Abstract
Background and objective: Venlafaxine and its major active metabolite O-desmethylvenlafaxine selectively inhibit serotonin and norepinephrine reuptake from the synaptic gap. The inhibition of norepinephrine uptake is assumed to enhance antidepressant efficacy when venlafaxine is given at higher therapeutic doses. Thus investigation of the concentration-response relationship of noradrenergic effects is of clinical interest. We used pupillography as a test system for the pharmacodynamic response to venlafaxine, since it had been shown to be useful for assessment of noradrenergic effects on the autonomous nervous system. The aim of the study was to develop a pharmacokinetic/pharmacodynamic model by means of nonlinear mixed-effects modelling in order to describe the time course of the noradrenergic response to venlafaxine.
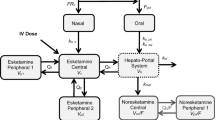
Subjects and methods: Twelve healthy male subjects received venlafaxine 37.5 mg or placebo orally twice daily for 7 days and subsequently 75 mg or placebo twice daily for another 7 days. After a 14-day washout phase, the two groups were crossed over. After the last dose of venlafaxine or placebo on day 14, blood samples were drawn to determine venlafaxine and O-desmethylvenlafaxine concentrations and the amplitude and recovery time of the pupillary light reflex were measured. A pharmacokinetic/pharmacodynamic model was developed to describe the data using nonlinear mixed-effects modelling.
Results: The pharmacokinetic part of the model could be simultaneously fitted to both venlafaxine and O-desmethylvenlafaxine data, yielding precise parameter estimates that were similar to published data. The model detected high variability of the intrinsic clearance of venlafaxine (94.8%), most likely due to cytochrome P450 2D6 polymorphism. Rapid development of tolerance of the pupillary light reflex parameters was seen and could be successfully accounted for in the pharmacodynamic part of the model. The half-life of development and regression of tolerance was estimated to be 30 minutes for the amplitude and 40 minutes for the recovery time.
Conclusion: The time course of the effect and the concentration-response relationship were successfully described by a pharmacokinetic/pharmacodynamic model that takes into account the rapid development of tolerance of pupillary light reflex parameters. This provides a basis for further investigations of the applicability of pupillography as a surrogate measurement of the effectivity of antidepressant drugs with norepinephrine reuptake-inhibiting properties.










Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References:
Muth EA, Haskins JT, Moyer JA, et al. Antidepressant biochemical profile of the novel bicyclic compound Wy-45,030, an ethyl cyclohexanol derivative. Biochem Pharmacol 1986 Dec; 35(24): 4493–7
Harvey AT, Rudolph RL, Preskorn SH. Evidence of the dual mechanisms of action of venlafaxine. Arch Gen Psychiatry 2000 May; 57(5): 503–9
Lecrubier Y. Clinical utility of venlafaxine in comparison with other antidepressants. Int Clin Psychopharmacol 1995 Mar; 10 Suppl. 2: 29–35
Bitsios P, Szabadi E, Bradshaw CM. Comparison of the effects of venlafaxine, paroxetine and desipramine on the pupillary light reflex in man. Psychopharmacology (Berl) 1999 Apr; 143(3): 286–92
Matoga M, Pehourcq F, Titier K, et al. Rapid high-performance liquid chromatographic measurement of venlafaxine and O-desmethylvenlafaxine in human plasma: application to management of acute intoxications. J Chromatogr B Biomed Sci Appl 2001 Sep; 760(2): 213–8
Siepmann T, Ziemssen T, Mueck-Weymann M, et al. The effects of venlafaxine on autonomic functions in healthy volunteers. J Clin Psychopharmacol 2007 Dec; 27(6): 687–91
Taft DR, Iyer GR, Behar L, et al. Application of a first-pass effect model to characterize the pharmacokinetic disposition of venlafaxine after oral administration to human subjects. Drug Metab Dispos 1997 Oct; 25(10): 1215–8
Howell SR, Husbands GE, Scatina JA, et al. Metabolic disposition of 14C-venlafaxine in mouse, rat, dog, rhesus monkey and man. Xenobiotica 1993 Apr; 23(4): 349–59
Troy SM, Schultz RW, Parker VD, et al. The effect of renal disease on the disposition of venlafaxine. Clin Pharmacol Ther 1994 Jul; 56(1): 14–21
Gordi T, Xie R, Huong NV, et al. A semiphysiological pharmacokinetic model for artemisinin in healthy subjects incorporating autoinduction of metabolism and saturable first-pass hepatic extraction. Br J Clin Pharmacol 2005 Feb; 59(2): 189–98
Rousseau A, Leger F, Le MY, et al. Population pharmacokinetic modeling of oral cyclosporin using NONMEM: comparison of absorption pharmacokinetic models and design of a Bayesian estimator. Ther Drug Monit 2004 Feb; 26(1): 23–30
Parker VD, Richards LS, Nichols AI, et al. The absolute bioavailability of an oral sustained-release formulation of desvenlafaxine succinate in healthy subjects. Clin Pharmacol Ther 2005 Feb; 77(2): P47
Porchet HC, Benowitz NL, Sheiner LB. Pharmacodynamic model of tolerance: application to nicotine. J Pharmacol Exp Ther 1988 Jan; 244(1): 231–6
Sheiner LB, Stanski DR, Vozeh S, et al. Simultaneous modeling of pharmacokinetics and pharmacodynamics: application to d-tubocurarine. Clin Pharmacol Ther 1979 Mar; 25(3): 358–71
Muth EA, Moyer JA, Haskins JT, et al. Biochemical, neurophysiological, and behavioral-effects of Wy-45,233 and other identified metabolites of the antidepressant venlafaxine. Drug Dev Res 1991; 23(2): 191–9
Karlsson MO, Savic RM. Diagnosing model diagnostics. Clin Pharmacol Ther 2007 Jul; 82(1): 17–20
Hooker A, Karlsson MO. Conditional weighted residuals: a diagnostic to improve population PK/PD model building and evaluation. AAPS PharmSci 2005; 7 Suppl. 2: W5321
Holford NH. The visual predictive check: superiority to standard diagnostic (Rorschach) plots [abstract]. Seventeenth Meeting of the Population Approach Group in Europe; 2008 Jun 18–20; Marseilles [online]. Available from URL: http://www.page-meeting.org/?.abstract=738 [Accessed 2008 Aug 21]
Effexor (venlafaxine hydrochloride) tablets: US prescribing information. Philadelphia (PA): Wyeth Pharmaceuticals Inc., 2007 [online]. Available from URL: http://www.fda.gov/cder/foi/label/2007/020151s043,020699s069lbl.pdf [Accessed 2007 Jul 11]
Klamerus KJ, Maloney K, Rudolph RL, et al. Introduction of a composite parameter to the pharmacokinetics of venlafaxine and its active O-desmethyl metabolite. J Clin Pharmacol 1992 Aug; 32(8): 716–24
Savic R, Jonker Dl, Kerbusch T, et al. Implementation of a transit compartment model for describing drug absorption in pharmacokinetic studies. J Pharmacokinet Pharmacodyn 2007 Oct; 34(5): 711–26
Shams ME, Arneth B, Hiemke C, et al. CYP2D6 polymorphism and clinical effect of the antidepressant venlafaxine. J Clin Pharm Ther 2006 Oct; 31(5): 493–502
Amchin J, Ereshefsky L, Zarycranski W, et al. Effect of venlafaxine versus fluoxetine on metabolism of dextromethorphan, a CYP2D6 probe. J Clin Pharmacol 2001 Apr; 41(4): 443–51
Fukuda T, Nishida Y, Zhou Q, et al. The impact of the CYP2D6 and CYP2C19 genotypes on venlafaxine pharmacokinetics in a Japanese population. Eur J Clin Pharmacol 2000 May; 56(2): 175–80
Fukuda T, Yamamoto I, Nishida Y, et al. Effect of the CYP2D6*10 genotype on venlafaxine pharmacokinetics in healthy adult volunteers. Br J Clin Pharmacol 1999 Apr; 47(4): 450–3
Grozinger M, Dragicevic A, Hiemke C, et al. Melperone is an inhibitor of the CYP2D6 catalyzed O-demethylation of venlafaxine. Pharmacopsychiatry 2003 Jan; 36(1): 3–6
Otton SV, Ball SE, Cheung SW, et al. Venlafaxine oxidation in vitro is catalysed by CYP2D6. Br J Clin Pharmacol 1996 Feb; 41(2): 149–56
Veefkind AH, Haffmans PM, Hoencamp E. Venlafaxine serum levels and CYP2D6 genotype. Ther Drug Monit 2000 Apr; 22(2): 202–8
Moyer JA, Muth EA, Haskins J, et al. In vivo antidepressant profiles of the novel bicyclic compounds Wy-45.030 and Wy-45.881 [abstract no. 10]. 14th Annual Meeting of the Society for Neuroscience; 1984 Oct 10–15; Anaheim (CA)
Franklin M, Clement EM, Campling G, et al. Effect of venlafaxine on pineal melatonin and noradrenaline in the male rat. J Psychopharmacol 1998; 12(4): 371–4
Ekblom M, Hammarlund-Udenaes M, Paalzow L. Modeling of tolerance development and rebound effect during different intravenous administrations of morphine to rats. J Pharmacol Exp Ther 1993 Jul; 266(1): 244–52
Shi J, Benowitz NL, Denaro CP, et al. Pharmacokinetic-pharmacodynamic modeling of caffeine: tolerance to pressor effects. Clin Pharmacol Ther 1993 Jan; 53(1): 6–14
Acknowledgements
The authors would like to thank Frederike Lentz, Silke Dittberner and Anne Drescher for their valuable suggestions during the model building and their careful proofreading of the manuscript. The reference compounds for analytical determination of venlafaxine and O-desmethylvenlafaxine were kindly provided by Wyeth Pharmaceuticals, Münster, Germany. No other sources of funding were used to assist in the preparation of this study. The authors have no conflicts of interest that are relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lindauer, A., Siepmann, T., Oertel, R. et al. Pharmacokinetic/Pharmacodynamic Modelling of Venlafaxine. Clin Pharmacokinet 47, 721–731 (2008). https://doi.org/10.2165/00003088-200847110-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200847110-00003