
Kiseleva A.V.: https://orcid.org/0000-0003-4765-8021; e‑mail: sanyutabe@gmail.com
Ershova A.I.: https://orcid.org/0000-0001-7989-0760; e‑mail: alersh@mail.ru
Sotnikova E.A.: https://orcid.org/0000-0002-8395-4146; e-mail: sotnikova.evgeniya@gmail.com
Divashuk M.G.: https://orcid.org/0000-0001-6221-3659; e-mail: divashuk@gmail.com
Khlebus E.Yu.: https://orcid.org/0000-0002-8355-8960; e-mail: elkhlebus@gmail.com
Skirko O.P.: https://orcid.org/0000-0003-3755-0279; e‑mail: ops_70@mail.ru
Efimova I.A.: https://orcid.org/0000-0002-3081-8415; e‑mail: biobank@gnicpm.ru
Shalnova S.A.: https://orcid.org/0000-0003-2087-6483; e‑mail: sshalnova@gnicpm.ru
Slominsky P.A.: https://orcid.org/0000-0003-3530-0655; e-mail: paslominsky@bk.ru
Meshkov A.N.: https://orcid.org/0000-0001-5989-6233; e-mail: meshkov@lipidclinic.ru
Drapkina O.M.: https://orcid.org/0000-0002-4453-8430; e-mail: odrapkina@gnicpm.ru
About this article
Cite this article
Kurilova, O.V., Klimushina, M.V., Kiseleva, A.V. et al. The Frequency of Heterozygous Carriage of the PAH Gene Nucleotide Sequence Variants Associated with the Development of Phenylketonuria in the ESSE-Vologda Population-Based Cohort Study. Mol. Genet. Microbiol. Virol. 36, 92–99 (2021). https://doi.org/10.3103/S0891416821020038
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0891416821020038